Clear Sky Science · en
Analysing cell death patterns to predict outcomes and treatment options in patients with high-grade serous ovarian carcinoma
Why Cell Death Matters for Ovarian Cancer
High-grade serous ovarian carcinoma is one of the deadliest cancers affecting women, largely because it is often found late and tends to come back after surgery and chemotherapy. This study asks a deceptively simple question with life-or-death stakes: can we learn enough from how cancer cells die to predict which patients will do poorly and which treatments might help them most? By turning patterns of cell death into a numerical score, the researchers aim to give doctors a clearer map for guiding care and designing new therapies.
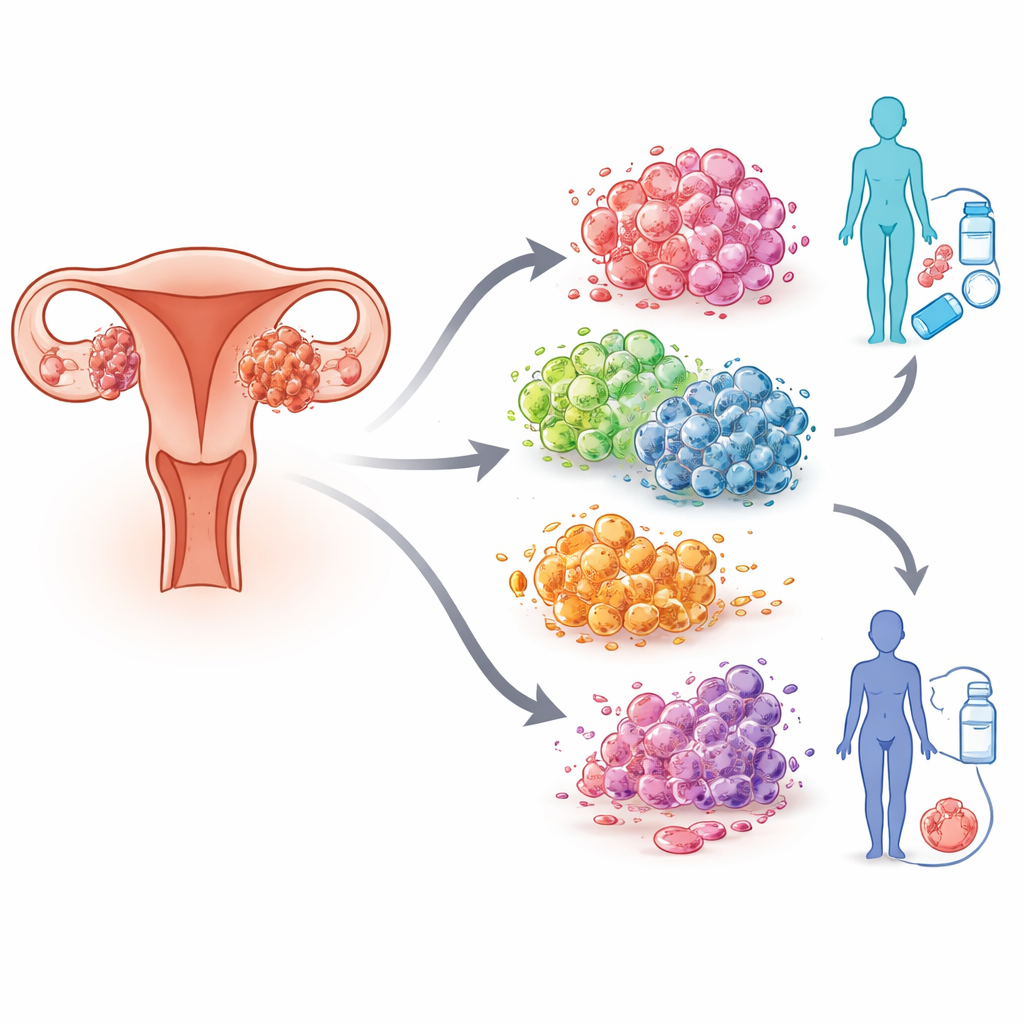
Different Ways for a Cell to Die
Cells do not all die the same way. Beyond simple damage and decay, many cells follow orderly “programs” of death that shape how our bodies grow, repair, and defend themselves. The team focused on 13 such programmed cell death routes, including familiar ones like apoptosis (a tidy form of self-destruction) and others with more exotic names, such as ferroptosis, pyroptosis, and newly described forms tied to metals or oxidative stress. Each route leaves its own molecular fingerprint. In tumors, these death programs can be hijacked, dulled, or flipped into states that help cancer cells evade the immune system, resist drugs, or spread.
Turning Gene Activity into a Risk Score
To capture these fingerprints, the researchers collected large gene-activity datasets from hundreds of ovarian tumor samples and compared them to normal ovarian tissue. They sifted through more than 24,000 genes associated with cell death, finding thousands that behaved differently in cancer. Using machine-learning tools, they narrowed this down to eight key genes whose combined activity best tracked with how long patients survived. From these eight genes they built a “cell death index,” or CDI, a single score that reflects the balance of death programs inside a tumor. Patients were then divided into high-CDI and low-CDI groups to test whether this score aligned with real-world outcomes.
Hidden Tumor Types and the Immune Landscape
When the scientists let the data group tumors on its own, three distinct molecular subtypes emerged, each with its own pattern of cell death genes and clinical behavior. One subtype was linked to poor survival, another to better outcomes, and each showed a different mix of immune cells in the tumor microenvironment—the community of cells, blood vessels, and signals that surround the cancer. Tumors with certain cell death profiles had fewer protective immune cells such as natural killer cells and cytotoxic T cells, hinting that disrupted death programs may help tumors hide from the body’s defenses. These patterns suggest that not all high-grade ovarian cancers are alike, even when they look similar under the microscope.
Predicting Survival and Drug Response
The CDI score proved to be a strong predictor of how patients fared. Across training and independent validation groups, those with a high CDI consistently had shorter overall survival than those with a low CDI. The team combined CDI with simple clinical information such as age and tumor grade into a graphical tool called a nomogram, which estimates an individual’s chance of living one, three, or five years after diagnosis. They also explored how CDI related to genetic mutations and drug sensitivity. Tumors with higher mutation burdens tended to have better outcomes, likely because they present more abnormal targets to the immune system. Meanwhile, the risk groups showed different predicted responses to a range of cancer drugs, suggesting that CDI could eventually help match patients to therapies they are more likely to benefit from, including targeted agents and immunotherapy.
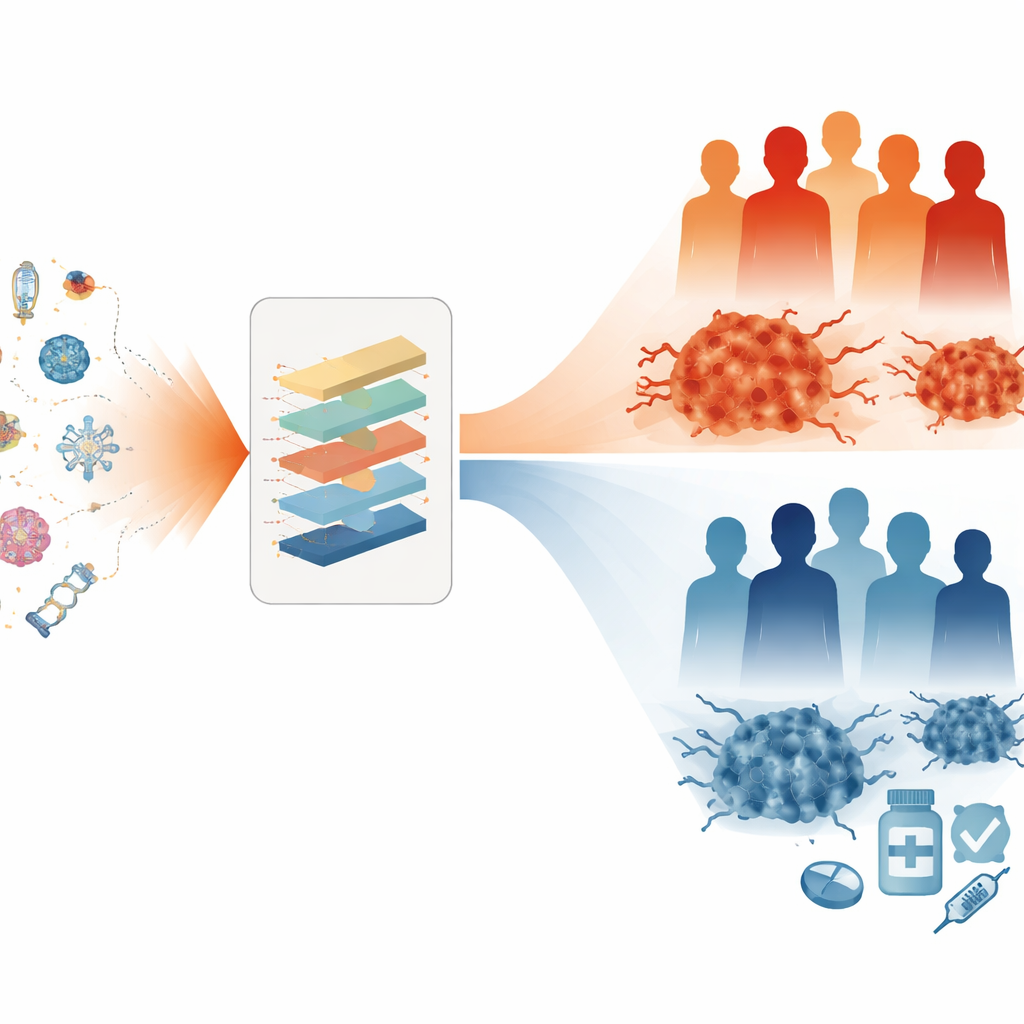
What This Could Mean for Patients
In plain terms, this study shows that reading a tumor’s “death signature” can offer powerful clues about a woman’s prognosis and how her ovarian cancer might respond to treatment. The eight-gene cell death index outperformed several previously published models and correlated with how active the immune system appeared within the tumor. While the work relies on existing datasets and still needs larger, prospective clinical testing, it points toward a future in which decisions about surgery, chemotherapy, and newer immunotherapies are guided not just by tumor size and stage, but by the inner life-and-death workings of the cancer cells themselves.
Citation: Li, XN., Wei, L., Shu-yi Wang et al. Analysing cell death patterns to predict outcomes and treatment options in patients with high-grade serous ovarian carcinoma. Sci Rep 16, 13132 (2026). https://doi.org/10.1038/s41598-026-42628-3
Keywords: ovarian cancer, cell death, prognostic model, tumor microenvironment, immunotherapy