Clear Sky Science · en
Effect of pH on niacinamide skin permeation
Why this matters for your skin
Niacinamide shows up in countless serums and creams promising smoother, calmer, better-hydrated skin. Yet even a star ingredient only works if it can slip past the skin’s outer shield to where living cells reside. This study asks a deceptively simple question with big implications for everyday products: does the acidity, or pH, of a formula change how easily niacinamide gets through the skin barrier, and if so, why?
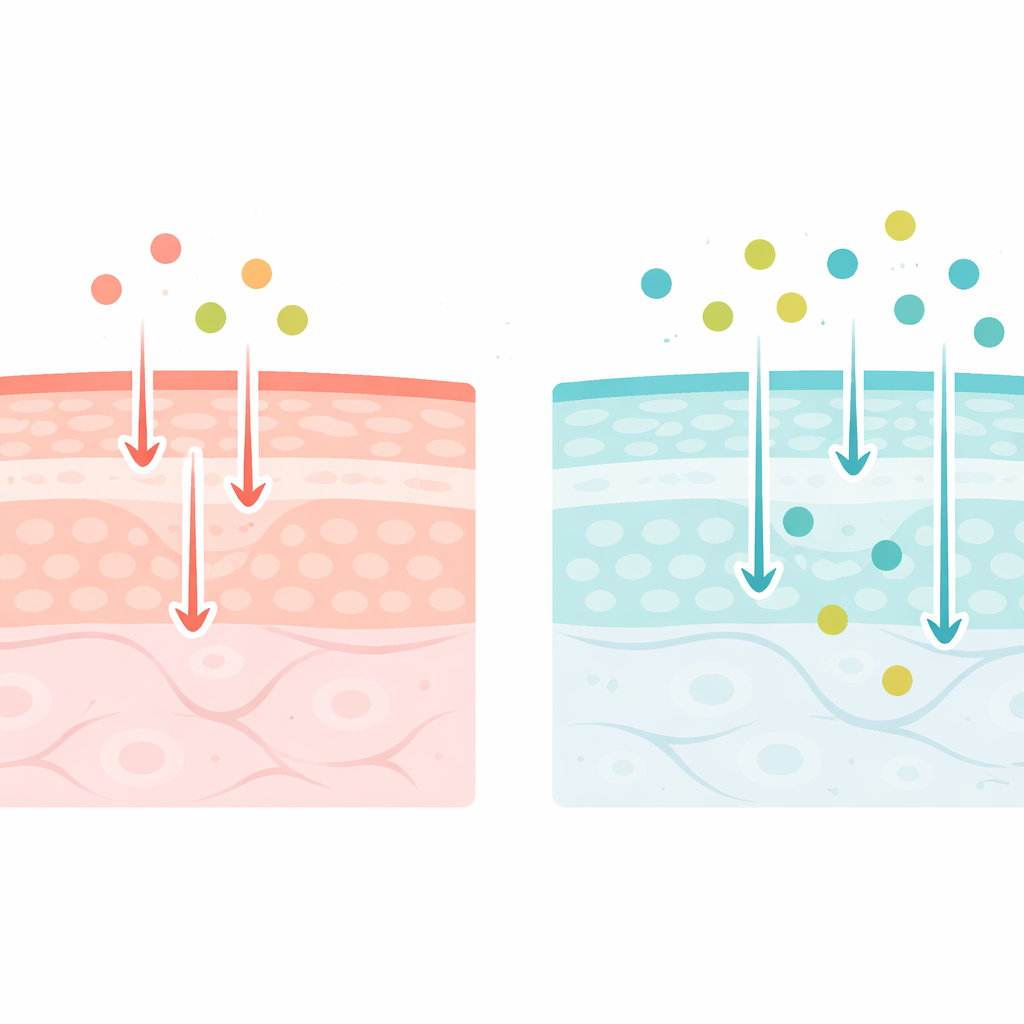
The skin’s shield and a familiar helper
The skin’s outermost layer, the stratum corneum, works like a brick wall: flattened dead cells are the bricks and a dense mix of fats is the mortar. This barrier keeps water in and irritants out, but it also slows down helpful ingredients. Niacinamide, a form of vitamin B3, is popular because it supports this barrier, reduces redness, and helps with fine lines and uneven tone. To do these jobs, it must cross the brick-and-mortar layer without disrupting it too aggressively. The surface of healthy skin is naturally mildly acidic—often called the “acid mantle”—and many products aim to respect or restore this acidity. The authors wanted to know whether tuning a formula to be closer to the skin’s natural pH changes how niacinamide travels across this wall.
Testing niacinamide at two everyday pH levels
Researchers compared niacinamide movement through samples of real human skin and lab-grown skin models. They used two water-based mixtures containing the same high niacinamide dose but with different acidity: one mildly acidic (pH 5.0, similar to the skin surface) and one closer to neutral (pH 7.4, like the fluid inside the body). Skin samples sat in diffusion cells that allowed the team to track how much niacinamide appeared on the far side over 24 to 48 hours. At the same time, they measured how easily electrical current passed through the skin, which reflects how tight or leaky the barrier is. By also repeating the experiment while switching the pH back and forth on the same piece of skin, they could see whether any changes were reversible rather than permanent damage.
Neutral formulas let more niacinamide through
The key finding is straightforward: neutral mixtures roughly doubled niacinamide permeation compared with acidic ones, in both real and reconstructed skin. When the researchers alternated the pH on the same skin sample, the barrier repeatedly became more permissive under neutral conditions and more restrictive again under acidic conditions, showing that the effect was reversible. Electrical measurements told a consistent story. At neutral pH, the skin’s resistance to electrical current dropped and its effective capacitance—linked to how charges and tiny ions arrange themselves in the barrier—increased. At acidic pH, resistance climbed and capacitance stayed low. Together, these patterns point to subtle structural shifts inside the fatty “mortar” rather than gross damage to the tissue.
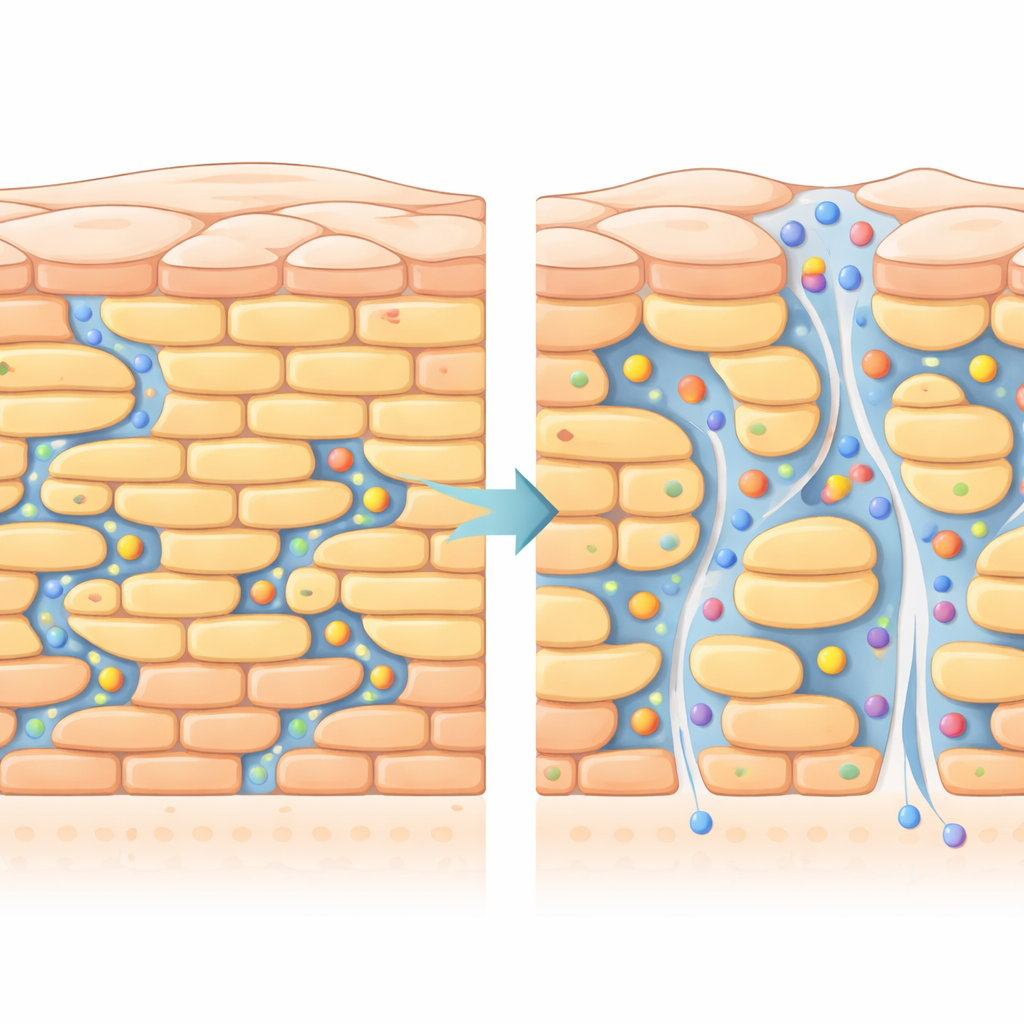
What may be changing inside the barrier
To explain these differences, the authors focused on a particular group of skin fats: free fatty acids. These molecules can carry or lose a positive charge depending on pH, which slightly alters how tightly they pack. At lower pH, they are mostly uncharged and help form a more rigid, well-ordered lipid matrix. At neutral pH, more of them become charged, which likely loosens the packing in small patches and creates microscopic defects and extra water-filled channels. These tiny structural imperfections make it easier for water-loving molecules like niacinamide and simple ions to move through, which is reflected in both higher niacinamide flux and lower electrical resistance. Calculations comparing experiment and theory suggest that transport occurs through a mix of lipid routes and narrow aqueous pathways; neutral pH seems to improve the efficiency of these routes rather than dramatically widening them.
The role of skin microbes over longer times
In longer experiments, the team noticed that niacinamide movement sometimes slowed slightly over time, which was unexpected under constant soaking. Careful chemical analysis revealed that a small fraction of niacinamide was being converted into nicotinic acid, another form of vitamin B3, but only when microbes were allowed to grow. When the researchers added a preservative that prevents microbial activity, this conversion disappeared. Although the amounts were tiny over 24 hours, the result shows that skin-dwelling bacteria can chemically modify niacinamide, a factor that could matter for products left on the skin for long periods or stored in conditions that favor microbial growth.
What this means for skincare users and formulators
Overall, the study concludes that the pH of a topical formula is a critical yet gentle lever for controlling how much niacinamide penetrates the skin. Neutral, water-based systems make the outer barrier slightly more open by reversibly rearranging lipids, leading to higher niacinamide delivery, while still leaving the barrier fundamentally intact. Mildly acidic formulas, closer to the skin’s natural surface pH, slow its passage. For everyday users, this means that not all niacinamide products are equal—how they are formulated, especially their pH and preservation system, can influence both how much niacinamide actually reaches deeper layers and how it might be transformed by the skin’s microbiome.
Citation: Sjöberg, T., Letasiova, S., Jankovskaja, S. et al. Effect of pH on niacinamide skin permeation. Sci Rep 16, 9821 (2026). https://doi.org/10.1038/s41598-026-41992-4
Keywords: niacinamide, skin barrier, pH, transdermal delivery, stratum corneum