Clear Sky Science · en
A radio-genomics biomarker for precision epidermal growth factor receptor mutation targeting therapy in non-small cell lung cancer
Why this research matters to people with lung cancer
Lung cancer is still one of the deadliest cancers, and for many patients the best hope comes from drugs that precisely target the genetic faults driving their tumors. Yet finding those faults usually requires invasive biopsies and complex lab tests that are not always possible or reliable. This study shows that information hidden in routine CT scans can help doctors identify a particularly important genetic pattern in non-small cell lung cancer, potentially guiding patients more quickly and safely to the treatments most likely to help them.
Using scans instead of needles
Today, treatment choices for lung cancer often depend on whether a tumor carries changes in a gene called EGFR, which can make it sensitive to a class of pills known as tyrosine kinase inhibitors. To discover these changes, doctors typically insert a needle or use a scope to remove tumor tissue, then sequence its DNA. This process can be uncomfortable, risky, and sometimes impossible if the tumor is hard to reach or the patient is frail. Blood-based “liquid biopsies” are less invasive but can miss mutations or pick up misleading signals from normal cells. The authors of this paper set out to see whether CT images, which almost every lung cancer patient already receives, could non‑invasively predict a tumor’s EGFR profile in a clinically useful way.
A fingerprint hidden in CT images
The team focused on a scenario they call “exclusive” EGFR mutation: tumors that have EGFR alterations but lack other major targetable mutations such as ALK or KRAS. These tumors tend to respond better to EGFR‑targeted drugs, making them especially attractive candidates for first‑line therapy. Using CT scans and genetic data from 304 patients at several hospitals, plus an independent public dataset of 51 patients, the researchers extracted a huge set of quantitative image measurements—called radiomic features—from three regions: the tumor itself, a thin ring of surrounding tissue, and nearby lung. They then applied statistical techniques to shrink this library to a compact 10‑feature signature they named EGFR‑RPV (radiomics predictive vector), designed to distinguish exclusive EGFR‑mutant cancers from all others.
How well the imaging marker performs
When tested on patients whose data were not used to build it, EGFR‑RPV correctly identified exclusive EGFR‑mutant tumors in roughly three out of four cases, in both internal and external validation groups. Many of the most informative image features came from the tissue immediately around the tumor rather than the tumor core, supporting the idea that cancer cells influence their surroundings in subtle but detectable ways. One feature from the broader lung region carried negative weight, echoing the clinical observation that EGFR‑mutant tumors are more common in people without heavy smoking‑related lung damage. Beyond predicting mutation status, the radiomic score also separated patients into higher‑ and lower‑risk survival groups, suggesting it captures broader aspects of tumor behavior.
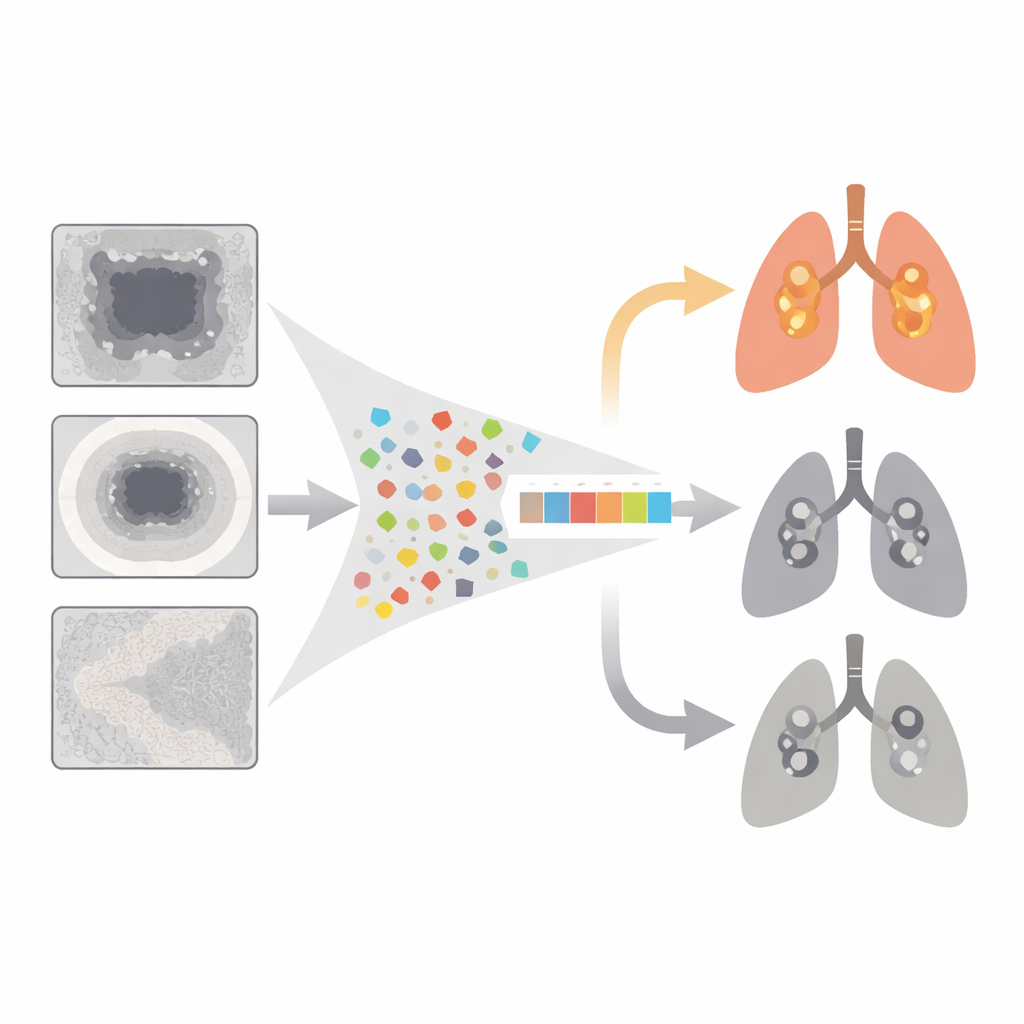
Linking images to tumor biology
To probe what might underlie the imaging patterns, the researchers turned to gene‑activity data from the public cohort. They found that tumors with exclusive EGFR mutations more often expressed two genes, FAM190A and BCMO1, which are involved in cell division control and vitamin A metabolism. A broader pathway analysis suggested that these tumors may rely on a developmental signaling route known as Hedgehog, while dialing down some of the usual programs for rapid cell growth and inflammation. Although these biological links need further confirmation, they hint that the CT‑based signature is not just a statistical trick, but reflects real differences in how these cancers grow and interact with their environment.
What this could mean for patients
From a patient’s perspective, the appeal of EGFR‑RPV is its practicality: it uses standard CT scans, can be computed quickly, and does not require an additional procedure. The authors emphasize that their tool is meant to complement, not replace, tissue and blood testing. In situations where biopsy samples are scarce, test results are delayed or unclear, or repeated invasive procedures would be burdensome, an imaging‑based estimate of exclusive EGFR status could help steer decisions—such as whether to prioritize EGFR‑targeted pills, broaden mutation testing, or consider other strategies. While larger and prospective studies are still needed before this approach can be widely adopted, the work showcases how modern image analysis may turn everyday scans into powerful guides for precision lung cancer care.
Citation: Chen, M., Copley, S.J., Linton-Reid, K. et al. A radio-genomics biomarker for precision epidermal growth factor receptor mutation targeting therapy in non-small cell lung cancer. Sci Rep 16, 12416 (2026). https://doi.org/10.1038/s41598-026-42948-4
Keywords: non-small cell lung cancer, EGFR-targeted therapy, radiomics, CT imaging biomarker, precision oncology