Clear Sky Science · en
Evaluating effects of tissue type, preservation method, and decomposition on DNA quality to support genetic sampling in stranded small cetaceans
Why dolphin strandings can still tell vital stories
When a dolphin washes ashore dead, it is a tragedy—but also a rare scientific opportunity. These stranded animals allow researchers to collect tissue that can reveal how dolphin populations are doing, how they move, and how they are coping with pollution and climate change. Yet all of that depends on one fragile ingredient: intact DNA. This study asks a practical but crucial question: from a decaying dolphin on a beach, which tissues should we sample, and how should we store them, to get DNA good enough for modern genetic analyses?
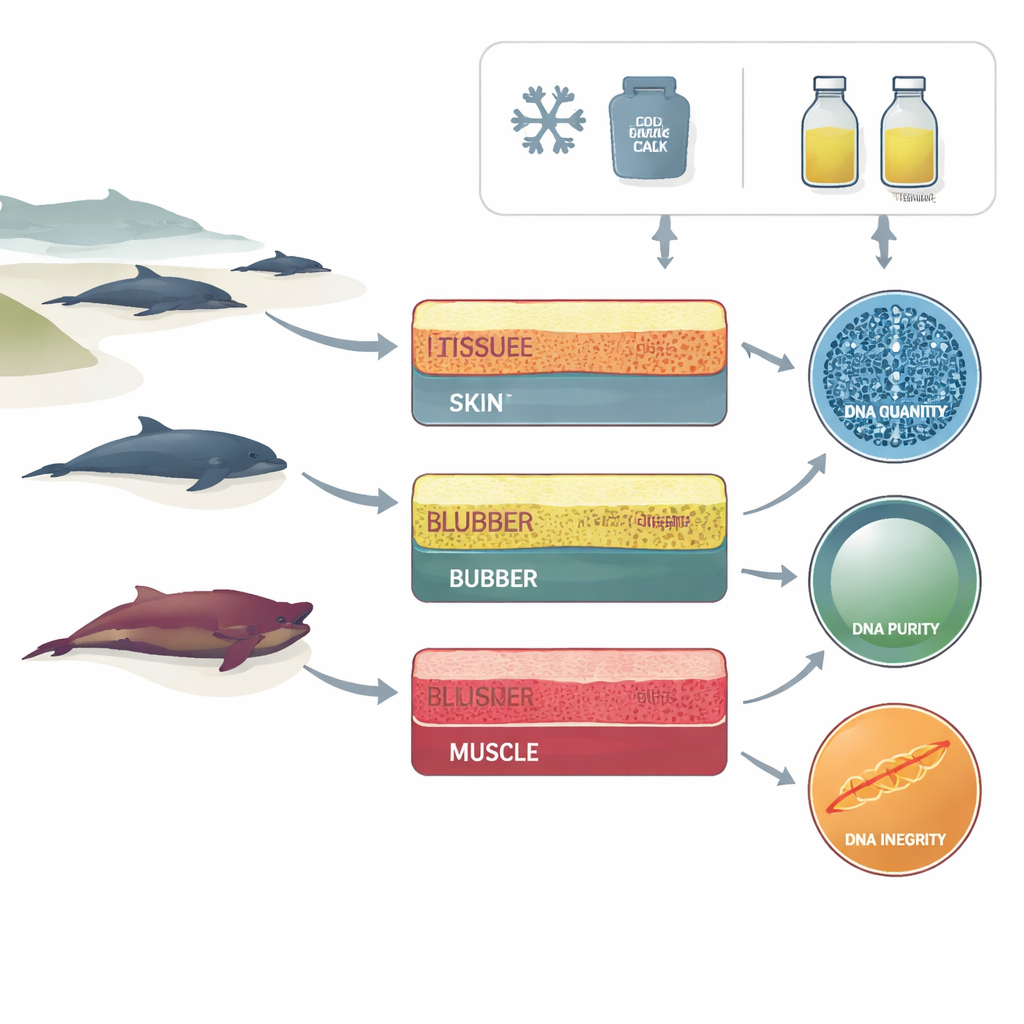
From beach to test tube
The researchers worked with a stranding network along the Portuguese coast, using carcasses of small dolphins that had died and come ashore in different conditions—from freshly dead to mummified remains. For each animal, they collected three types of tissue that are commonly available during necropsies: the outer skin, the fatty blubber beneath it, and the underlying muscle. Each tissue sample was then preserved in one of two simple ways that most field teams can realistically use: submerged in strong (96%) ethanol at room temperature, or kept frozen at about minus 20 degrees Celsius.
Measuring what makes DNA "good"
In the lab, the team extracted DNA from all these combinations of tissue, preservation method, and decomposition level. They then looked at three aspects of DNA quality. First was how much DNA they could obtain from a given piece of tissue. Second was "purity," which reflects how much the sample is contaminated by proteins, salts, or other chemicals that can interfere with analyses. Third was "integrity"—whether the DNA strands were long and relatively intact, or broken into short fragments. Using standard instruments, they measured concentration and purity and assigned each sample a DNA integrity number, a score that summarizes how fragmented the DNA is.
Skin for plenty, muscle for strength
The results showed a clear trade-off between how much DNA could be obtained and how intact it was. Skin was the clear winner for yield: across almost all decomposition stages and both preservation methods, skin produced much higher DNA concentrations than muscle or blubber. Skin samples also generally had good purity, meaning they contained few interfering substances. However, as carcasses became more decomposed, DNA in the skin broke down rapidly. In contrast, muscle produced less DNA overall but kept it in better shape at advanced decomposition stages, particularly when the tissue had been frozen. Blubber usually performed worst, with low DNA amounts and more contamination, though it still provided usable DNA when skin or muscle were unavailable.
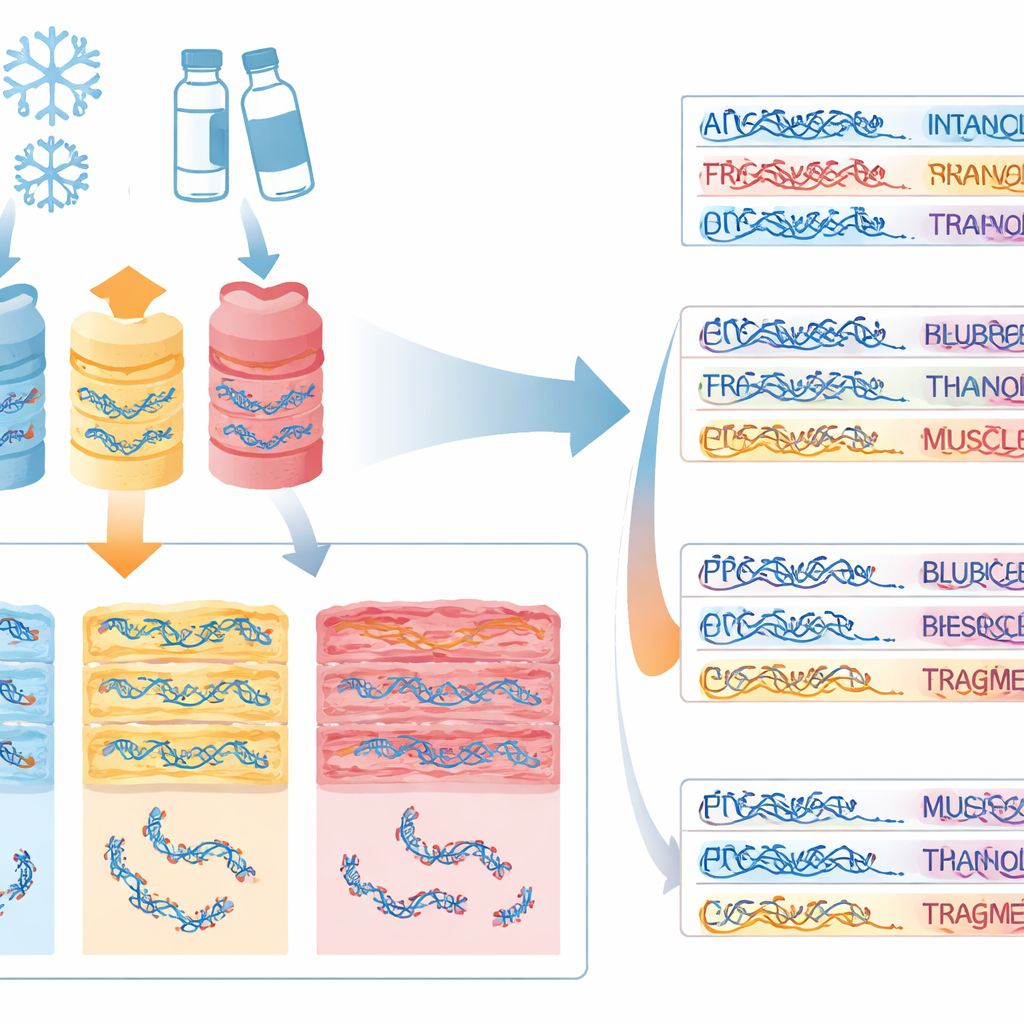
What happens as the body breaks down
As expected, DNA quality declined as decomposition advanced: fresher carcasses (early stages) had higher DNA concentrations and better integrity, while heavily decomposed ones (late stages and mummified remains) showed more fragmentation. Yet there were surprises. Muscle from some mummified carcasses that had been frozen actually showed relatively high DNA integrity, probably because the inner layers of tissue are shielded from sun, heat, and scavengers, and later drying can slow further decay. The study also found that how much DNA is present does not reliably predict how intact it is: a sample can be rich in DNA but still too broken for demanding techniques such as whole-genome sequencing.
Simple rules for real-world responders
Based on these patterns, the authors propose two practical frameworks: a detailed table for researchers planning specific genetic methods, and a simplified decision guide for stranding teams working in the field. For animals that are fresh to moderately decomposed, they recommend prioritizing skin, preserved either in ethanol or by freezing, as it offers high yield, good purity, and acceptable integrity. For carcasses in advanced decomposition or mummified condition, they advise focusing on deeper muscle, ideally frozen, to maximize the chance of obtaining DNA that is still long and intact enough for more sophisticated analyses.
What this means for dolphin conservation
By systematically testing how tissue type, preservation method, and decomposition stage interact, this study turns a messy reality—dead dolphins in varying states on remote beaches—into clear, evidence-based guidelines. For conservationists and geneticists, this means fewer wasted samples, more reliable data, and better comparisons between regions and years. For the dolphins, it means that even after death, their bodies can more effectively contribute to understanding population health, migration routes, and responses to human pressures, improving the scientific foundation for protecting these animals in a changing ocean.
Citation: Grilo, M.L., Leal e Rigor, M., Moura, A.E. et al. Evaluating effects of tissue type, preservation method, and decomposition on DNA quality to support genetic sampling in stranded small cetaceans. Sci Rep 16, 13555 (2026). https://doi.org/10.1038/s41598-026-41686-x
Keywords: cetacean genetics, dolphin strandings, DNA preservation, marine conservation, wildlife sampling