Clear Sky Science · en
Streptomyces koyangensis L-asparaginase: computational prediction of dual-mechanism BCL-2 interaction in acute lymphoblastic leukemia
Why a Bacteria Enzyme Matters for Childhood Leukemia
Acute lymphoblastic leukemia (ALL) is the most common cancer in children, and one of its key drugs is an enzyme called L-asparaginase. This medicine starves leukemia cells by removing an amino acid they cannot make for themselves. But today’s versions, made from common bacteria like E. coli, can trigger strong allergic reactions and other side effects. This study explores whether an L-asparaginase from a different microbe, Streptomyces koyangensis, could offer a safer and more powerful option—and even attack leukemia cells in two ways instead of one.
A New Twist on a Familiar Cancer Drug
Leukemia cells depend on a nutrient called asparagine, floating in the bloodstream, to grow and divide. Standard L-asparaginase drugs work by breaking down this nutrient so the cancer cells essentially starve, while healthy cells, which can make their own asparagine, cope much better. Unfortunately, the current drugs are large bacterial proteins that the immune system often treats as dangerous invaders, causing allergic reactions and sometimes forcing treatment to stop. The researchers looked to alternative sources of the enzyme—from two Streptomyces bacteria and from date palm (a plant)—to see whether any might be better suited for therapy.
Using Computers to Explore Invisible Molecules
Instead of going straight to lab animals or patients, the team first built detailed computer models of each candidate enzyme. They examined basic physical traits such as size, stability, and how water-loving or water-repelling each enzyme was—features that influence how long a drug may last in the body and how likely it is to clump or break down. Then they used several independent simulation methods to predict how these enzymes might stick to key leukemia-related proteins inside or on the surface of cancer cells. By comparing different algorithms and running long, physics-based simulations of molecular motion, they could test whether the predicted “docking” between enzyme and cancer protein was strong and stable over time.
Finding a Promising Partner in Streptomyces
Across all these tests, the L-asparaginase from Streptomyces koyangensis consistently stood out. Computer docking suggested it binds very tightly to a protein called BCL-2, which is often overproduced in leukemia cells and acts like a bodyguard that prevents them from dying. Follow-up simulations showed that the enzyme and BCL-2 form a large, snug contact surface held together by many hydrogen bonds and favorable electrostatic and hydrophobic interactions. The complex remained remarkably stable during a 100-nanosecond virtual “stress test,” with only small wobbles in shape and a strongly negative calculated binding energy—signs that this partnership would likely hold up in real life. In contrast, enzymes from the other sources showed weaker and less stable interactions.
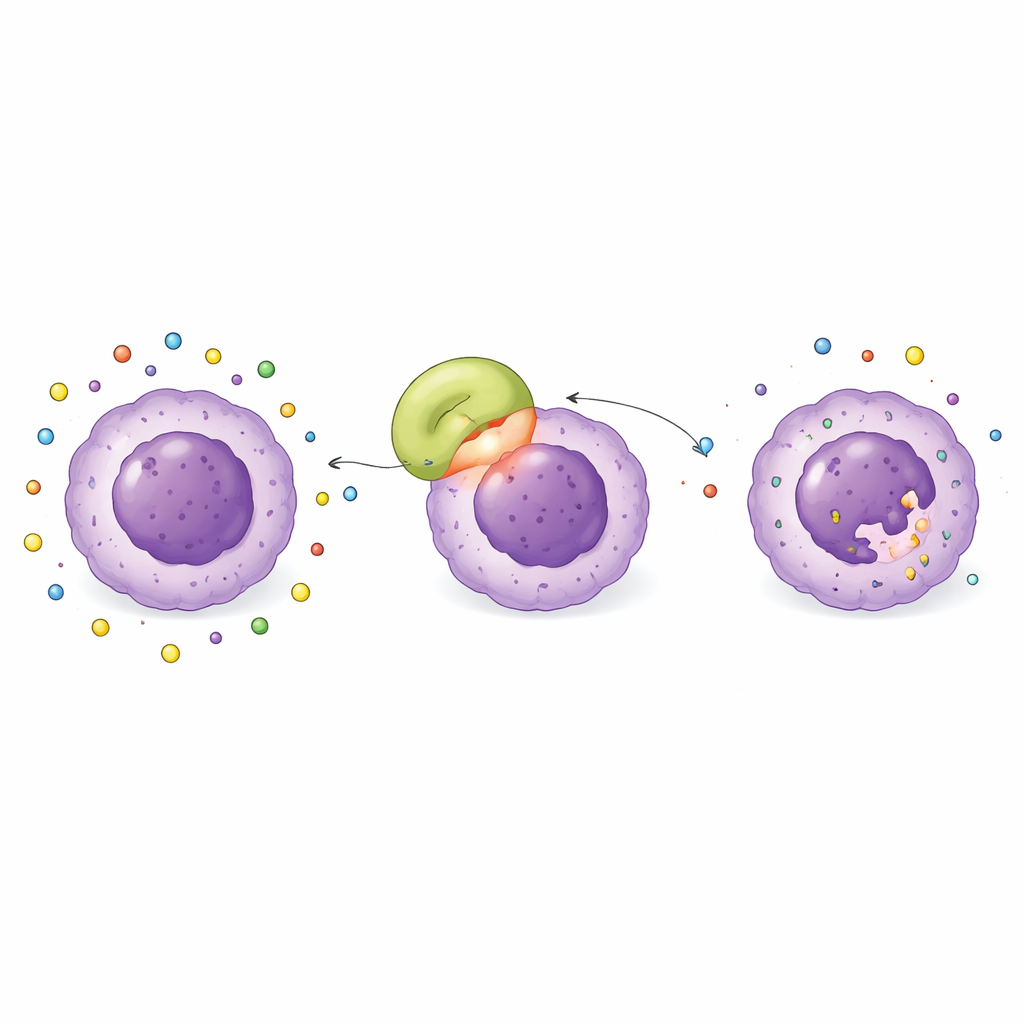
A Potential Double Hit on Leukemia Cells
These findings hint that Streptomyces koyangensis L-asparaginase might not only starve leukemia cells by removing asparagine, but also directly latch onto BCL-2, potentially weakening the cells’ defenses against programmed cell death. In principle, that would amount to a double hit: cutting off a vital nutrient while also disarming a key survival shield. The study even identified specific “hot spot” amino acids on the enzyme that are most important for this binding, providing clear targets for future fine-tuning to balance potency and safety. Because Streptomyces enzymes may provoke fewer immune reactions than standard bacterial sources, this candidate could eventually address both resistance and side-effect problems seen with current therapy.
What This Means and What Comes Next
For non-specialists, the take-home message is that powerful computers can now screen and shape potential cancer drugs before they ever reach a test tube. Here, an in-depth battery of simulations points to Streptomyces koyangensis L-asparaginase as a promising next-generation treatment for ALL, with the intriguing possibility of a dual mechanism—nutrient deprivation plus direct attack on a survival protein. However, the work so far is entirely virtual. The authors stress that laboratory experiments with purified enzyme, leukemia cells, and animal models are essential to confirm whether this predicted double action really occurs and is safe. If those studies succeed, this microbe-derived enzyme could help refine leukemia treatment and inspire similar computational searches for smarter, more precise biologic drugs.
Citation: Solanki, G., Prajapati, C., Gadhvi, R. et al. Streptomyces koyangensis L-asparaginase: computational prediction of dual-mechanism BCL-2 interaction in acute lymphoblastic leukemia. Sci Rep 16, 12675 (2026). https://doi.org/10.1038/s41598-026-42798-0
Keywords: acute lymphoblastic leukemia, L-asparaginase, BCL-2, Streptomyces koyangensis, computational drug design