Clear Sky Science · en
Integrated machine learning and multi-omics analysis identifies ALOX5 as a potential therapeutic target for tubulointerstitial inflammation in diabetic kidney disease
Why this matters for people with diabetes
Many people with diabetes worry about their kidneys, but most explanations focus on blood sugar and blood pressure. This study looks deeper, asking what actually happens inside the kidney tissue as damage develops. By combining big data, single-cell analysis, and lab imaging, the researchers pinpoint a specific inflammatory switch in immune cells that could be turned off with a natural compound, potentially opening a new way to protect kidneys in diabetes.
A closer look at kidney damage in diabetes
Doctors have long known that diabetic kidney disease can lead to kidney failure, but attention has often centered on the tiny filters called glomeruli. Recent evidence, however, suggests that the spaces between the filters and the small tubes that process urine are even more important for predicting how quickly kidney function will decline. In these regions, waves of immune cells move in, drive inflammation, and leave behind scars. The authors set out to identify which genes, within this inflamed tissue, might be acting as master switches for the harmful immune response.
Finding an inflammatory switch in kidney immune cells
To hunt for these switches, the team pooled several public gene-expression datasets from kidney tubule samples of people with and without diabetic kidney disease. Using methods that group genes by how they turn on and off together, and then applying two independent machine-learning approaches, they narrowed thousands of genes down to a single standout: ALOX5. This gene encodes an enzyme that helps convert fatty molecules into leukotrienes, powerful chemical messengers that attract and activate immune cells. Across multiple patient groups, ALOX5 was consistently higher in diseased kidneys, and its levels closely tracked with worse kidney function and a more aggressively inflamed immune environment.
How one pathway fuels kidney inflammation
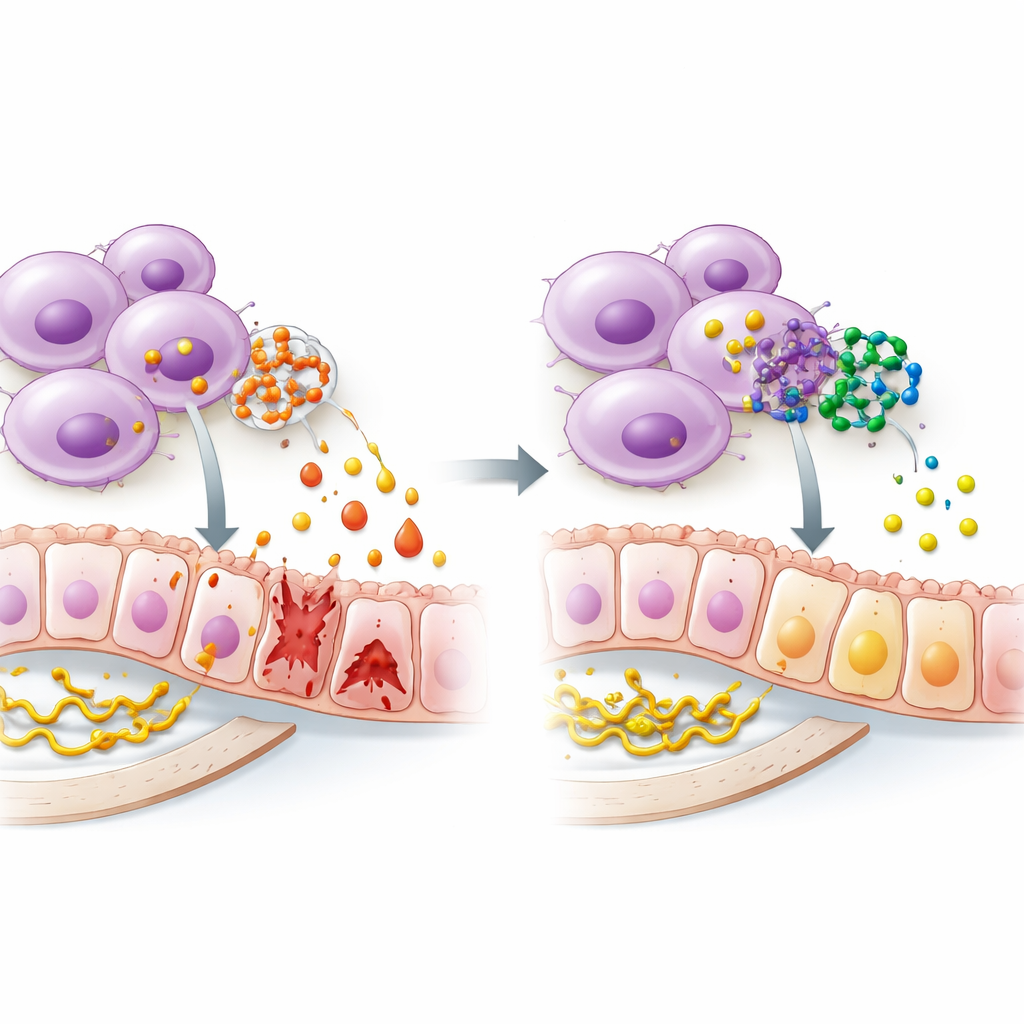
The researchers then asked where in the kidney this pathway is most active and how it might be driving damage. Single-cell sequencing data allowed them to look at individual cells rather than bulk tissue. They found that ALOX5 and its helper protein, ALOX5AP, were most strongly expressed in certain macrophages—front-line immune cells that can either calm or inflame tissues. As these cells shifted toward a pro-inflammatory state, the helper protein ramped up, suggesting that the leukotriene-making machinery was being primed. Multiplex immunohistochemistry, a technique that labels several proteins in tissue slices at once, confirmed that ALOX5, its partner, and markers of an “angry” macrophage state all clustered together around damaged tubules, while a receptor for leukotrienes appeared on both immune cells and nearby kidney cells. This pattern supports a self-reinforcing loop: macrophages make leukotrienes, those signals activate nearby cells through the receptor, and a central control system inside the cells, often called NF-kappa B, pushes them further into an inflammatory mode.
Searching for a drug to calm the fire
Identifying a pathway is only half the story; the next question is whether it can be safely blocked. Using a drug-signature database, the team screened for small molecules predicted to affect ALOX5, then used computer docking to see how tightly each candidate might bind to the enzyme’s 3D structure. Four compounds stood out, including a known ALOX5 blocker and honokiol, a natural substance derived from magnolia bark. Follow-up analysis of absorption and drug-like properties suggested that honokiol in particular might be suitable as an oral agent, with good uptake, reasonable chemical stability, and a low likelihood of confusing lab tests with nonspecific effects. While these predictions still need real-world testing, they point toward a feasible path from molecular insight to treatment.
What this could mean for future care
In everyday terms, the study proposes that a specific enzyme system in kidney-invading immune cells acts like a volume knob for inflammation in diabetic kidney disease. When ALOX5 and its partner protein are turned up, macrophages produce more inflammatory messengers, switch into a damaging mode, and help drive scarring and loss of kidney function. By dialing down this pathway—possibly with compounds such as honokiol—it might be possible to restore a healthier balance in the kidney’s immune environment and slow or prevent damage. Although clinical trials are still far off, the work offers a clear biological target and a promising candidate molecule, bringing the idea of precision anti-inflammatory therapy for diabetic kidney disease a step closer.
Citation: Lu, W., Deng, Y., Zhai, L. et al. Integrated machine learning and multi-omics analysis identifies ALOX5 as a potential therapeutic target for tubulointerstitial inflammation in diabetic kidney disease. Sci Rep 16, 14194 (2026). https://doi.org/10.1038/s41598-026-44445-0
Keywords: diabetic kidney disease, kidney inflammation, macrophages, ALOX5 pathway, honokiol