Clear Sky Science · en
Natural low methoxyl pectin extracted from sunflower heads serves as an efficient biosorbent for lead removal
Turning Crop Waste into a Water Cleaner
Lead pollution in drinking water is a serious threat worldwide, and removing tiny amounts of dissolved lead is both technically difficult and expensive. This study explores an unexpected ally in the fight against lead: a type of natural gelling sugar called pectin, extracted not from fruit peels as usual, but from discarded sunflower heads. By carefully tuning how this pectin is extracted, the researchers show it can act as a powerful sponge for lead in water, pointing to a way to turn agricultural waste into a low-cost, sustainable water treatment material.
Why Lead in Water Is Hard to Remove
Lead ions in water are dangerous even at very low levels because they accumulate in the body and are hard to get rid of. Standard treatment methods—such as chemical precipitation, membrane filtration, and ion exchange—often require complex equipment, lots of energy, or generate secondary waste. Adsorption, where a solid material simply pulls contaminants out of water and holds them on its surface, is much simpler and can be very effective. The challenge is to find inexpensive, renewable materials that can grab lead strongly and in large amounts, without needing heavy chemical processing.
A New Life for Sunflower Heads
Sunflower heads, usually left in the field or discarded after seeds are removed, contain large amounts of pectin. Pectin molecules are rich in special chemical groups that can latch onto metal ions. The team compared three pectins: one extracted from sunflower heads at a relatively mild temperature (LHP), one extracted at a higher temperature for longer (AHP), and a typical commercial citrus pectin (CP). By heating sunflower heads more strongly, they produced AHP with shorter, more flexible chains and many exposed binding sites for metals. In contrast, the commercial citrus pectin had more of its binding sites blocked by small chemical “caps,” making it less able to hold lead.

How Structure Controls Lead-Grabbing Power
Although the sunflower pectins and citrus pectin had broadly similar basic building blocks, they differed in two key ways that determine how well they capture lead: how many active sites are available and how easy those sites are to reach. Both sunflower pectins naturally had a low level of chemical “capping,” leaving many negatively charged sites open to bind lead. However, AHP had much shorter chains than LHP, which reduced tangling between molecules and opened up its structure. Experiments showed that AHP could hold almost 296 milligrams of lead per gram of pectin—about one quarter more than LHP and three quarters more than citrus pectin. Tests of pH, temperature, and starting lead concentration all confirmed that AHP consistently outperformed the other two.
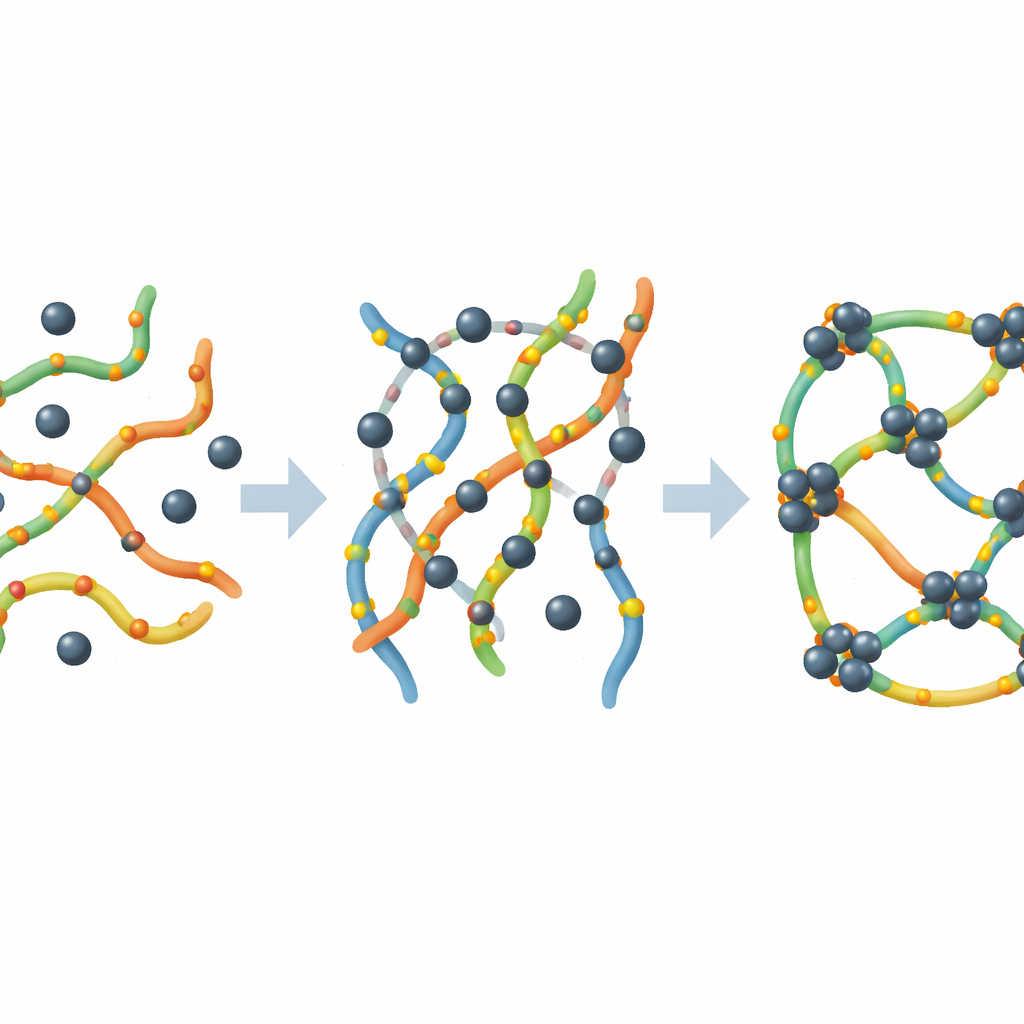
Watching Lead Bind and the Network Transform
To understand what was happening at the microscopic level, the researchers followed how lead interacted with pectin using several advanced techniques. Spectroscopy and surface analysis showed that lead attaches directly to oxygen-containing groups on the pectin chains, forming strong chemical bonds rather than just weak attraction. Microscopy images revealed that, as lead binds, the pectin chains reorganize themselves into a denser, more connected network, similar to a soft gel. Measurements of surface area and pore structure confirmed that this network actually becomes more internally textured as it traps lead, creating extra internal surfaces and tiny voids that help hold even more metal.
From Lab Mechanism to Real-World Potential
The study also tested how other common ions in water, such as calcium and aluminum, compete with lead for binding spots on pectin. Multicharged ions interfered the most, showing that real wastewater conditions will matter for performance. Even so, the sunflower pectin compared favorably with many chemically modified or composite pectin materials reported in previous work, despite being produced by a relatively simple extraction. The authors suggest that the next step is to embed this optimized pectin into solid beads, gels, or magnetic particles that can be easily separated and reused in treatment systems.
What This Means for Safer Water
In plain terms, the researchers discovered that the way you cook sunflower heads to extract pectin can turn an agricultural byproduct into a particularly effective lead sponge. Heating at higher temperature for longer shortens the pectin chains just enough to untangle them and expose more “grabbing hands” for lead, without destroying the material. This carefully tuned pectin forms a flexible, porous web that locks lead in place through strong chemical bonding. By showing that extraction conditions alone can optimize both the chemistry and the physical openness of this web, the work points to a practical, reagent-free strategy for transforming plant waste into powerful, greener tools for cleaning heavy metals from water.
Citation: Peng, X., Gong, Q., Gao, R. et al. Natural low methoxyl pectin extracted from sunflower heads serves as an efficient biosorbent for lead removal. Sci Rep 16, 11557 (2026). https://doi.org/10.1038/s41598-026-40672-7
Keywords: lead removal, sunflower pectin, biosorbent, water purification, agricultural waste reuse