Clear Sky Science · en
Numerical study on the performance of a forced ventilation system under hydrogen leakage in an underground hydrogen equipment room
Keeping Underground Hydrogen Safe
Hydrogen is often hailed as a clean fuel of the future, but it is also highly flammable and very light, which makes it prone to pooling under ceilings if it leaks indoors. As cities look to tuck hydrogen equipment and refueling stations below ground to save space and improve safety at street level, a pressing question arises: how can we keep leaked hydrogen from building up to explosive levels in tight underground rooms? This study uses detailed computer simulations to explore how well different fan and vent layouts can sweep leaked hydrogen out of a compact underground equipment room.
Why Move Hydrogen Systems Underground?
Aboveground hydrogen stations must be separated from homes, schools, and other buildings by generous safety distances. In dense cities, finding large open plots for such facilities is difficult, which slows the spread of hydrogen-powered transport. Burying key equipment beneath the surface can shrink the blast impact on nearby structures and make refueling points easier to place. But going underground eliminates natural breezes and introduces structural limits on how many openings can be cut into walls and ceilings. The authors focus on a realistically small concrete room, just a few meters on a side, to see how hydrogen would spread if a leak occurred and how forced ventilation could clear it again.
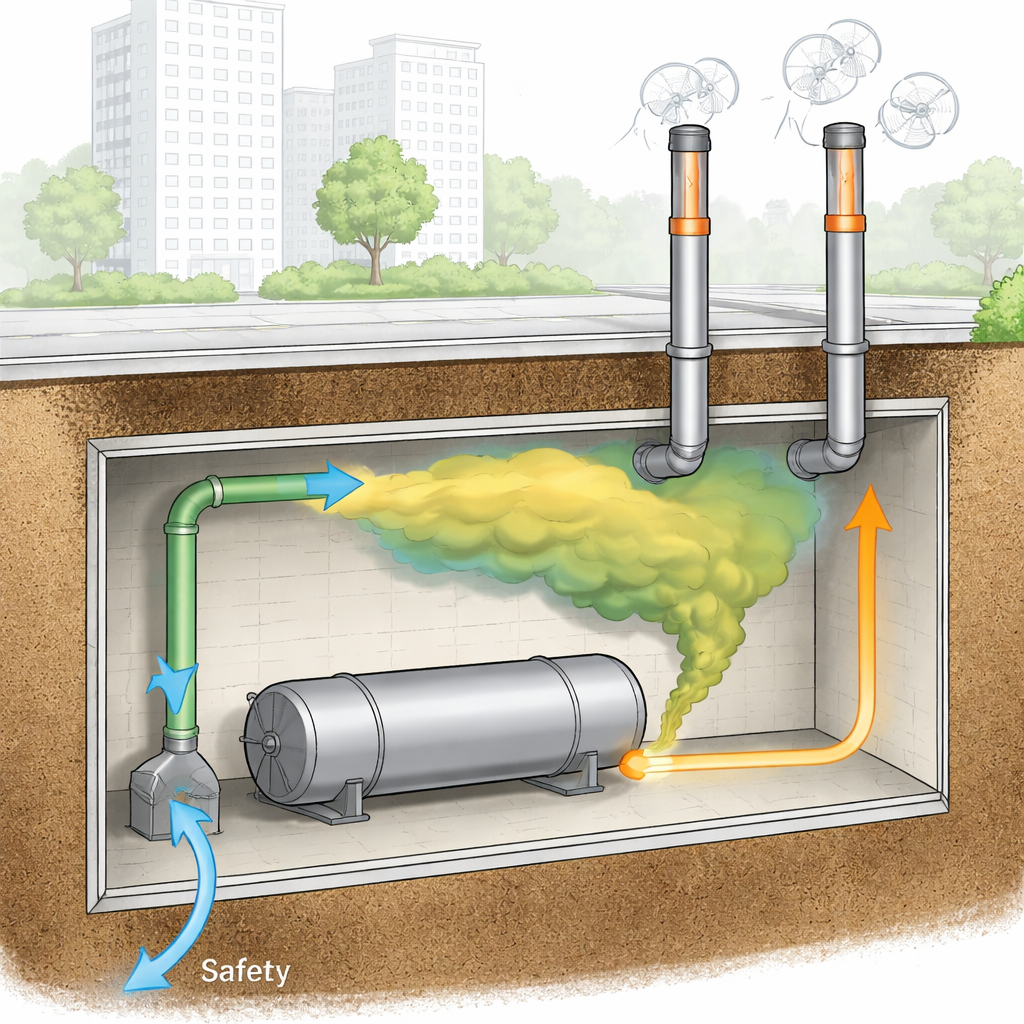
How the Leak and Ventilation Were Tested on a Computer
The team built a three-dimensional model of an underground room with a hydrogen nozzle on the floor, a small side vent near the floor, a larger main inflow vent, and one or two ceiling-level exhaust fans. Using commercial fluid-flow software, they simulated hydrogen jetting upward from the floor and mixing with still air, then later being removed when fans switched on. Nine “what-if” cases changed three main factors: how many fans ran, whether the main inflow vent was open or closed, and how fast hydrogen leaked. The simulations tracked how hydrogen concentration changed at many points in the room and, importantly, how long it took for the average level to cross key safety thresholds linked to flammability.
What Happens When Hydrogen Leaks Indoors
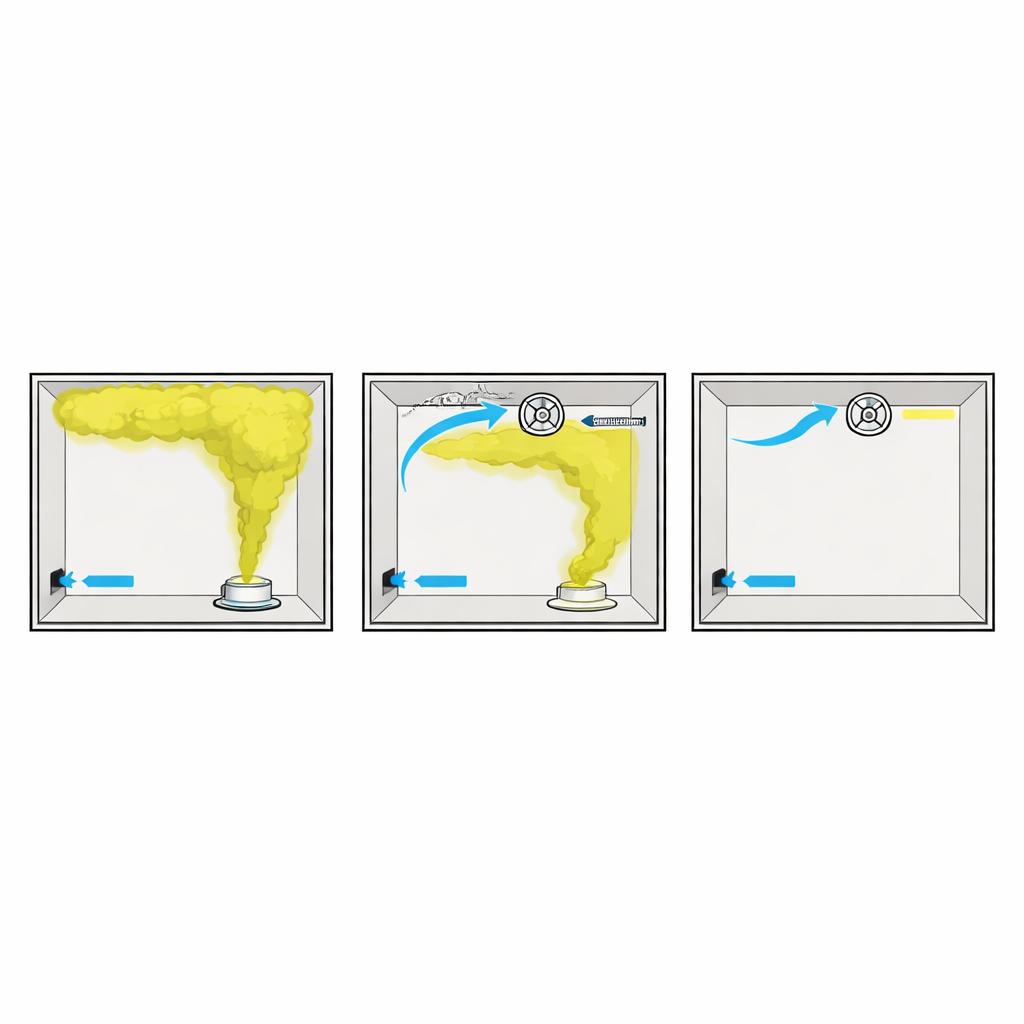
Across all leak cases, the same pattern appeared: hydrogen shot upward as a narrow jet, hit the ceiling, then spread sideways to form a layer of gas that gradually thickened and crept downward. Points at the same height saw similar concentrations, while higher locations became dangerous first. At realistic leak rates, large parts of the room crossed into a clearly flammable range in under a minute, and without any fan running, much of the space exceeded 10 percent hydrogen within five minutes. Opening a vent to the outside did provide some natural relief, but not enough to keep levels safely below the lower limit for ignition.
How Fan Layouts Change Safety Margins
When the researchers turned on forced ventilation after a leak, the details of fan and vent placement made a striking difference. If the main inflow vent remained closed, even adding a second exhaust fan only modestly sped up dilution. In contrast, opening the main inflow vent so that fresh air could enter freely cut the time needed for hydrogen near the ceiling to fall below the flammable limit by roughly 75 to 90 percent. Placing the inflow vent and exhaust vent on the same wall worked better than placing them on opposite walls, because poor layouts created stagnant pockets near a ceiling protrusion where hydrogen lingered. Under the best configurations, the upper part of the room dropped below the flammable level in just a few seconds; under poor ones, it could take more than a minute.
A Simple Rule-of-Thumb Tool
Alongside the detailed simulations, the authors developed a simple mathematical model that treats the whole room as if its gas were perfectly mixed. This model uses only the room volume, leak rate, and fan inflow and outflow rates to estimate how the average hydrogen level rises and falls over time. When the simulated room behaved fairly evenly—without strong pockets of high or low concentration—the simple model predicted the time to reach or drop below flammability thresholds within about 7 percent. In cases with strong layering or stagnant zones, its error grew to roughly 15 percent but still gave a reasonable first estimate. This makes the model useful during early design work, with detailed simulations reserved for final checks.
What This Means for Underground Hydrogen Rooms
For people designing hydrogen equipment rooms below ground, the study’s message is straightforward: how you arrange vents can matter as much as, or more than, how powerful your fans are. Ensuring a generously sized fresh-air inlet and placing that inlet in a coordinated way with the exhaust fan—ideally on the same side and aimed to sweep across areas where hydrogen collects—can shrink the time that the room stays in a flammable state by up to 90 percent. The authors show that with smart geometry and modest fan flow rates, underground rooms can meet strict hydrogen safety targets without carving large, structurally weakening openings in walls and ceilings.
Citation: Shin, HC., Hwang, I. & Seo, H. Numerical study on the performance of a forced ventilation system under hydrogen leakage in an underground hydrogen equipment room. Sci Rep 16, 10782 (2026). https://doi.org/10.1038/s41598-026-43773-5
Keywords: hydrogen safety, underground refueling, ventilation design, gas leak dispersion, computational fluid dynamics