Clear Sky Science · en
Efficacy of a novel bacteriophage in controlling Escherichia coli associated with swine farm environments and its potential for biofilm disruption
Why pig farms and germs matter to all of us
What happens in a pig barn might seem far removed from everyday life, but the bacteria that thrive there can travel through meat, water, and the wider environment to reach people. As antibiotics lose their punch against these microbes, scientists are urgently hunting for new ways to keep animals healthy and food safe. This study explores whether a naturally occurring virus that preys on bacteria can help control a troublesome strain of Escherichia coli in swine farms, especially when the bacteria huddle together in stubborn, protective layers called biofilms.
A tiny hunter found in river water
The researchers began by collecting water from the Chao Phraya River in Bangkok and searching it for viruses that specifically attack multidrug-resistant E. coli from swine farm wastewater. They isolated one promising candidate and named it vECPPW9. Under an electron microscope, this virus has a classic "space-lander" shape, with a many-sided head and a contractile tail that lets it latch onto bacterial cells. In lab tests, vECPPW9 was able to kill about 40 percent of the resistant E. coli strains they examined, giving it a reasonably broad reach among problem bacteria found in these farm environments.
Fast attack, strong punch, and tough under stress
To see how well vECPPW9 performs as a bacterial predator, the team tracked how quickly it sticks to its targets and multiplies. At body temperature, more than 90 percent of the virus particles attached to E. coli cells within ten minutes. Once inside, the virus took about 20 minutes of quiet preparation before releasing hundreds of offspring from each infected cell, bursting the bacteria open. Even when the scientists added relatively few virus particles compared with bacteria, E. coli growth was sharply suppressed within an hour, and bacterial numbers stayed far below untreated cultures over 24 hours. The phage also remained stable at temperatures typical of storage and animal housing and across a fairly wide range of acidity, important traits if it is to be used in real barns and water systems.
A safe genetic blueprint with powerful tools
The team sequenced the entire genetic material of vECPPW9 to check both its identity and its safety. The virus carries a large double-stranded DNA genome packed with hundreds of genes that build its structure, copy its DNA, and break open host cells. Crucially, the researchers found no genes linked to bacterial toxins, antibiotic resistance, or the ability to hide quietly inside bacterial chromosomes. Instead, they identified enzymes that can punch holes in bacterial cell walls and likely others that chew through the slimy sugars making up biofilms. Comparisons with known viruses showed that vECPPW9 belongs to a group called Phapecoctavirus but is distinct enough to count as a new species within this family of strictly lytic, or always-killing, phages.
Breaking down stubborn bacterial communities
Because many farm bacteria live in biofilms on plastic, rubber, and metal surfaces, the scientists asked whether vECPPW9 could stop these communities from forming or tear them down once established. In simple plastic wells, mixing the phage with E. coli from the start cut biofilm mass by up to about three-quarters and reduced the number of living cells inside by more than a hundred-fold, depending on the dose and exposure time. When the team allowed biofilms to grow for a day or three before adding phage, vECPPW9 still reduced the thickness of the slime and the number of surviving bacteria. Under a scanning electron microscope, untreated biofilms looked like dense, well-organized cities of intact rod-shaped cells, while phage-treated surfaces were littered with ruptured bacteria and debris, and the smooth matrix was clearly disrupted.
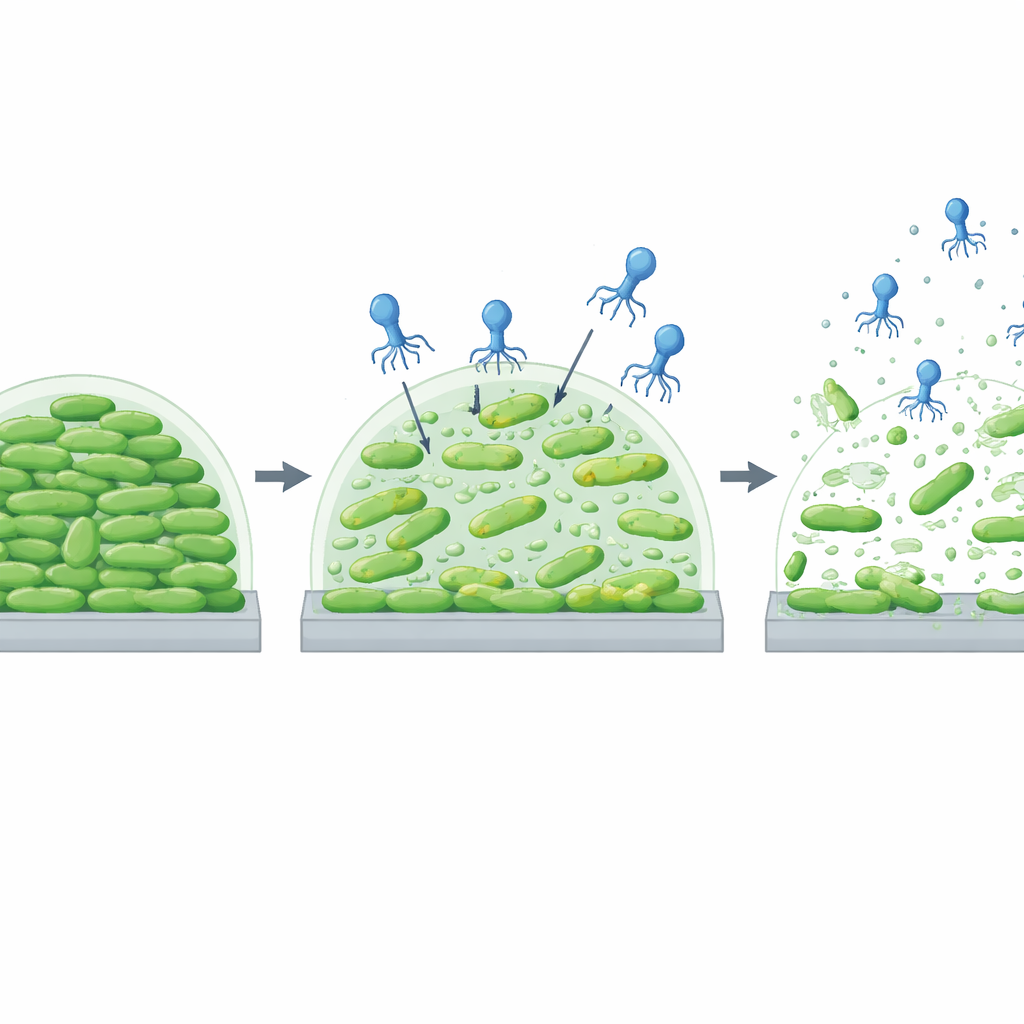
Cleaning up real farm-like surfaces
To mimic conditions in swine barns more closely, the researchers grew E. coli biofilms on rubber tubing and stainless-steel coupons similar to those used in drinkers and pipelines. Over three days, biofilms steadily thickened in untreated controls, but surfaces exposed to vECPPW9 had much less buildup at every time point. When biofilms were allowed to form first and only then treated with phage, the virus again trimmed down the biomass compared with controls, even as the communities matured. These findings suggest that vECPPW9 can both prevent and chip away at biofilms on the types of materials found in water and feed systems, potentially improving hygiene and reducing the spread of resistant bacteria.
What this could mean for farms and food
Taken together, the study paints vECPPW9 as a fast-acting, genetically safe viral hunter that can kill multidrug-resistant E. coli and weaken the biofilms that shelter them on farm surfaces. While all experiments were done under controlled lab conditions, and more work is needed in live animals and real barns, the results point toward practical uses: adding phages to drinking water or feed, or spraying them onto equipment and pipes to keep harmful bacteria in check. If such approaches prove effective and safe at scale, they could help farmers reduce their reliance on antibiotics, slow the spread of resistance, and ultimately make pork production safer for animals, workers, and consumers alike.
Citation: Wintachai, P., Thonguppatham, R., Smith, D.R. et al. Efficacy of a novel bacteriophage in controlling Escherichia coli associated with swine farm environments and its potential for biofilm disruption. Sci Rep 16, 12937 (2026). https://doi.org/10.1038/s41598-026-42644-3
Keywords: bacteriophage therapy, Escherichia coli, swine farming, biofilm control, antibiotic resistance