Clear Sky Science · en
Numerical investigation of ultrasound-induced acoustic streaming and shear stress for blood clot manipulation
Why breaking clots without drugs matters
Blood clots in veins and brain vessels can block circulation and cause stroke, heart attack, or dangerous lung clots. Today’s main treatments rely on blood-thinning or clot-dissolving drugs, which can save lives but also raise the risk of serious bleeding and are not suitable for every patient. This study explores a drug-free, non-invasive alternative: using carefully tuned ultrasound to stir the fluid around a clot so strongly that the clot starts to break apart on its own.
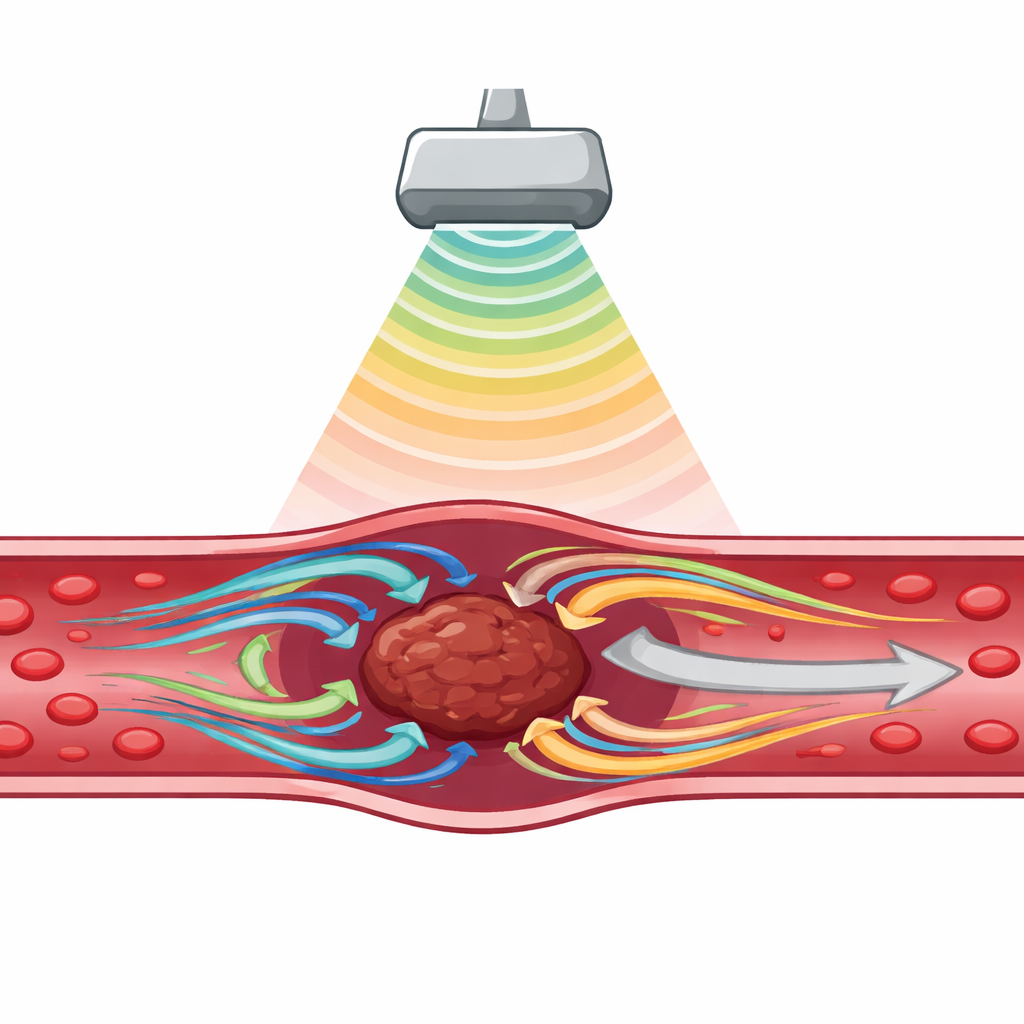
Using sound to push on a hidden blockage
The researchers focused on a subtle effect called acoustic streaming. When an ultrasound beam passes through fluid, it does not just send waves; it can also drive a slow but steady flow, like a gentle underwater wind. Near an obstruction such as a blood clot, this flow can whirl into tiny vortices that drag along the clot surface and create a sideways pulling force known as shear stress. If that stress exceeds the clot’s mechanical strength, the clot’s internal fibers can rupture and the mass begins to fragment. Instead of adding drugs to weaken the clot, the team asked whether streaming alone, created by realistic ultrasound settings, could reach those stress levels.
Building a digital blood vessel
To answer this, the authors constructed a detailed computer model of a blood vessel with a clot inside, using the simulation software COMSOL Multiphysics. They represented the vessel as a two-dimensional tube and the clot as an elliptical region with thick, viscous fluid-like behavior. An ultrasound source, modeled as a straight transducer above the clot, emitted continuous sound waves into the vessel. By coupling two sets of equations—one describing how sound waves travel and another describing how fluids flow—they calculated how the ultrasound field generated streaming around the clot and how much shear stress appeared on its surface under different conditions.
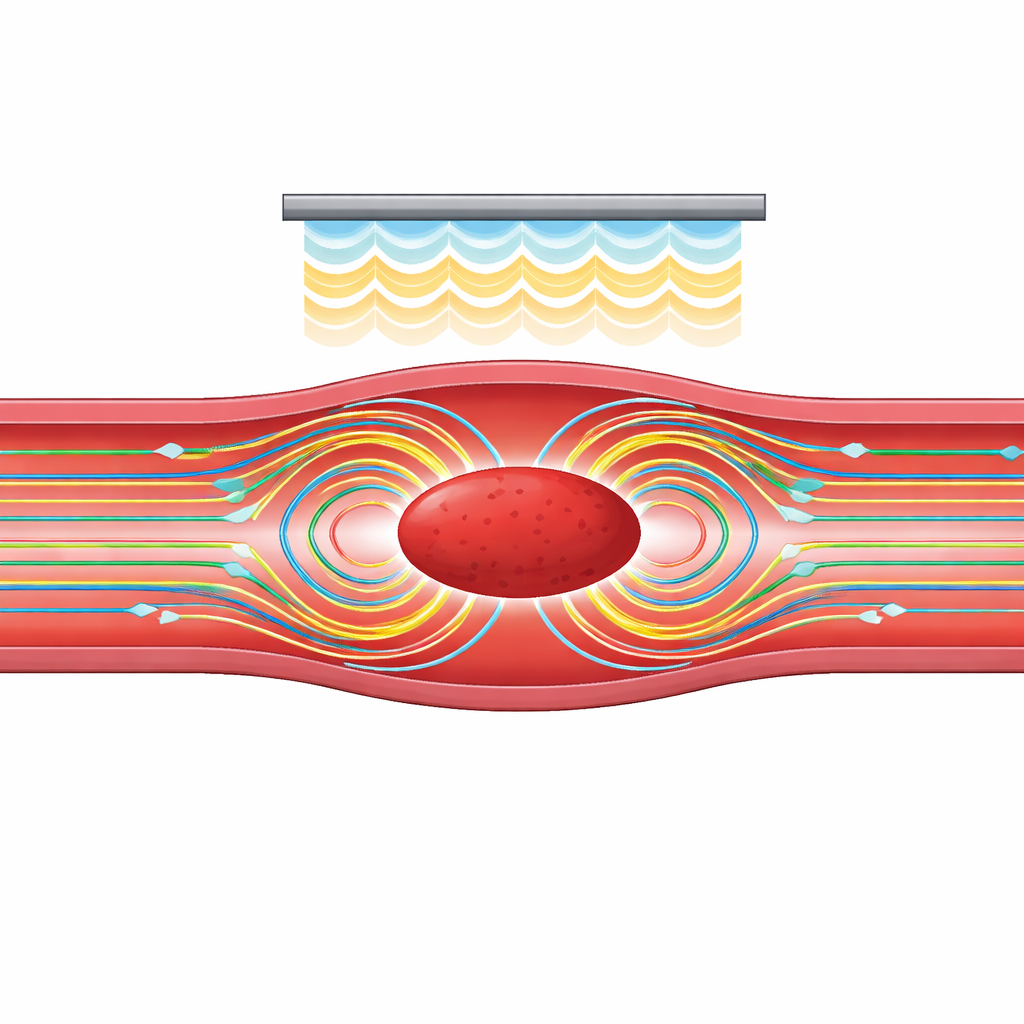
Finding the right sound settings
The team systematically varied three key factors: where the clot sat relative to the transducer, the frequency of the ultrasound, and the strength (pressure) of the sound waves. They found that streaming and shear stress changed in a complex, non-linear way as the clot moved along the vessel, a signature of standing wave patterns formed by incoming and reflected sound. At certain positions, strong vortices formed on both sides of the clot and created intense stresses; at others, the flow was much weaker. Frequency also mattered. Very low frequencies penetrated well but risked unwanted side effects such as uncontrolled bubble activity, while very high frequencies were quickly absorbed and mostly turned into heat. Around 2 MHz, the model predicted a sweet spot where the streaming remained strong without excessive absorption, making it particularly attractive for therapy.
How hard the sound must push
By gradually increasing acoustic pressure, the researchers showed that shear stress on the clot surface rose steadily and then began to level off as viscous resistance in the fluid balanced the driving force. Under optimized conditions—about 2 MHz and 2 MPa acoustic pressure with the clot at a favorable location—the computed shear stress peaked at roughly 10.9 pascals, more than double an estimated threshold of 4.1 pascals needed to start breaking the clot’s internal network. The model also explored a more realistic scenario where the vessel wall was thicker, as might happen in disease. In that case, more of the sound was lost before reaching the clot, and the initial shear stress fell to about 2.7 pascals. Modestly increasing the pressure could raise it to about 3.0 pascals, but still below the breakup threshold, underscoring how body tissues between the skin and the vessel can blunt the effect.
Next steps toward safer clot treatment
Overall, the simulations suggest that ultrasound-driven streaming can, in principle, generate enough mechanical stress to fragment clots without using clot-dissolving drugs, provided the sound settings and geometry are favorable. At the same time, the work highlights important caveats: the current model uses simplified vessel and clot structures, assumes no background blood flow, and even places the transducer inside the vessel rather than on the skin. The authors argue that more realistic three-dimensional models, deformable vessel walls, moving blood, and experimental tests are needed. Still, their results map out promising ranges of frequency, pressure, and positioning, and point to acoustic streaming as a potential building block for future, safer ultrasound-based clot therapies.
Citation: Hisham, A., Hassan, M.A. & Wahba, A.A. Numerical investigation of ultrasound-induced acoustic streaming and shear stress for blood clot manipulation. Sci Rep 16, 12891 (2026). https://doi.org/10.1038/s41598-026-44521-5
Keywords: ultrasound clot therapy, acoustic streaming, blood thrombus, shear stress, computational modeling