Clear Sky Science · en
Transient adenovirus-Cre infection causes long-lasting remodeling of the mammary gland immune landscape
Why this matters for breast cancer research
Most of what we know about how breast cancer starts comes from studies in mice, where scientists can switch cancer-linked genes on or off in carefully chosen cells. This paper shows that one common way of flipping those genetic switches—using a harmless laboratory virus—quietly rewires the immune system in the breast for months. For anyone interested in how cancers begin, or how to harness immunity to prevent them, this work is a reminder that the tools we use to model disease can themselves shape the story we read out.
Two different paths to the same tumor
To study the earliest steps of breast cancer, researchers often delete a powerful tumor suppressor gene called p53 in milk duct cells of mice. The team compared two ways of doing this. One used a virus carrying an enzyme (Cre) that, once delivered into the mammary ducts, quickly deletes p53 in targeted cells. The other used a drug, tamoxifen, to activate a switchable form of the same enzyme already present in those cells. Both approaches ultimately produced similar mammary tumors with similar timing. At first glance, they might seem interchangeable—but the authors asked whether the way p53 loss is triggered leaves a lasting imprint on immune cells that share this tissue environment.
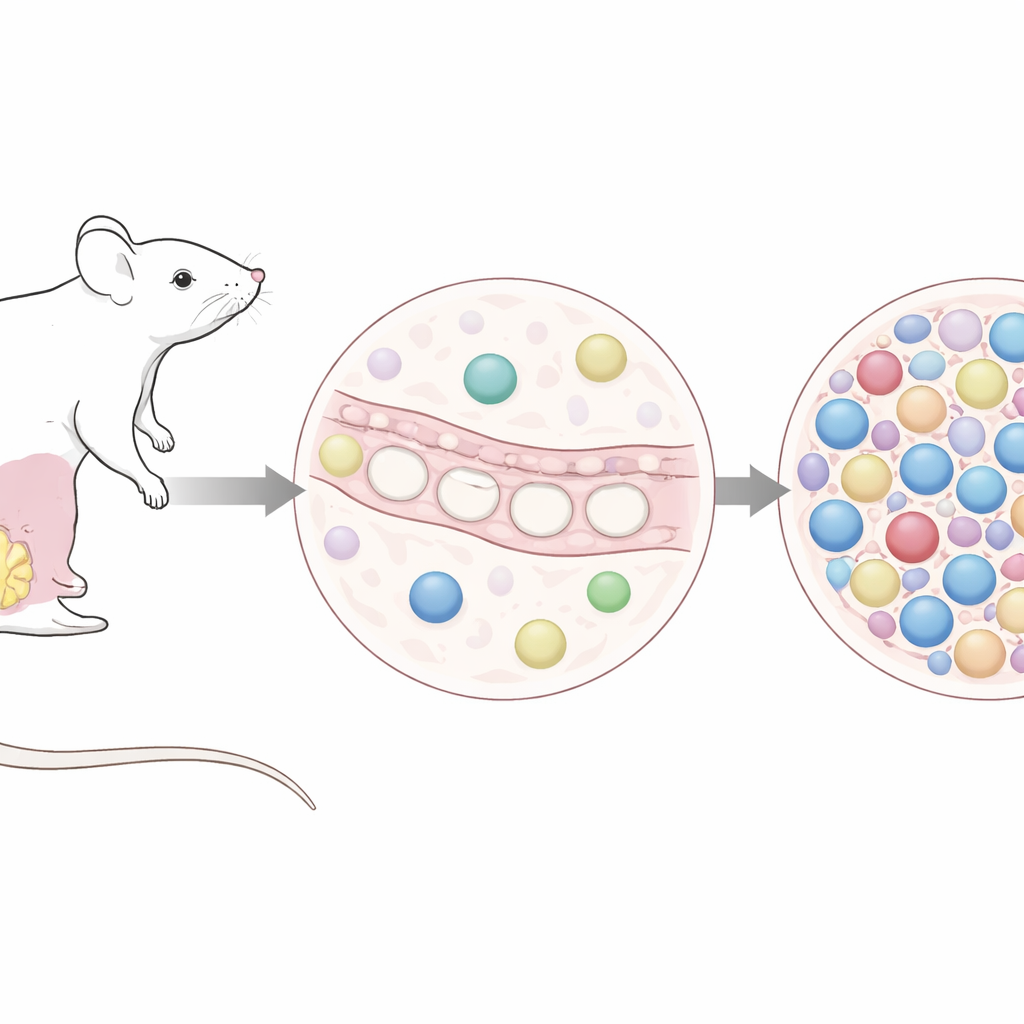
When a short viral visit leaves a long immune memory
Shortly after adenovirus was injected into the mammary ducts, the affected glands filled with white blood cells, as expected for a classic antiviral response. What was surprising is how durable this effect proved to be. Months later—well before full-blown tumors appeared—virus-exposed glands still contained many more immune cells than glands in tamoxifen-treated mice or in uninjected controls. Detailed cell profiling showed that this sustained increase was driven mainly by killer T cells (CD8 T cells). Many of these bore surface features of “tissue-resident” cells: they had settled in for the long term, poised in a heightened state of activation that did not subside back to baseline.
Virus noise hides the cancer signal
The authors then asked how this viral footprint affected the immune system’s ability to sense and respond to p53-deficient precancerous cells. In the tamoxifen model, as p53-null cells expanded over time, CD8 T cells in the mammary gland gradually became more activated and produced more inflammatory molecules, consistent with an emerging anti-tumor response. But in the virus-based model, a strong antiviral CD8 T cell presence was already in place and stayed highly activated regardless of whether p53 had been deleted. In other words, the persistent antiviral “buzz” made it very hard to detect additional changes driven specifically by the growing population of mutant cells. Virus exposure also temporarily pushed helper T cells (CD4 T cells) toward an antiviral state, potentially limiting their availability to support anti-tumor responses.
Shifts in other immune guardians
The study also examined macrophages and other myeloid cells, which help patrol tissues and can either support or suppress emerging cancers. In mice where p53 loss was induced with tamoxifen, the number of macrophages in the mammary gland rose as precancerous changes advanced. In contrast, virus-exposed glands showed a lower proportion of macrophages within the immune cell pool, likely because T cells had expanded so dramatically. Some subtle changes in macrophage subtypes tracked with p53 loss in both systems, but overall the viral imprint on T cells was the dominant alteration. Despite these immune differences, both induction methods led to tumors with similar onset times, suggesting that other factors—such as additional genetic hits—also strongly shape when tumors appear.
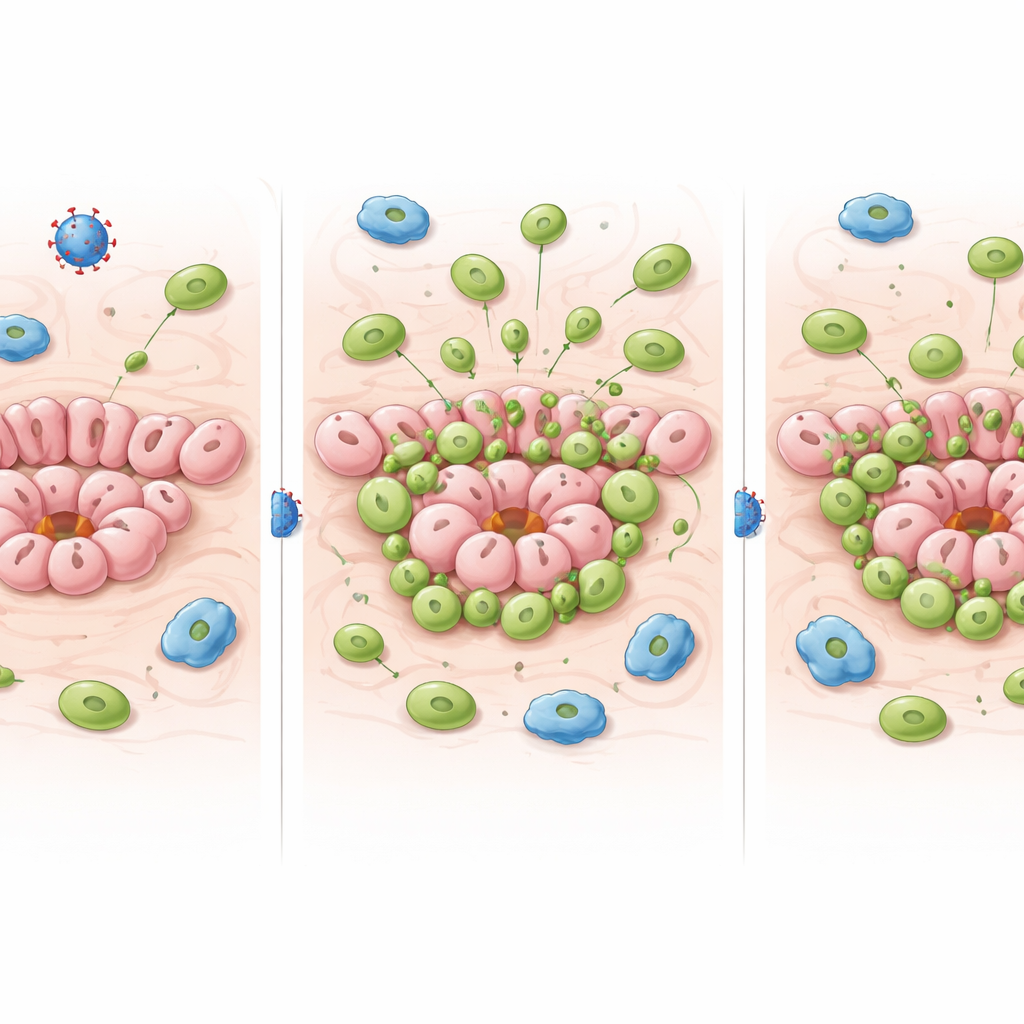
What this means for models and for people
For scientists, the message is clear: how you initiate cancer in a mouse can profoundly influence the local immune landscape, even long after the initial trigger is gone. Transient adenoviral infection leaves behind armies of resident, virus-focused T cells that can mask or distort the immune system’s natural response to early cancerous changes. That matters not only for interpreting data from breast cancer models, but also for similar virus-based systems in lung, ovary, bladder, and other tissues. More broadly, the work raises the possibility that even short-lived viral infections in human breast tissue could leave lasting immune scars that might, in subtle ways, alter future cancer risk. Choosing or designing models that minimize these hidden effects will be crucial for turning insights about early immune surveillance into reliable strategies to intercept breast cancer before it takes hold.
Citation: Han, S., Zhao, D., Chen, X. et al. Transient adenovirus-Cre infection causes long-lasting remodeling of the mammary gland immune landscape. Sci Rep 16, 13250 (2026). https://doi.org/10.1038/s41598-026-43069-8
Keywords: breast cancer, immune microenvironment, adenovirus, mouse models, tissue-resident T cells