Clear Sky Science · en
DFT based photophysical assessment of 2-substituted-3-(pyridin-2-yl)-benzo-[d][1,3]-azaphosphole P-oxide for organic optoelectronic applications
Why glowing plastics matter
Imagine phone screens, solar panels and smart windows made from thin, flexible films that can be printed like newspaper. To reach that future, scientists are searching for organic (carbon‑based) materials that can move electric charges efficiently, shine brightly in different colors and survive years of use. This study explores a new family of phosphorus‑containing molecules and shows, using computer simulations, how small changes in their structure can switch and fine‑tune their light emission and charge‑transport behavior for such devices.
Designing smarter light‑emitting building blocks
The researchers focus on a ring‑shaped scaffold called a benzazaphosphole oxide, which already behaves well as a light‑emitting semiconductor. They redesign one end of this scaffold by swapping a common benzene ring for a pyridine ring—a closely related “cousin” that contains nitrogen. Around this new core they attach different companion groups, creating nine related molecules. All calculations are carried out with density functional theory, a quantum‑mechanical method that allows the team to predict how electrons are arranged, how easily they move and what colors of light the molecules will absorb and emit, without yet having to synthesize each one.
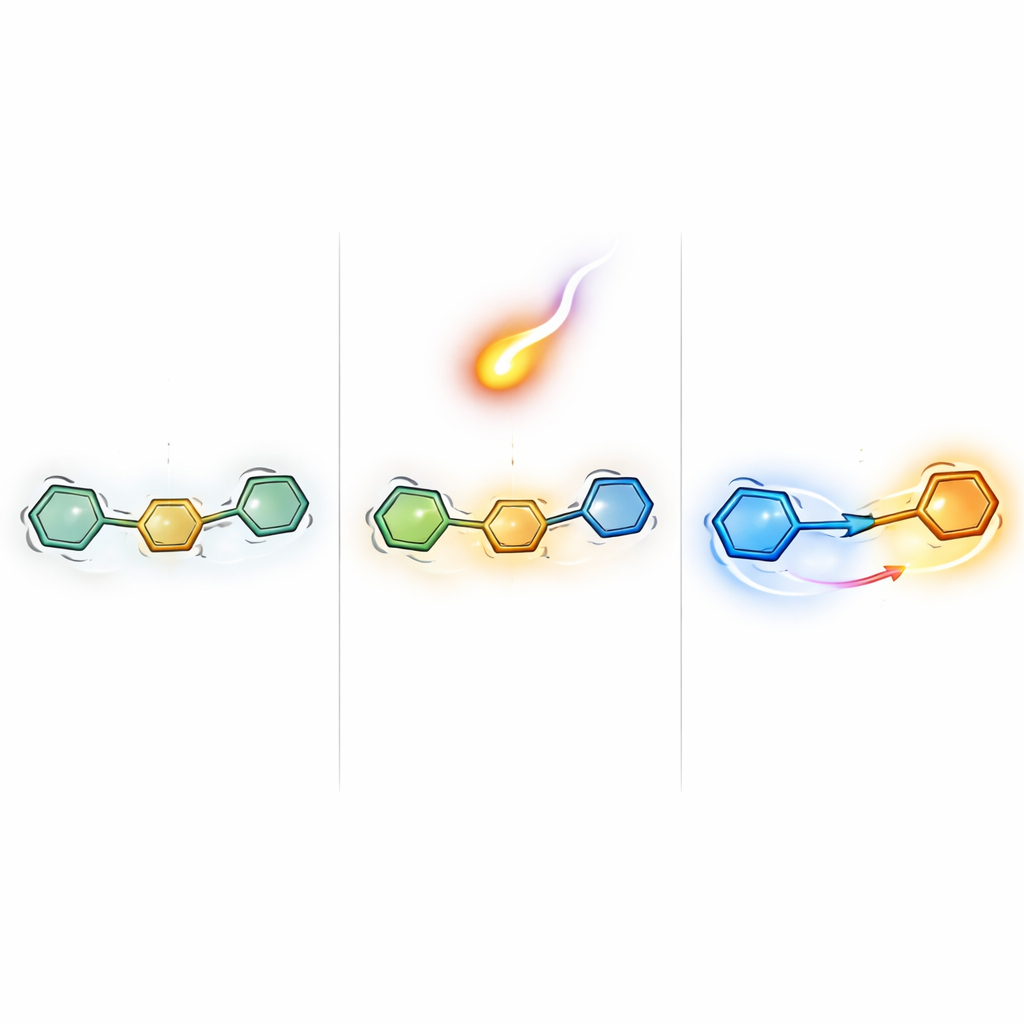
Flipping who gives and who takes electrons
A central finding is that the pyridine end dramatically rearranges how electrons flow inside these molecules. In earlier versions, the main scaffold acted as the electron donor and the outer ring as the acceptor. In the new designs, this role is flipped: the pyridine end now donates electron density, while the opposite aryl group becomes the main electron acceptor. This "push–pull" arrangement, where one side of the molecule pushes electrons and the other pulls them, is especially valuable in optoelectronics because it strengthens internal charge transfer. The authors quantify this by dissecting the frontier orbitals (the highest occupied and lowest unoccupied energy levels) and showing that the donor side dominates the filled orbital, while the acceptor side dominates the empty one.
Tuning color and charge flow with simple substitutions
By changing only the aryl group on the acceptor side, the team creates three clear classes of behavior. When that group carries halogen atoms such as fluorine, chlorine or bromine, the molecules keep relatively wide energy gaps and emit greenish light with large shifts between absorbed and emitted wavelengths—signatures of strong charge‑transfer in the excited state but with limited communication across the whole framework. When the aryl group is extended into sulfur‑rich ring systems, the electronic cloud spreads out more evenly across the molecule. These versions show narrower energy gaps, brighter emission and much lower “reorganization energies,” meaning they can move positive charges (holes) more easily—an advantage for use as hole‑transport layers.
Balancing brightness, stability and non‑linear response
A third set of molecules fuses the acceptor side to larger ring systems containing sulfur and nitrogen. These designs combine strong charge transfer with high predicted bond strengths, suggesting good resistance to thermal breakdown—crucial for devices that must operate for thousands of hours. Across all nine molecules, one simple measure—the spatial separation between the donor‑dominated and acceptor‑dominated orbitals—turns out to control many properties at once: as this separation grows, emission shifts from blue toward orange, the ease of hole motion improves, the gap between singlet and triplet excited states narrows and the calculated non‑linear optical response becomes stronger. Three particular structures stand out: one that emits cyan light efficiently, one that gives bright deep‑blue light with very fast emission, and one that emits orange light while balancing electron and hole transport and offering the highest calculated bond strength.
From computer predictions to future devices
For a non‑specialist, the key message is that the authors have discovered a simple “knob” for tuning organic semiconductors: invert the internal push–pull direction by adding a pyridine end group, then adjust the opposite end to dial in the desired color, charge‑transport balance and robustness. Their quantum‑chemical analysis suggests that several members of this new family should be particularly promising as active layers in light‑emitting diodes, solar cells or non‑linear optical components. While these predictions still need to be tested experimentally, the work provides a clear recipe for chemists who want to build next‑generation glowing plastics with properties tailored for flexible, low‑cost optoelectronic technologies.
Citation: Shoaib, M.M., Iftikhar, F., Mahmood, T. et al. DFT based photophysical assessment of 2-substituted-3-(pyridin-2-yl)-benzo-[d][1,3]-azaphosphole P-oxide for organic optoelectronic applications. Sci Rep 16, 14530 (2026). https://doi.org/10.1038/s41598-026-43447-2
Keywords: organic semiconductors, charge transfer, light emitting materials, phosphorus heterocycles, optoelectronic devices