Clear Sky Science · en
Hierarchical Co–Ni hydroxides integrated with carbon nanotubes via ZIF-67 templates for high-performance supercapacitors
Why better energy storage matters
From electric cars to backup power for homes, we increasingly rely on devices that can store energy quickly, safely, and for a long time. Supercapacitors are one such technology: they can charge and discharge in seconds and last for hundreds of thousands of cycles. But today’s supercapacitors still struggle to hold enough energy for many real‑world uses. This study explores a new way to build the tiny, structured materials that sit inside supercapacitors, with the goal of making them store more energy, charge faster, and survive years of repeated use.
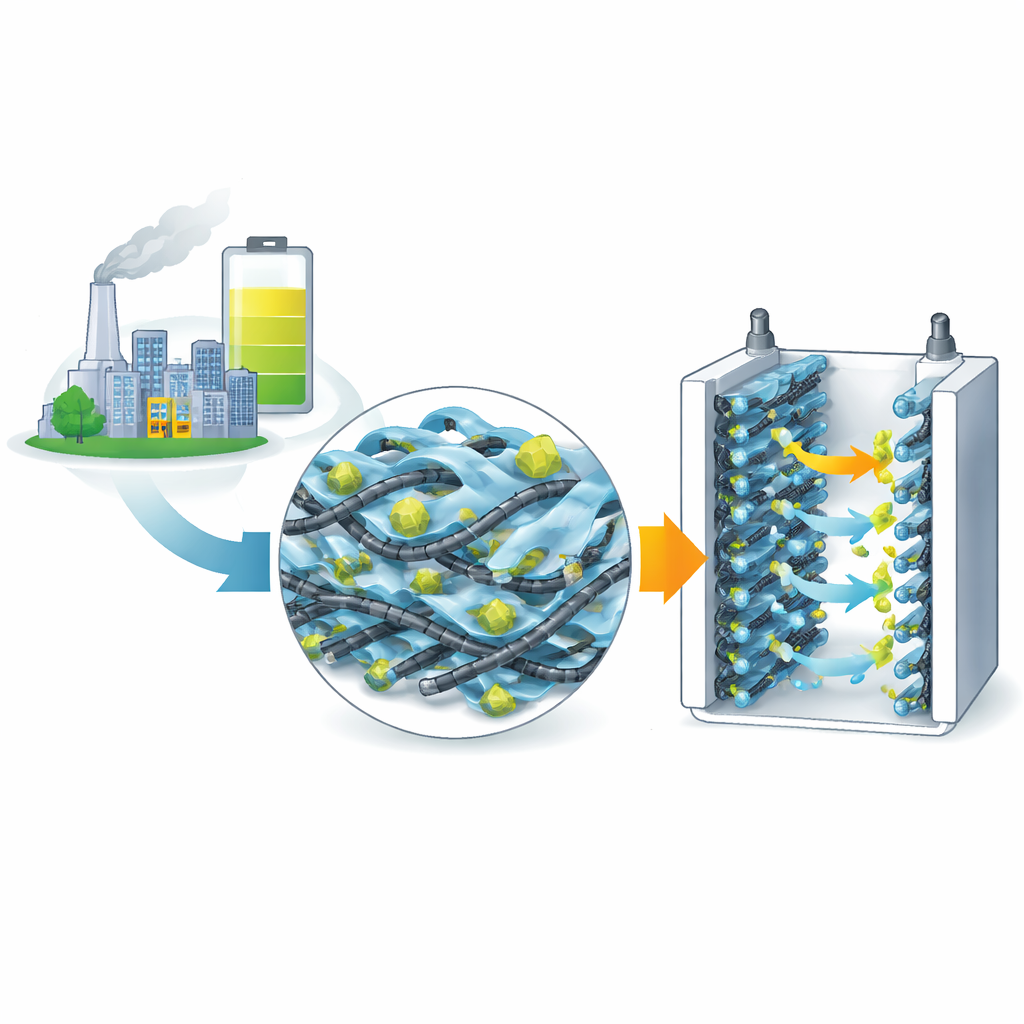
Building blocks for fast power
The heart of any supercapacitor is its electrode, the material that touches the liquid or gel electrolyte and actually stores charge. The researchers focused on combining three different ingredients that each bring something useful. First are cobalt–nickel layered double hydroxides, which are thin, sheet‑like structures rich in chemical sites that can temporarily hold charge through fast redox reactions. On their own, however, these sheets conduct electricity poorly and allow ions to move through them only slowly. Second are carbon nanotubes, tiny hollow carbon cylinders that offer excellent electrical pathways and mechanical strength. Third is a porous crystal called ZIF‑67, which acts like a template or scaffold to help shape the final material, while also adding enormous internal surface area.
A gentle recipe for a complex structure
Instead of using high‑temperature treatments that can damage delicate frameworks, the team designed a low‑temperature, solution‑based route to assemble these ingredients. They first treated commercial carbon nanotubes with acids so that their surfaces carried chemical groups able to anchor the metal hydroxide sheets. Then they grew cobalt–nickel hydroxide directly on the nanotubes, forming thin, interwoven nanosheets around the carbon network. In a final step, they introduced ZIF‑67 crystals, which served both as a source of cobalt and as a sacrificial mold that guided further growth. During this process, much of the ZIF‑67 was gradually consumed, leaving behind a highly porous, hierarchical network where nanotubes, metal hydroxide sheets, and remnants of the original framework were tightly integrated.
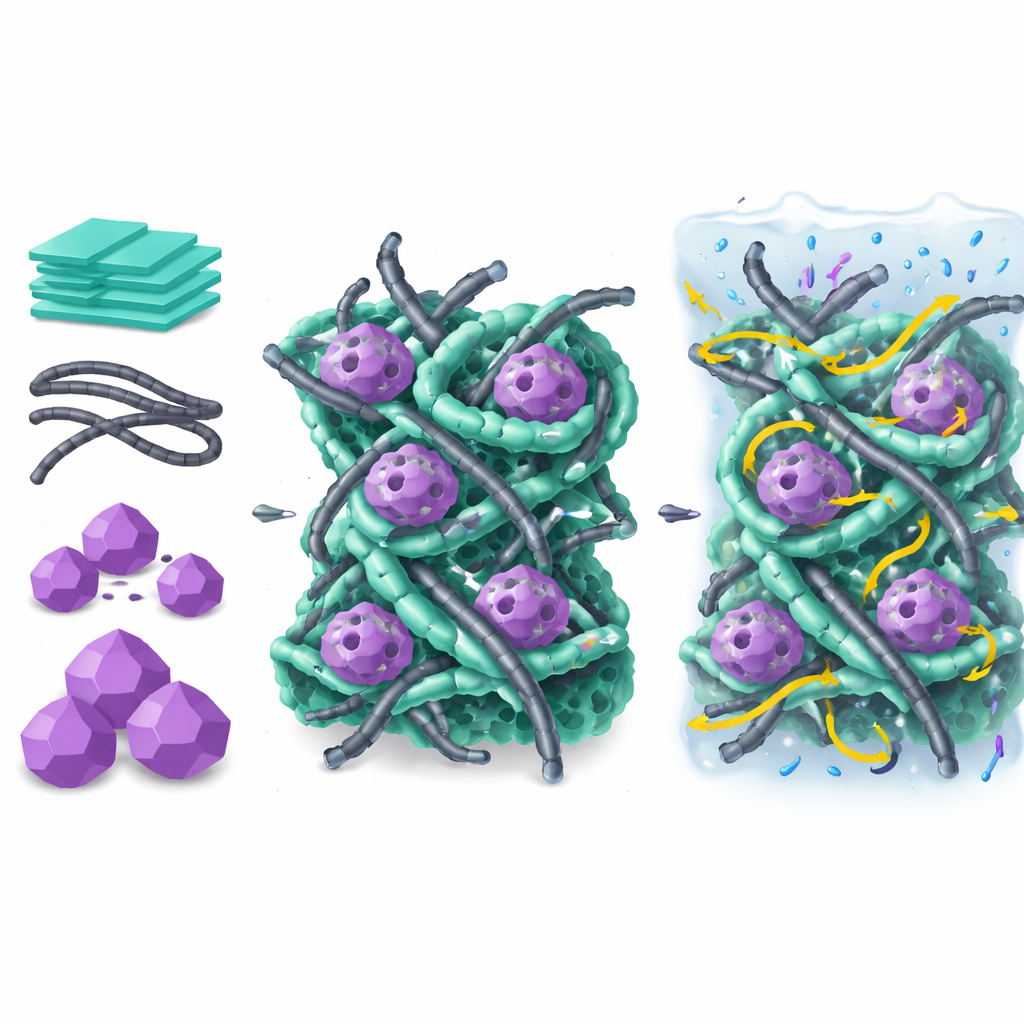
Seeing inside the new material
To confirm what they had made, the authors used a suite of structural and surface‑sensitive tools. X‑ray diffraction showed that the crystal structures of both the layered hydroxides and ZIF‑67 were successfully incorporated in the composite and remained stable, even as more carbon nanotubes were added. Electron microscopy images revealed ultrathin sheets draped over and between nanotube bundles, with small crystalline particles embedded in between—evidence of an intimately mixed network. Gas adsorption measurements demonstrated that the best samples, especially those containing both nanotubes and ZIF‑67, had extremely high internal surface areas and a well‑balanced mixture of small and medium‑sized pores. These pores act like a maze of tiny hallways, allowing electrolyte ions to reach active sites quickly while providing plenty of area on which to store charge.
From structure to performance
The real test was how well these materials performed as supercapacitor electrodes. In electrochemical measurements, simply adding carbon nanotubes to the cobalt–nickel hydroxide greatly boosted its ability to store charge and to keep working at high charging rates. The best nanotube‑based sample reached a specific capacitance of 870 farads per gram at a modest current and still retained 85 percent of that value at ten times higher current, indicating rapid charge and discharge. When ZIF‑67 was included in the design, performance improved even further. The top composite, called CNCZ30, delivered about 903 farads per gram and sustained 72 percent of its capacitance at high current. It also showed remarkably low resistance to charge flow and maintained 96.6 percent of its initial capacitance after 7,000 charge–discharge cycles, a sign of excellent long‑term stability.
What this means for future devices
For non‑specialists, the key message is that carefully arranging different nanoscale ingredients can produce electrodes that are both fast and durable. In this work, the carbon nanotubes act like highways for electrons, the cobalt–nickel hydroxide sheets supply abundant charge‑storing sites, and the ZIF‑derived framework opens up a vast interior landscape for ions to move through. Together, they create a “conductive‑redox” synergy that overcomes the weaknesses of each component alone. While these materials are still at the laboratory stage, their high capacity, strong rate performance, and long life suggest they could help power future generations of quick‑charging, high‑power energy storage devices used in everything from portable electronics to electric transport and grid‑level backup.
Citation: Gohr, M.S., Rafea, M.A., Wazeer, W. et al. Hierarchical Co–Ni hydroxides integrated with carbon nanotubes via ZIF-67 templates for high-performance supercapacitors. Sci Rep 16, 14226 (2026). https://doi.org/10.1038/s41598-026-42472-5
Keywords: supercapacitors, nanocomposites, carbon nanotubes, energy storage, electrode materials