Clear Sky Science · en
Potential biomarkers of Ewing sarcoma identified through a Europe-wide analysis of prospectively collected samples
Why this childhood cancer study matters
Ewing sarcoma is a rare but aggressive cancer that strikes mainly children and young adults, often appearing in bones and quickly spreading to the lungs or other bones. Doctors can see how far the disease has spread, but they still struggle to predict which patients will do well and which are at highest risk of relapse, even when they receive the same modern chemotherapy. This Europe‑wide study set out to find biological warning signs inside the tumor’s DNA and RNA that could help personalize treatment, much like blood pressure numbers guide heart‑disease care. 
A closer look inside Ewing sarcoma tumors
The research team brought together 335 tumor samples from newly diagnosed Ewing sarcoma patients treated in large European clinical trials over two decades. All samples were collected before chemotherapy and processed using shared protocols in specialized biobanks. Instead of focusing on a single mutation, the scientists measured ten different “biomarkers.” Some were individual genes or small regulatory RNAs, such as MIR34A, STEAP1, EZH2, DKK2, and LGALS3BP, assessed by staining tumor sections or by quantitative PCR. Others reflected broader changes across the genome, such as gains or losses of whole chromosome arms (like chromosome 1q or 16q), tiny deletions affecting a gene region called ADAM3A, the overall percentage of the genome that was altered, and how much of the genome had lost normal pairs of gene copies, known as loss of heterozygosity.
Linking tumor signals to patient outcomes
For each biomarker, the investigators asked two questions: how it related to established clinical features (such as age, tumor size, pelvic location, or the presence of metastases at diagnosis) and how strongly it predicted survival. To do this, they grouped patients into “high” or “low” categories for each marker and used statistical models that account for other risk factors. They also calculated effect sizes to judge how clinically meaningful each signal was, not just whether it met a strict P‑value cut‑off. The study found that most markers were only weakly linked to each other, suggesting they captured different aspects of tumor biology rather than reflecting a single underlying change.
Genomic chaos as a warning sign
The most powerful signals came from markers that captured how disordered the tumor’s DNA had become. A high percentage of the genome altered (PGA) and extensive loss of heterozygosity were both associated with a clearly higher risk of events such as relapse and death, even after adjusting for metastases, tumor size, and other known factors. Patients whose tumors showed more widespread copy‑number changes or LOH tended to have pelvic tumors, responded less well to chemotherapy under the microscope, and were more likely to die during follow‑up. Gains of chromosome 1q and losses of 16q frequently traveled together and correlated with this broader genomic instability, reinforcing the idea that large‑scale DNA imbalances, rather than single point mutations, help drive aggressive disease. 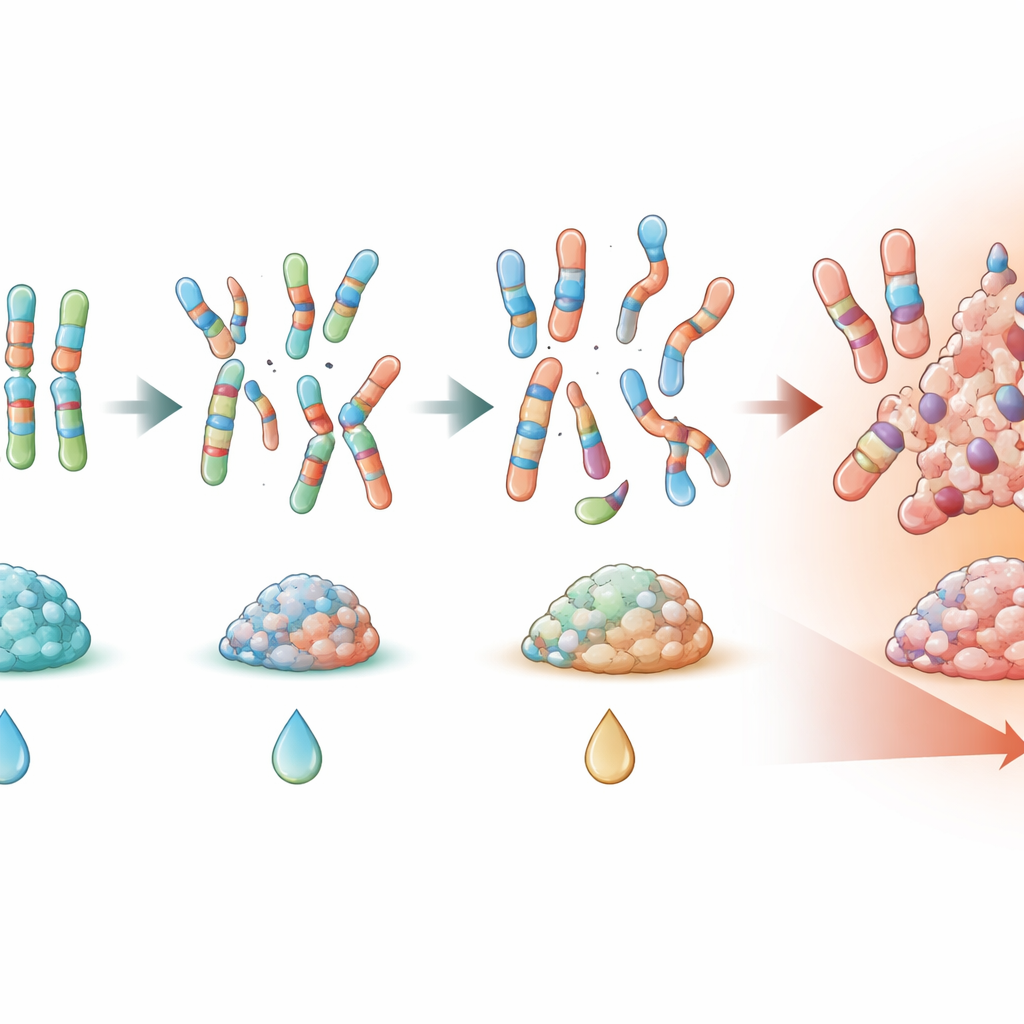
A protective signal from a tiny RNA
In contrast to markers of genomic chaos, higher expression of MIR34A, a small regulatory RNA previously linked to cell death and chemotherapy sensitivity, was tied to better outcomes. Patients with tumors rich in MIR34A experienced fewer relapses and deaths, and this effect remained strong when the analysis adjusted for standard clinical factors. Earlier laboratory work has shown that MIR34A can shut down genes that promote cell division, survival, and spread; this large clinical dataset now supports its role as a tumor‑suppressive influence in Ewing sarcoma. High membranous expression of the surface protein STEAP1 also tracked with better survival in patients with localized disease, while high EZH2 expression was more common in patients who already had metastases at diagnosis, although EZH2 itself did not clearly predict survival in this cohort.
What this means for future care
Taken together, the pan‑European analysis suggests that three markers stand out as particularly informative for Ewing sarcoma prognosis: widespread loss of heterozygosity, a high fraction of the genome altered, and low levels of MIR34A. Tumors that have highly disrupted DNA and lack this protective RNA are more likely to behave aggressively, whereas those with more orderly genomes and higher MIR34A tend to follow a more favorable course. While these markers are not yet part of routine care, they offer a path toward blood or tissue tests that could help doctors identify children who need intensified treatment and others who might safely avoid extra toxicity, bringing genuinely personalized therapy a step closer for this challenging cancer.
Citation: Ranft, A., Richter, G.H.S., Diaz-Martin, J. et al. Potential biomarkers of Ewing sarcoma identified through a Europe-wide analysis of prospectively collected samples. Sci Rep 16, 11613 (2026). https://doi.org/10.1038/s41598-026-43071-0
Keywords: Ewing sarcoma, cancer biomarkers, genomic instability, pediatric oncology, prognosis