Clear Sky Science · en
IRX4204 sensitizes multiple myeloma to ferroptosis and improves lenalidomide efficacy through the HMOX1-GPX4 axis
Why this research matters
Multiple myeloma is a cancer of antibody-producing cells in the bone marrow that remains incurable for most patients, largely because tumors eventually outsmart existing drugs. This study explores a newly appreciated form of cell death called ferroptosis—an iron-fueled kind of "rusting" inside cells—and shows how a highly selective experimental drug, IRX4204, can push myeloma cells toward this fate. By revealing how IRX4204 rewires the cell’s internal defenses and makes standard treatments like lenalidomide work better, the work points to a fresh angle for tackling drug-resistant myeloma.
A new way to kill cancer cells
Traditional cancer therapies often aim to trigger apoptosis, a tidy, programmed form of cell death. Myeloma cells, however, frequently learn to dodge this process. Ferroptosis offers a very different route: it relies on iron and the build-up of destructive fats in cell membranes, ultimately causing the cells to fall apart. Myeloma cells are especially dependent on iron and produce large amounts of antibodies, which puts them under constant oxidative strain. The authors reasoned that if they could tip the balance slightly further, these cells might become uniquely vulnerable to ferroptosis without harming normal tissues.
The drug that primes cells for iron-driven death
IRX4204 is a third-generation activator of retinoid X receptors (RXRs), master switches in the nucleus that control genes involved in metabolism and stress responses. The researchers treated several myeloma cell lines with IRX4204 and found that it slowed their growth and strongly increased their sensitivity to known ferroptosis-inducing compounds. A protective molecule called Ferrostatin-1, which blocks ferroptosis, largely rescued the cells from IRX4204’s toxic effects. This indicated that the drug was not simply poisoning the cells, but specifically pushing them into ferroptosis. When IRX4204 was combined with lenalidomide—a widely used myeloma drug—the two worked together to kill more cancer cells than either alone, and this added effect also depended on ferroptosis.
How the cell’s stress machinery is rewired
Digging into the mechanism, the team showed that IRX4204 chips away at the cell’s antioxidant shield. It lowers levels of GPX4 and SLC7A11, two proteins that normally prevent harmful fats from accumulating in membranes. At the same time, the drug increases free iron inside the cells and boosts lipid peroxidation, a form of oxidative damage tightly linked to ferroptosis. A key player turned out to be HMOX1, an enzyme that breaks down heme and releases iron. The researchers found that myeloma cells exposed to ferroptosis triggers switched on the HMOX1 gene, and that higher HMOX1 levels in patient samples were associated with better survival. IRX4204 was shown to activate a nuclear receptor pair, PPARα–RXRα, which binds directly to the HMOX1 gene’s control region and ramps up its activity.
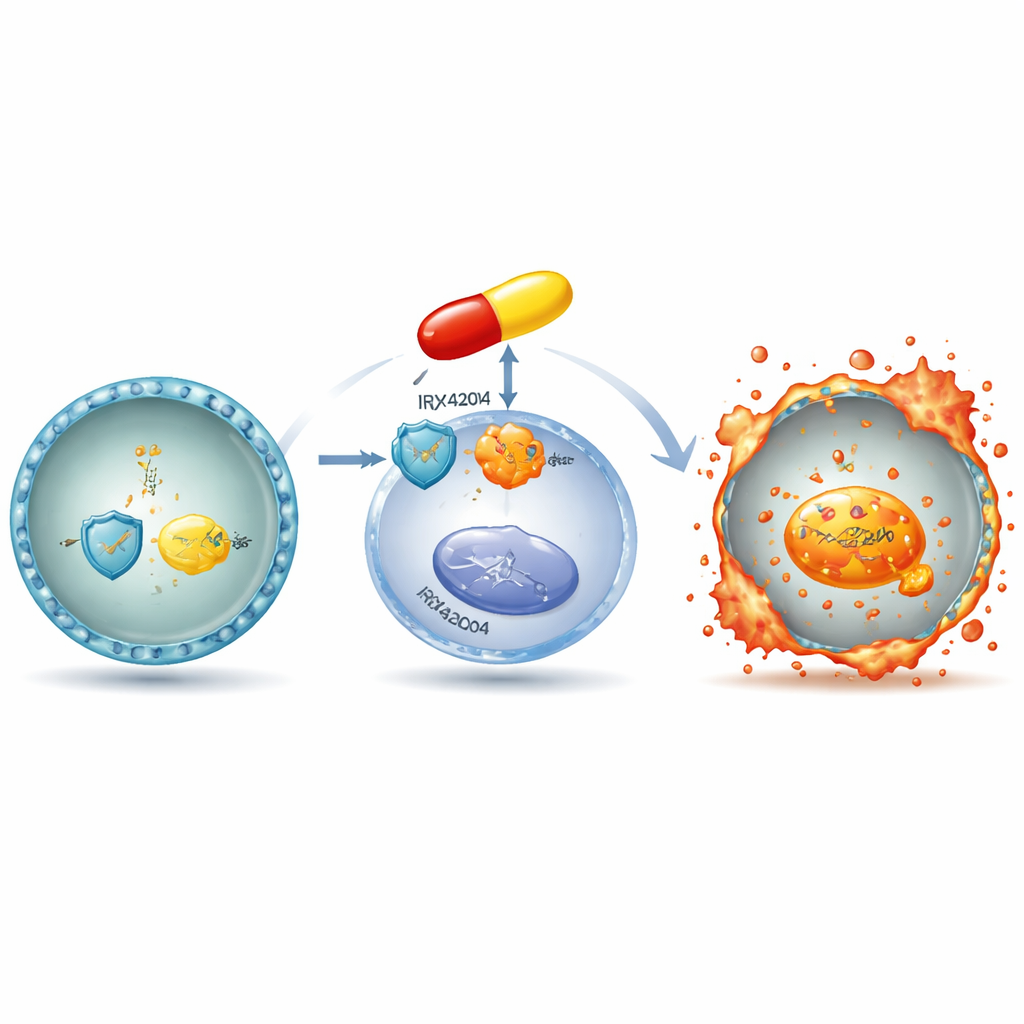
The crucial iron-releasing switch
To test whether HMOX1 is just a bystander or truly essential, the group used CRISPR gene editing to delete it from myeloma cells. Without HMOX1, IRX4204 could no longer efficiently lower GPX4 levels, nor could it drive iron accumulation and lipid damage. These edited cells were far less sensitive to ferroptotic death, demonstrating that HMOX1 sits upstream of the entire cascade. In mice carrying human myeloma tumors, treatment with IRX4204 plus lenalidomide shrank tumors more than either drug alone and extended survival, while also producing the same molecular signature seen in dishes: higher HMOX1 and lower GPX4 in tumor tissue, without obvious added toxicity.
What this could mean for patients
Taken together, the findings reveal a previously unrecognized control circuit—an RXR–HMOX1–GPX4 axis—that tunes how ready myeloma cells are to undergo ferroptosis. By turning this dial with IRX4204, the researchers were able to make standard therapy more effective and expose a weakness in otherwise resilient cancer cells. Although further work in immune-competent models and clinical trials is needed, the study suggests that carefully harnessing iron-driven cell death could complement existing treatments and help outmaneuver drug resistance in multiple myeloma.
Citation: Wu, J., Yan, Z., Burcher, K. et al. IRX4204 sensitizes multiple myeloma to ferroptosis and improves lenalidomide efficacy through the HMOX1-GPX4 axis. Sci Rep 16, 13832 (2026). https://doi.org/10.1038/s41598-026-42123-9
Keywords: multiple myeloma, ferroptosis, RXR agonist, HMOX1, lenalidomide