Clear Sky Science · en
Multimerin1 and not Galectin-8 tempers WNT signaling to promote gastric chief cell differentiation
Why stomach cell timing matters
Our stomach lining is constantly renewed by specialized cells that must grow up, or mature, at the right time. If this process is disturbed, it can set the stage for disease, including cancer. This study looks at how cells that line tiny blood vessels in the mouse stomach quietly guide the maturation of nearby digestive cells, and how a hidden genetic glitch in a common research mouse led scientists to an unexpected player in this process.
A surprise in a familiar mouse strain
The team began by studying a protein called galectin 8, which can bind certain sugar patterns that are overrepresented in high risk precancerous tissues in the gut. Because a well known mouse strain lacks the gene for galectin 8, the researchers expected this strain to reveal how galectin 8 affects a key stomach cell type called the chief cell, which makes digestive enzymes. They found that, compared with normal mice, young knockout mice had a clear delay in chief cell maturation, judged by the late appearance of specific markers that signal fully developed digestive cells.
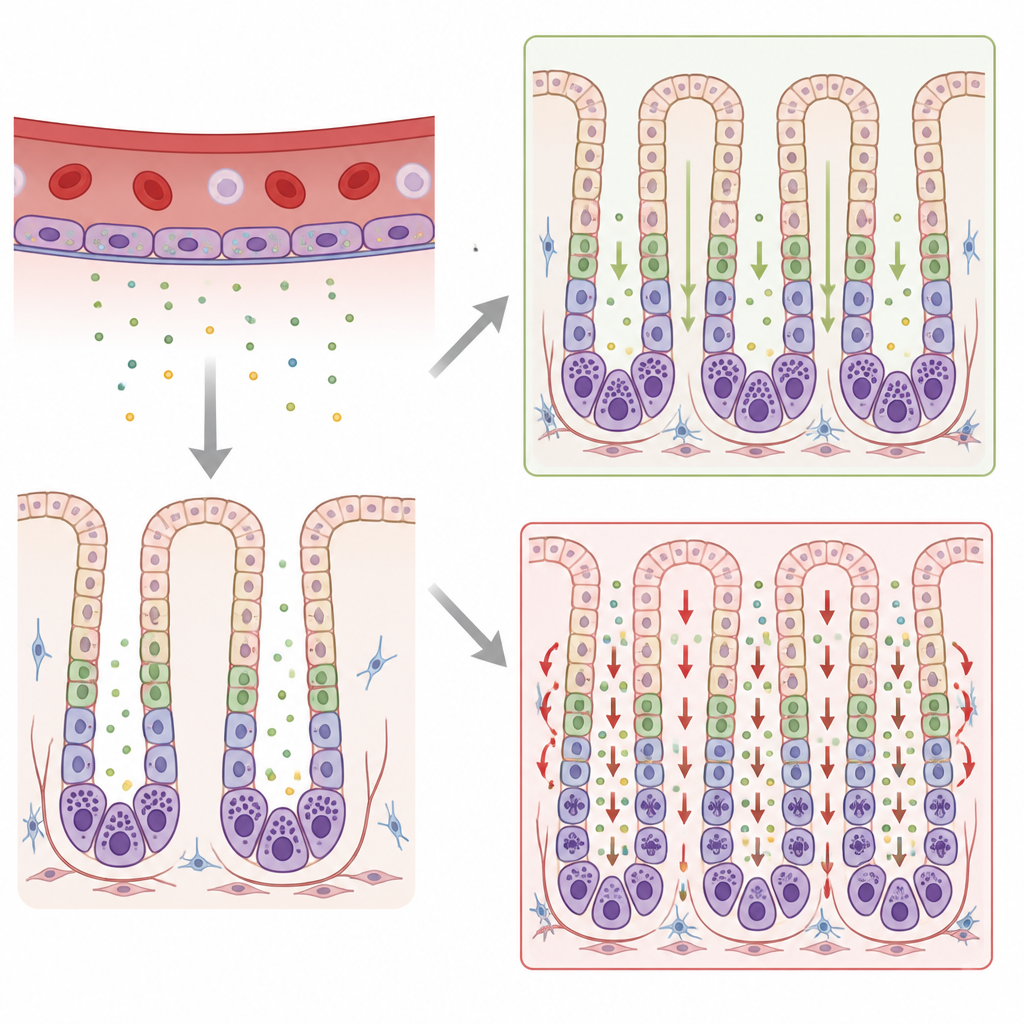
Narrowing down the true culprit
At first glance, this delay seemed to confirm a role for galectin 8 in stomach development. But further analysis raised doubts. Other stomach cell types, such as acid producing cells and surface lining cells, looked normal. Single cell RNA sequencing also showed that the galectin 8 gene was not even active in chief cells themselves, but instead in other cell types. Bulk RNA sequencing, followed by targeted genetic tests, revealed something unexpected: the galectin 8 knockout line also carried a previously unrecognized deletion of two neighboring genes on a different chromosome, including one called Mmrn1, which encodes a large blood vessel protein named multimerin 1.
Separating linked genes to test their roles
To work out which missing gene truly caused the chief cell delay, the scientists bred the mice so that the galectin 8 mutation could be separated from the Mmrn1 deletion. When they examined animals lacking only galectin 8 but carrying normal Mmrn1, chief cells matured on schedule, just like in standard mice. In contrast, mice lacking multimerin 1 (and a second gene, alpha synuclein) but having normal galectin 8 still showed the delayed chief cell maturation. Alpha synuclein, best known for its roles in the brain, was not detectable in the stomach, making multimerin 1 the most likely driver of the effect.
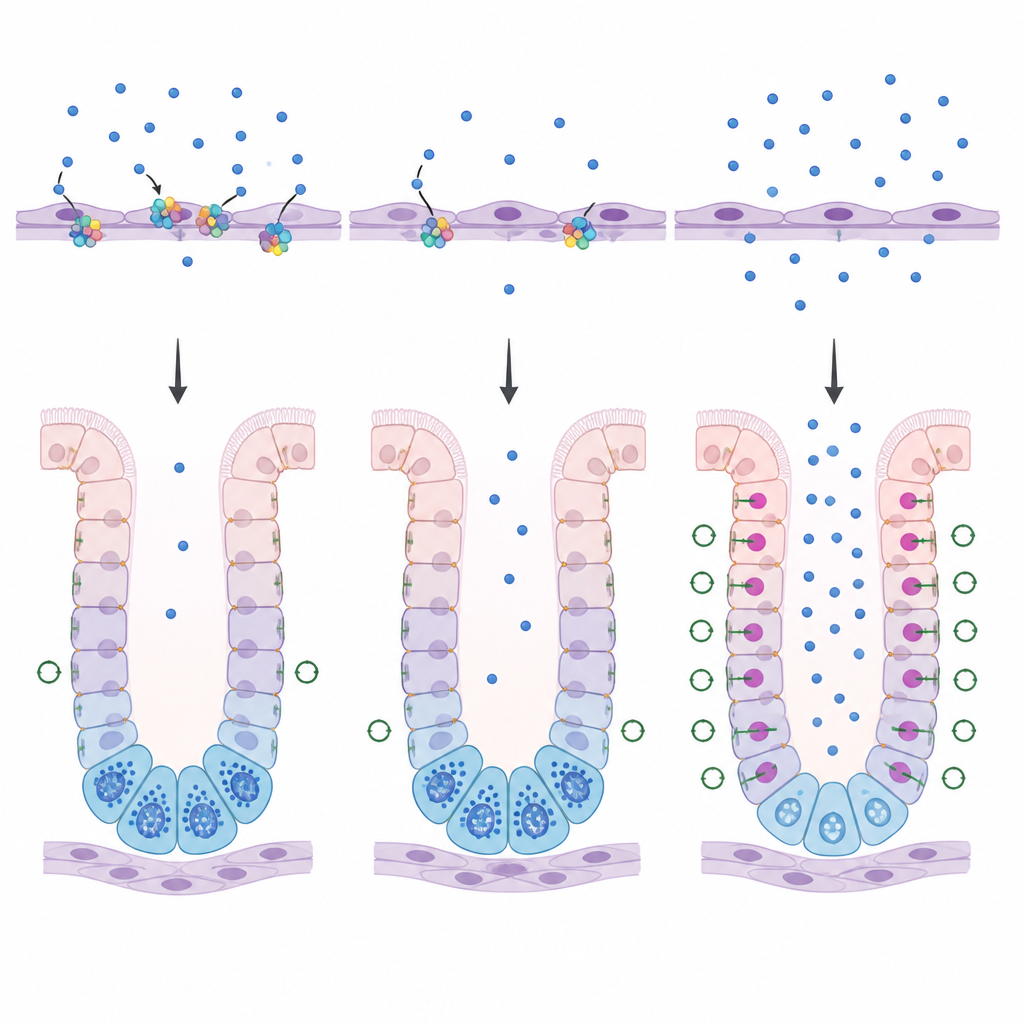
How blood vessel cells steer stomach growth
Multimerin 1 is made by cells that line blood vessels, not by the stomach surface cells themselves. Previous work on related proteins suggested they can dampen WNT signaling, a powerful growth signal that influences how stem cells decide their fate. In young mice lacking multimerin 1, the researchers saw more beta catenin inside the nuclei of stomach gland cells and more cells in an active growth state, both signs of heightened WNT activity. As the animals aged, this pattern faded, implying that other factors eventually take over the job of restraining WNT. The findings point to multimerin 1 as a kind of buffer that keeps WNT signals at a level that allows chief cells to mature properly at a critical early window.
What this means for stomach health and research
In simple terms, this study shows that a protein released from blood vessel cells helps nearby stomach cells grow up on time by keeping a powerful growth signal in check. It also illustrates how hidden genetic changes in lab animals can mislead scientists about which gene is responsible for a given trait. The authors conclude that multimerin 1, not galectin 8, tempers WNT signaling to promote normal development of enzyme producing chief cells in the mouse stomach, and they argue that future work using cleaner genetic models will be needed to explore how this pathway may connect to metaplasia and cancer risk.
Citation: Lin, X., Nicolazzi, G., Liu, X. et al. Multimerin1 and not Galectin-8 tempers WNT signaling to promote gastric chief cell differentiation. Sci Rep 16, 15011 (2026). https://doi.org/10.1038/s41598-026-43742-y
Keywords: gastric chief cells, WNT signaling, multimerin 1, gastric maturation, mouse stomach