Clear Sky Science · en
Electrochemical determination of dihydroxybenzene isomers utilising poly-L-cystine-AgTiCrO2 nanohybrids
Why tracking tiny toxins in water matters
Many industrial products we rely on—such as dyes, pesticides, rubber, cosmetics, and some medicines—release small chemical compounds into air and water. Three closely related substances, catechol, hydroquinone, and resorcinol, belong to a family called dihydroxybenzenes. Even at very low levels, they can harm the heart, liver, kidneys, and DNA, and some are linked to cancer. Because they often appear together and look almost identical to standard instruments, scientists need smarter, more selective sensors to spot them quickly and cheaply in real-world samples like river water or cosmetics.
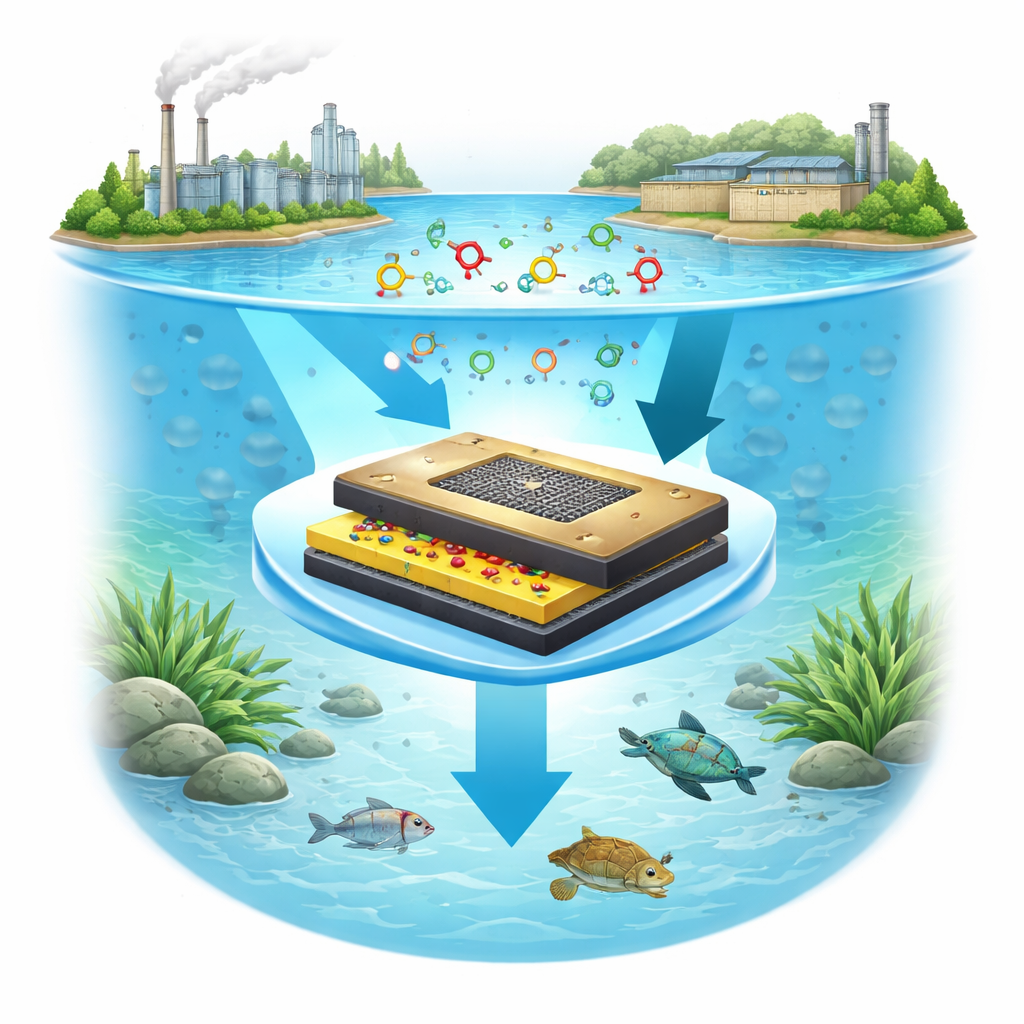
Building a new kind of tiny detector
The authors designed a new electrochemical sensor—essentially a tiny, wire-connected surface whose electrical response changes when certain molecules touch it. To make this surface highly sensitive, they built it from a layered "nanohybrid" structure: silver, titanium, and chromium oxides combined at the nanometer scale, supported on sheets of conductive carbon known as reduced graphene oxide, and coated with a thin polymer grown from the amino acid L-cystine. This stack is formed on a standard glassy carbon electrode, a common lab component, using a relatively simple “combustion” process to create the oxide nanohybrid, followed by solution coating and electrochemical film formation.
How the smart surface works
The sensor works by measuring how easily electrons flow when the three target chemicals are present in a water-based solution. Catechol, hydroquinone, and resorcinol can each undergo oxidation and reduction—reversible gain and loss of electrons—forming related structures called benzoquinones. The special surface layers help this electron exchange happen quickly and cleanly. The metal oxides provide abundant active sites and good electrical conductivity; graphene offers a large, conductive area; and the L-cystine–based polymer presents chemical groups that attract the pollutants and speed up electron transfer. Together, they make the sensor more responsive than a bare carbon electrode, which tends to foul, give overlapping signals, and miss very low concentrations.
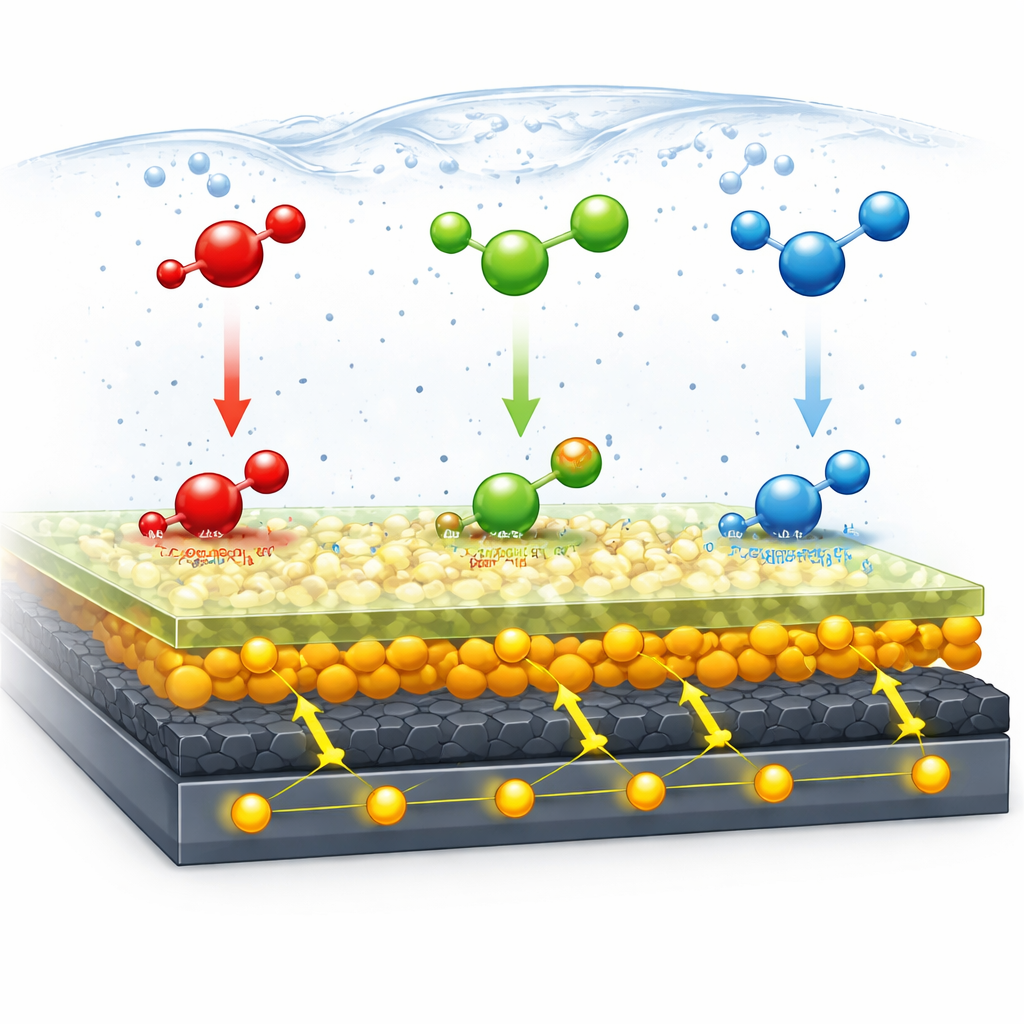
Tuning the sensor for real performance
To get the best performance, the team carefully adjusted several knobs. They optimized how much nanohybrid material was coated onto the electrode so that the film was thick enough to be active but not so thick that it blocked electron flow. They also tuned the formation of the L-cystine film, finding the number of electrochemical growth cycles that maximized signal without overbuilding the layer. Next, they studied how the acidity (pH) of the solution and the speed of the voltage scan influenced the current signals. These tests showed that the reactions involve a tightly coupled exchange of two electrons and two protons, and that the process is mainly controlled by how quickly molecules can diffuse to the surface. By choosing slightly acidic, near-neutral conditions, they achieved clear, well-separated peaks for each compound.
Seeing three look‑alike pollutants at once
In practical terms, the upgraded electrode could detect catechol, hydroquinone, and resorcinol at extremely low levels—down to tens to a few hundred parts per billion in solution—with a linear, easy-to-calibrate response over useful concentration ranges. Importantly, it could distinguish their signals when all three were present together, addressing a key challenge since these molecules are structurally very similar. Tests showed that common substances like salts, dyes, and urea caused only minor interference, and repeated measurements over time demonstrated good stability and reproducibility. The researchers also applied the sensor to commercial products containing related ingredients, such as a cosmetic cream and pharmaceutical formulations, and recovered realistic concentrations of the target chemicals.
From lab bench to cleaner environments
For a non-specialist, the main takeaway is that the authors have engineered a highly responsive, layered sensing surface that can spot three toxic, look‑alike pollutants at tiny concentrations in complex mixtures. By combining a cleverly designed metal-oxide nanohybrid with conductive carbon and a biologically inspired polymer, they improved sensitivity and selectivity beyond many existing devices. This kind of portable, low-cost electrochemical sensor could ultimately help regulators and manufacturers monitor water, cosmetics, and industrial streams more effectively, reducing exposure to harmful chemicals and contributing to safer products and cleaner environments.
Citation: Achar, S., Bhat, R.S., Sajankila, S.P. et al. Electrochemical determination of dihydroxybenzene isomers utilising poly-L-cystine-AgTiCrO2 nanohybrids. Sci Rep 16, 14340 (2026). https://doi.org/10.1038/s41598-026-41391-9
Keywords: electrochemical sensor, water pollutants, nanomaterials, environmental monitoring, phenolic compounds