Clear Sky Science · en
Development of hesperidin loaded lipid-chitosan nanoparticles: physicochemical characterization, molecular docking and ex vivo study
Why a Citrus Compound Needs a High-Tech Lift
Many of the health-boosting substances in foods, such as the citrus compound hesperidin, look powerful on paper but underperform in the body. Hesperidin has been linked to heart, immune, and anti-cancer benefits, yet it dissolves poorly in water and struggles to cross the gut wall, so much of it is simply wasted. This study explores a way to “repackage” hesperidin into tiny hybrid particles made from fats and a natural polymer, so it can dissolve better, linger longer in the intestine, and more effectively deliver its antioxidant and anti-inflammatory effects.
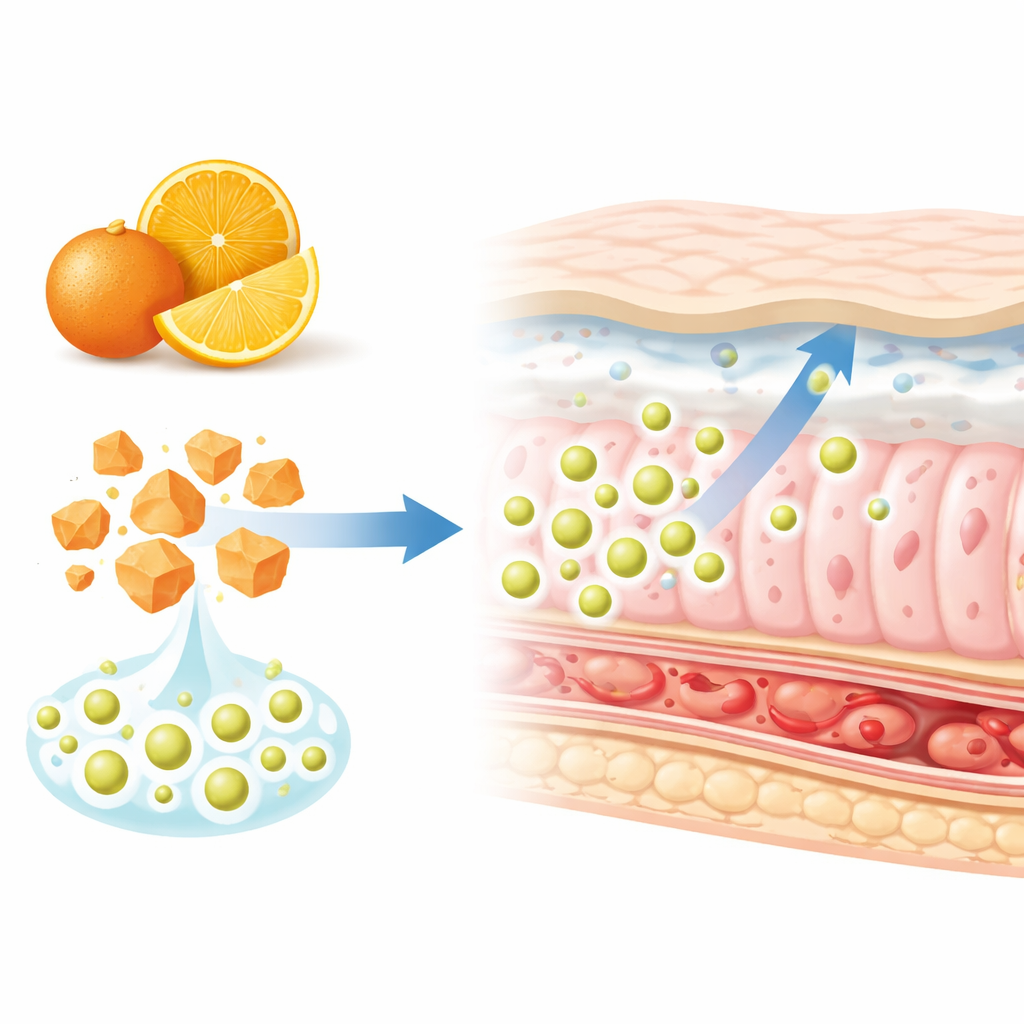
From Oranges to Tiny Delivery Vehicles
Hesperidin is a plant flavonoid found mainly in oranges, lemons, and other citrus fruits. Laboratory and animal studies have tied it to antiviral, anti-cancer, blood-pressure-lowering, antioxidant, and anti-inflammatory actions. Yet when swallowed as a conventional supplement, only a small fraction is absorbed. It dissolves poorly in water, is broken down by gut enzymes, and is pumped back out of intestinal cells. To overcome these obstacles, the researchers designed “hybrid” nanoparticles built from a solid fat (glyceryl monostearate) mixed with chitosan, a biodegradable sugar-based polymer derived from shellfish, plus helper molecules that stabilize the particles. Their goal was to trap hesperidin inside this nano-sized carrier to improve its solubility and its journey through the digestive tract.
Building and Inspecting the Nano-Carriers
The team used heating, magnetic stirring, and high-energy sound waves to disperse melted fat, chitosan, and hesperidin into water, forming uniform nanoparticles that were then freeze-dried into a stable powder. They produced several versions with different fat and chitosan amounts and measured properties such as particle size, surface charge, and how much drug was actually trapped inside. The best-performing formula contained particles about 200 nanometers across—thousands of times smaller than the width of a human hair—with a positive surface charge and high hesperidin loading. Advanced tests showed that within these particles, hesperidin shifted from a rigid, crystalline form to a more disordered, amorphous state, a change known to make poorly soluble compounds dissolve faster.
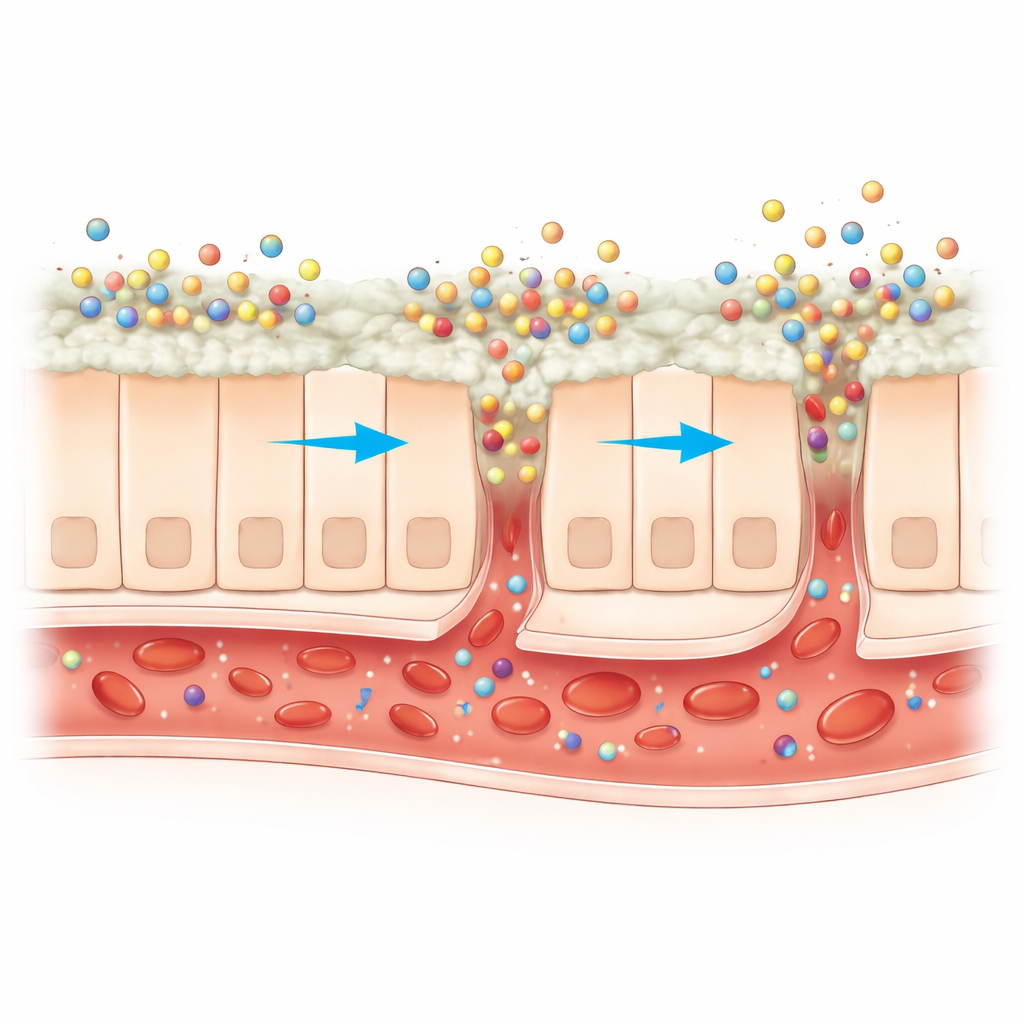
Helping the Drug Cross the Gut Wall
Next, the scientists asked whether these nanoparticles actually improved how hesperidin behaves in conditions mimicking the human intestine. In a lab-based release test, the nano-formulation steadily released more than 70 percent of its hesperidin content over 24 hours—around three-and-a-half times more than the raw compound. In experiments using goat intestine as a stand-in for the human gut, the nanoparticles delivered roughly 3.5 times more hesperidin across the tissue than free hesperidin. The chitosan coating, which carries a positive charge, bound well to negatively charged mucus on the intestinal surface and seemed to gently loosen the tight seams between neighboring cells, letting more of the tiny particles slip through. This combination of better dissolution, stronger sticking to the gut, and easier passage between cells suggests that, in living organisms, more of the swallowed dose could reach the bloodstream.
Boosting Antioxidant and Anti-Inflammatory Power
Because hesperidin is valued for its ability to neutralize harmful free radicals and calm inflammation, the researchers also compared the biological activity of the nano-formulation with that of the plain compound. In standard chemical tests of antioxidant capacity, the nanoparticles showed stronger, concentration-dependent free-radical scavenging than free hesperidin at several doses. In a protein-based assay used as a simple model of inflammation, the nanoparticle form again performed better at higher concentrations, approaching the effect of a common anti-inflammatory drug. To probe how hesperidin and its carrier materials might interact with biological targets at the molecular level, the team ran computer docking simulations. These suggested that both hesperidin and the carrier molecules could form favorable bonds with key antioxidant and inflammatory enzymes, supporting the lab observations.
What This Could Mean for Future Supplements
In plain terms, the study shows that carefully engineered fat–chitosan nanoparticles can help a stubborn, poorly soluble plant compound dissolve more easily, cling to the intestinal lining, and cross it more effectively, while maintaining or even enhancing its antioxidant and anti-inflammatory actions. If similar benefits are confirmed in animal and human studies, this strategy could turn hesperidin into a more reliable oral supplement or medicine, potentially allowing lower doses to achieve stronger effects. More broadly, the approach points to a general recipe for upgrading other plant-derived compounds that currently look promising in the lab but fail to live up to their potential in the human body.
Citation: Gilani, S.J., Altwaijry, N., Sultan, A.M. et al. Development of hesperidin loaded lipid-chitosan nanoparticles: physicochemical characterization, molecular docking and ex vivo study. Sci Rep 16, 13530 (2026). https://doi.org/10.1038/s41598-026-43743-x
Keywords: hesperidin, nanoparticles, drug delivery, antioxidant, chitosan