Clear Sky Science · en
ACSL4 mediates ferroptosis to promote immunoglobulin A nephropathy progression: scRNA-seq analysis
Why this kidney study matters
Chronic kidney disease can quietly progress for years before symptoms appear, ultimately leading some patients to dialysis or kidney transplantation. One of the most common culprits is immunoglobulin A nephropathy (IgAN), an autoimmune disorder that scars the kidney’s filters. This study looks under the microscope—down to single cells—to ask a pressing question: is a newly recognized form of cell death helping to drive IgAN toward kidney failure, and could it offer a new target for treatment?
A closer look at a common kidney disease
IgAN is a widespread kidney condition in which immune proteins build up in the tiny filters that clean the blood. Over time, this buildup can inflame and damage kidney tissue, and up to 40% of patients may eventually reach end-stage kidney disease. Although researchers know that genetics, abnormal antibodies, and environmental triggers are involved, the exact cell types and internal processes that push IgAN from early damage to lasting scarring have remained unclear. Understanding which cells go wrong, and how, is essential for designing therapies that can slow or stop disease progression.
A new kind of cell death in the spotlight
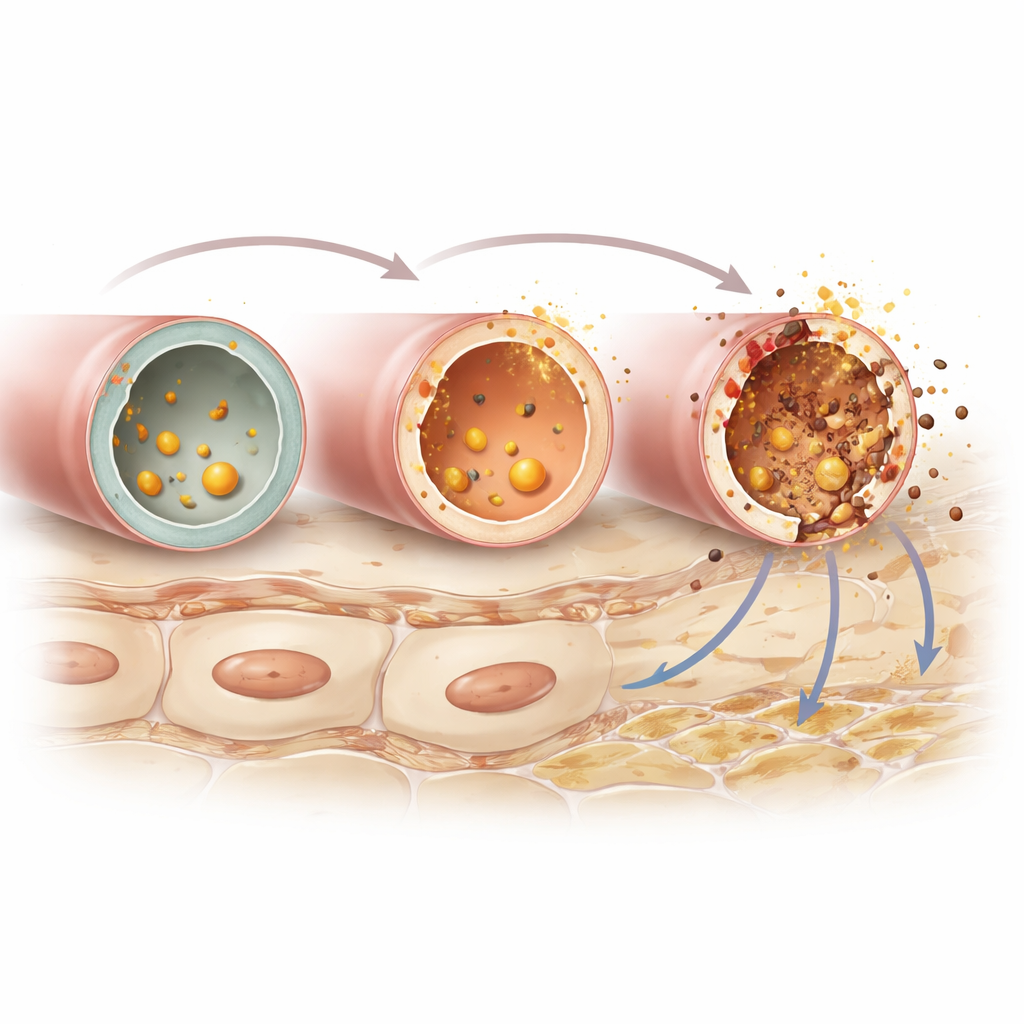
In recent years, scientists have discovered a distinct type of cell death called ferroptosis. Unlike familiar forms such as apoptosis, ferroptosis depends on iron and the toxic breakdown of fats in cell membranes. When this process accelerates, it can rupture cells and release irritating molecules into nearby tissue, fueling inflammation. A key player in this process is an enzyme called ACSL4, which helps pack certain fats into cell membranes, making them especially vulnerable to iron-driven damage. Because ferroptosis has been linked to kidney injury in other conditions, the authors asked whether ACSL4-driven ferroptosis might also be involved in worsening IgAN.
Zooming in on a specific kidney cell type
To answer this, the team analyzed a public single-cell RNA sequencing dataset from kidneys of patients with IgAN and a healthy control. This technology reads out which genes are active in thousands of individual cells, allowing researchers to sort them into distinct types. The authors identified twelve major cell populations, including blood vessel cells, immune cells, and different tubule cells that fine-tune the composition of urine. One cell type stood out: cells in the loop of Henle, a key segment that helps concentrate urine and balance salts. These cells were more abundant in IgAN kidneys than in the healthy sample, and they showed a striking rise in ACSL4 activity, while protective anti-ferroptosis genes were reduced, pointing toward a strong tilt in favor of ferroptotic cell death.
How stressed cells may talk to their neighbors
The study did more than catalog which genes were turned on. Using computational tools, the researchers modeled how loop of Henle cells might communicate with surrounding cells through signaling molecules. They found that ACSL4-high loop cells showed stronger predicted interactions with blood vessel cells, supporting cells in the kidney filters, and nearby tubule cells. Many of these signals involved the inflammatory messenger interleukin-6. This suggests that once loop cells are pushed toward ferroptosis, they may not die silently; instead, they could send distress signals that amplify inflammation and oxidative stress throughout the kidney tissue, encouraging further damage and scarring.
Evidence from real kidney tissue
To back up their computational findings, the authors examined kidney tissue from patients with IgAN and from people without the disease. Standard staining showed classic signs of IgAN: overgrowth of certain filter cells, inflammatory cell buildup between structures, and increased collagen fibers that mark scarring. When they labeled ACSL4 specifically, they saw that loop of Henle cells in healthy kidneys had very little of the enzyme, while those in IgAN kidneys glowed brightly, confirming the strong increase suggested by the gene activity data. Together, these images support the idea that loop cells expand in number and become more prone to ferroptotic damage as IgAN progresses.
What this means for future treatments
Overall, the study proposes that in IgAN, loop of Henle cells switch into a high-ACSL4, ferroptosis-prone state that both suffers damage and stirs up inflammatory signals such as interleukin-6. This combination may accelerate scarring and loss of kidney function. While the work relies heavily on computational analysis and tissue staining—and still needs follow-up experiments in cells and animals—it highlights ACSL4 and ferroptosis as promising targets. If future research confirms that blocking this pathway can protect loop cells and calm harmful signaling, it could open a new route to therapies that help IgAN patients keep their kidneys healthier for longer.
Citation: Shao, N., Tan, K., Chen, P. et al. ACSL4 mediates ferroptosis to promote immunoglobulin A nephropathy progression: scRNA-seq analysis. Sci Rep 16, 12543 (2026). https://doi.org/10.1038/s41598-026-43023-8
Keywords: IgA nephropathy, ferroptosis, ACSL4, kidney tubule cells, single-cell RNA sequencing