Clear Sky Science · en
Integrated multi-omics analysis identifies and validates endoplasmic reticulum stress and mitophagy-related biomarkers in MASLD
Why stressed liver cells matter to everyday health
Metabolic dysfunction-associated steatotic liver disease (MASLD) is now one of the most common liver problems worldwide, closely tied to obesity and type 2 diabetes. In MASLD, fat gradually builds up in the liver and can progress to inflammation, scarring, and even liver cancer. This study takes a deep look inside liver cells to understand how two key forms of cellular stress, and a single control molecule called NR4A1, might drive that progression and open doors to new treatments.
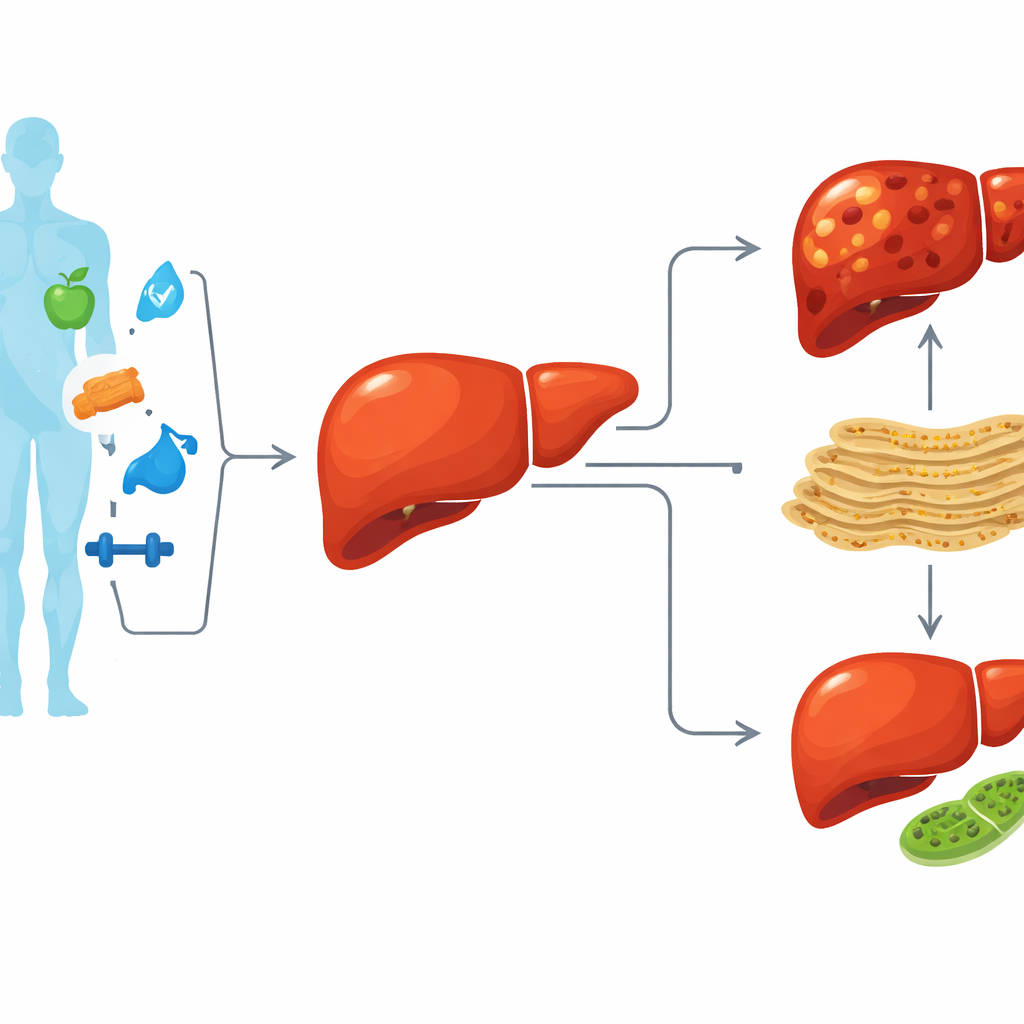
Fatty liver as a whole-body warning sign
MASLD is more than just a “fatty liver”; it reflects a broader breakdown in how the body handles energy. When the liver is overloaded with fat and nutrients for long periods, it can move from simple fat accumulation to more serious damage, including inflammation, fibrosis, cirrhosis, and liver cancer. Current therapies mainly focus on diet and lifestyle, which help but are often not enough. Researchers therefore want to understand the molecular switches inside liver cells that tip the balance from a stressed-but-adapting organ to one that is progressively failing.
Two stressed cell factories inside the liver
The authors focus on two critical structures inside liver cells. One is the endoplasmic reticulum, a membrane “factory” that processes proteins. When overloaded, it enters a state called stress, which first helps cells cope but, if prolonged, leads to inflammation and cell injury. The other is the mitochondrion, the cell’s power plant, which is kept healthy by a cleanup process called mitophagy, where damaged power plants are selectively removed. In MASLD, both this protein factory and these power plants are under chronic stress, and the balance between repair and damage can be lost. The study asks which genes sit at the crossroads of these stress responses and the buildup of liver fat.
Finding a central switch using big-data biology
The team combined several layers of “omics” data: bulk liver gene profiles from hundreds of patients, single-cell sequencing that tracks individual cell types, and advanced computer methods including machine learning. From thousands of genes linked to protein-factory stress and mitochondrial cleanup, they homed in on a small set active in MASLD. Among these, a gene called NR4A1 stood out as a central node. It was consistently lower in the livers of MASLD patients compared with healthy individuals, and its levels helped distinguish diseased from normal tissue with high accuracy. In single-cell data, NR4A1 was especially important in macrophages, the immune cells that patrol the liver, where its expression dropped as these cells shifted from early, adaptive states to more aggressive, inflammatory ones.
From cell models and mice to possible medicines
To go beyond computer predictions, the researchers created fatty liver–like conditions in human liver cell lines by exposing them to excess fatty acids. These cells accumulated fat and, at the same time, showed a clear drop in NR4A1 at both the RNA and protein levels. Similar reductions appeared in the livers of mice fed a high-fat diet and in tissue samples from MASLD patients, with NR4A1 levels declining further as disease advanced toward cirrhosis and cancer. Analyses of gene activity suggested that when NR4A1 is relatively high, cells show stronger stress-adaptation and repair pathways, especially those tied to protein quality control and mitochondrial health. Finally, the team used virtual screening to search for small molecules that might bind and stabilize NR4A1, identifying several candidate compounds for future experimental testing.
What this means for future diagnosis and treatment
Taken together, the findings present NR4A1 as a kind of internal thermostat that helps liver cells adjust to metabolic overload and maintain healthier protein factories and power plants. When this thermostat is turned down, cells appear more likely to accumulate fat, suffer from persistent stress, and drift toward chronic inflammation and scarring. While more work is needed to confirm cause-and-effect and to test drug candidates in living systems, this study suggests that measuring and eventually boosting NR4A1 activity could become part of a more targeted strategy to diagnose, monitor, and treat fatty liver disease before it reaches irreversible stages.
Citation: Chen, Q., Liu, L., Feng, J. et al. Integrated multi-omics analysis identifies and validates endoplasmic reticulum stress and mitophagy-related biomarkers in MASLD. Sci Rep 16, 12606 (2026). https://doi.org/10.1038/s41598-026-43311-3
Keywords: fatty liver disease, cell stress, mitochondria, immune cells, biomarkers