Clear Sky Science · en
Integrating genetics, age and imaging to predict treatment outcomes in neovascular age-related macular degeneration: a proof-of-concept study
Why this matters for vision and aging
As people live longer, more of us are at risk of age-related macular degeneration, a disease that can steal central vision and make everyday tasks like reading or driving difficult or impossible. Doctors now have effective eye injections that can slow or halt one aggressive form of the disease, but patients differ widely in how well they respond and how often they need treatment. This study explores whether combining information from our genes, eye scans, and age can help doctors forecast who is most likely to benefit from therapy and how the disease will behave over time, opening the door to more personalized eye care.
Bringing three clues together
The researchers focused on neovascular age-related macular degeneration, the “wet” form driven by leaky new blood vessels under the retina. All 106 participants were treated at a single specialist eye hospital under a common “treat-and-extend” schedule of anti-VEGF injections, which are standard drugs used to control the disease. For each person, the team gathered three types of information routinely available in modern clinics: detailed cross-sectional scans of the retina (OCT scans), a blood sample for genetic testing, and simple demographic data such as age at the first injection. 
Reading the retina with artificial intelligence
OCT scans can reveal tiny pockets of fluid and other structural changes in the retina that signal active disease. Instead of relying on human graders, the team used a deep learning model to automatically measure the volume of several key features, including fluid within and beneath the retina and bulges under the light-sensing layer. These measurements served as objective indicators of how damaged or active the eye was at the start of treatment. The main goal was to predict whether an eye would be “dry” – essentially free of visible fluid – two years after treatment began, a state that reflects quiet disease and is straightforward to verify on imaging.
Genetic load as an added piece of the puzzle
The study also incorporated a polygenic risk score, a single number that summarizes the combined effect of many common genetic variants linked to macular degeneration. Rather than predicting treatment response on its own, this score mainly captures how strongly someone is predisposed to develop the disease and how early it might appear. In this cohort, people tended to have higher genetic risk than the general population, and those with higher scores tended to start treatment at a younger age, suggesting that genetic load brings on disease earlier in life even if it does not directly dictate how the eye reacts to injections.
How well the combined model performed
To test whether blending these clues improved forecasting, the researchers built machine-learning models that used different combinations of age, imaging, and genetic data. When they tried to predict a dry macula at two years using only the eye-scan features, performance was fair. When they added the genetic risk score and age, the model’s ability to distinguish between eyes that would end up dry and those that would not improved substantially, reaching a high level of accuracy by common statistical standards. 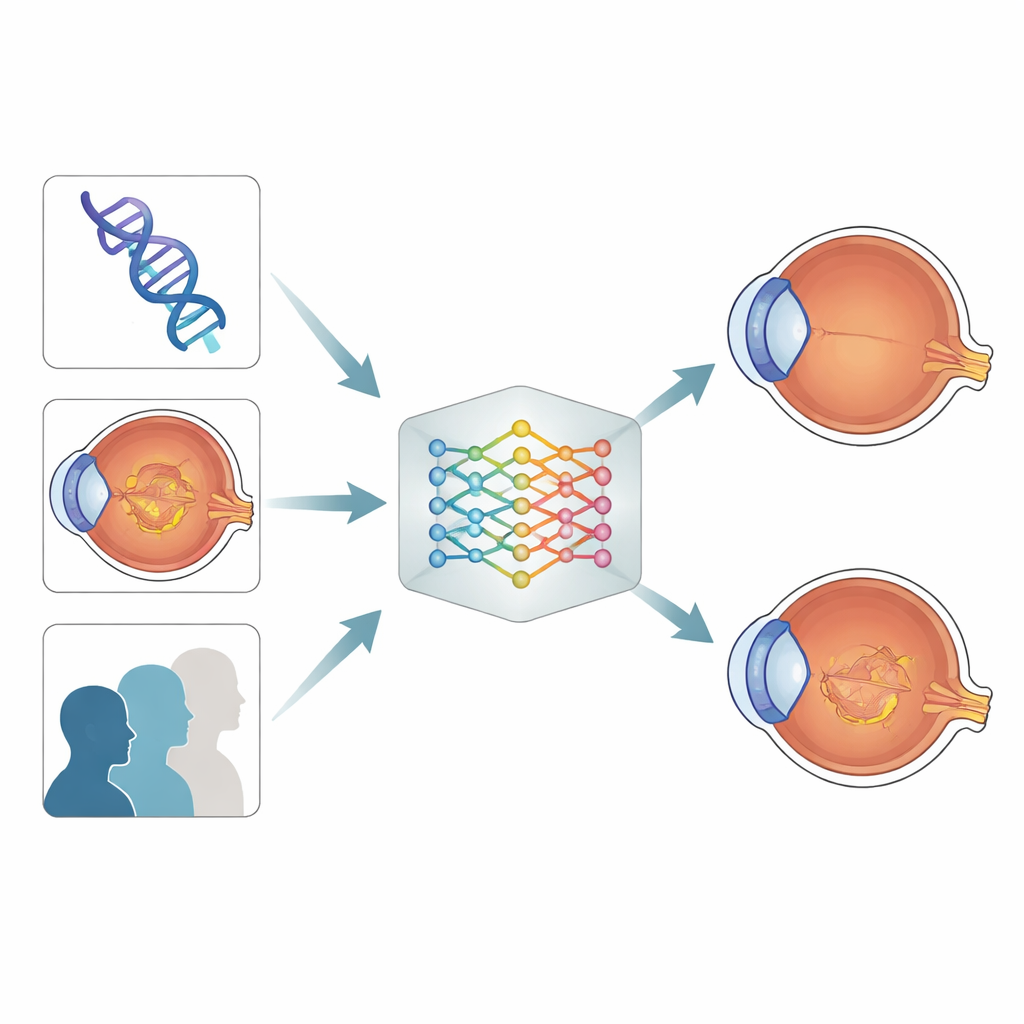
What this means for patients and the future
For people living with, or at risk of, macular degeneration, this proof-of-concept work suggests that a more tailored approach to treatment may be on the horizon. By combining a person’s inherited risk, the fine details of their retinal structure, and their age, doctors may eventually be able to estimate the chances of achieving a quiet, fluid-free macula and to plan follow-up accordingly. The current study is small and drawn from a single center, so its models are not ready for routine clinical use, and they focus on anatomical outcomes rather than day-to-day visual experience. Nonetheless, the results show that weaving genetics and advanced imaging into a single artificial-intelligence framework is technically feasible and biologically sensible, laying the groundwork for larger studies that could bring precision medicine to the front lines of eye care.
Citation: Moghul, I., Pontikos, N., Sharma, A. et al. Integrating genetics, age and imaging to predict treatment outcomes in neovascular age-related macular degeneration: a proof-of-concept study. Sci Rep 16, 12489 (2026). https://doi.org/10.1038/s41598-026-41931-3
Keywords: age-related macular degeneration, genetic risk score, retinal imaging, artificial intelligence, personalized eye treatment