Clear Sky Science · en
Nox4 and circulating free MPO synergistically promote aortic aneurysm formation
Why weak spots in the body’s main artery matter
Abdominal aortic aneurysms are dangerous balloon-like bulges in the main artery that runs through the belly. They often grow silently for years and can burst without warning, causing life-threatening internal bleeding. This study asks a basic but crucial question: what chemical forces inside the vessel wall turn a healthy aorta into a fragile one, and could understanding these forces point to new ways to prevent aneurysms?
Two harmful partners in the bloodstream
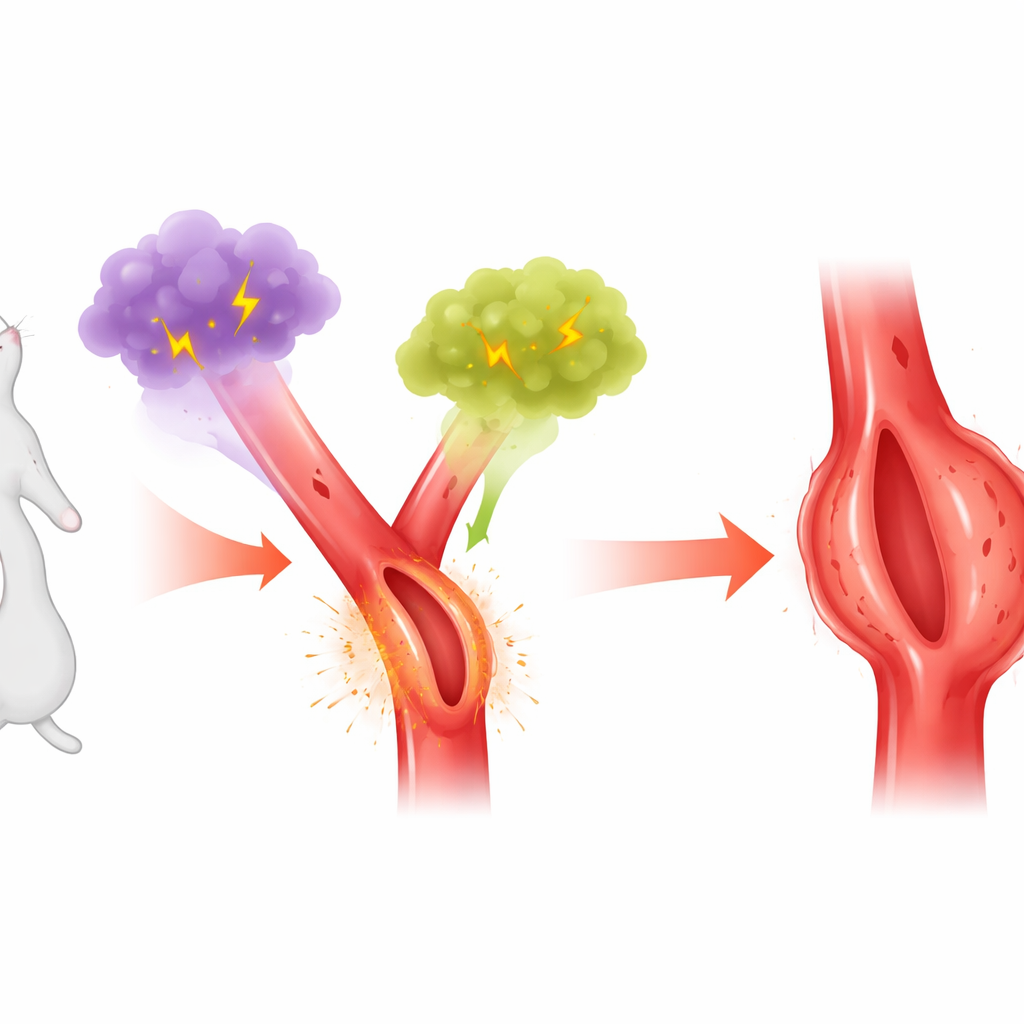
The researchers focus on a pair of enzyme systems that generate powerful oxidants—chemically reactive molecules that can damage tissues. One, called myeloperoxidase (MPO), is released from white blood cells during inflammation and can circulate freely in the blood. The other, Nox4, is produced by cells in the vessel wall and steadily generates hydrogen peroxide. MPO uses hydrogen peroxide as fuel to make even more aggressive oxidants. Because both have been linked separately to cardiovascular disease, the team wondered whether Nox4 and circulating MPO might work together in the abdominal aorta to drive aneurysm formation.
Watching enzymes gather in weak artery segments
Using established mouse models of abdominal aortic aneurysm triggered by the hormone angiotensin II or by applying calcium chloride around the aorta, the scientists first confirmed that aneurysm segments contained much more MPO protein and activity than normal aortas. Strikingly, this buildup was not due to local cells making extra MPO, since gene activity for MPO in the aortic wall did not rise. Instead, the pattern suggested that MPO circulating in the blood was being taken up into the vessel wall. When isolated aortas were briefly exposed to angiotensin II and then to purified human MPO in a dish, they absorbed substantially more MPO, especially during the early stages when aneurysms were just beginning to form.
A vessel enzyme that invites trouble in
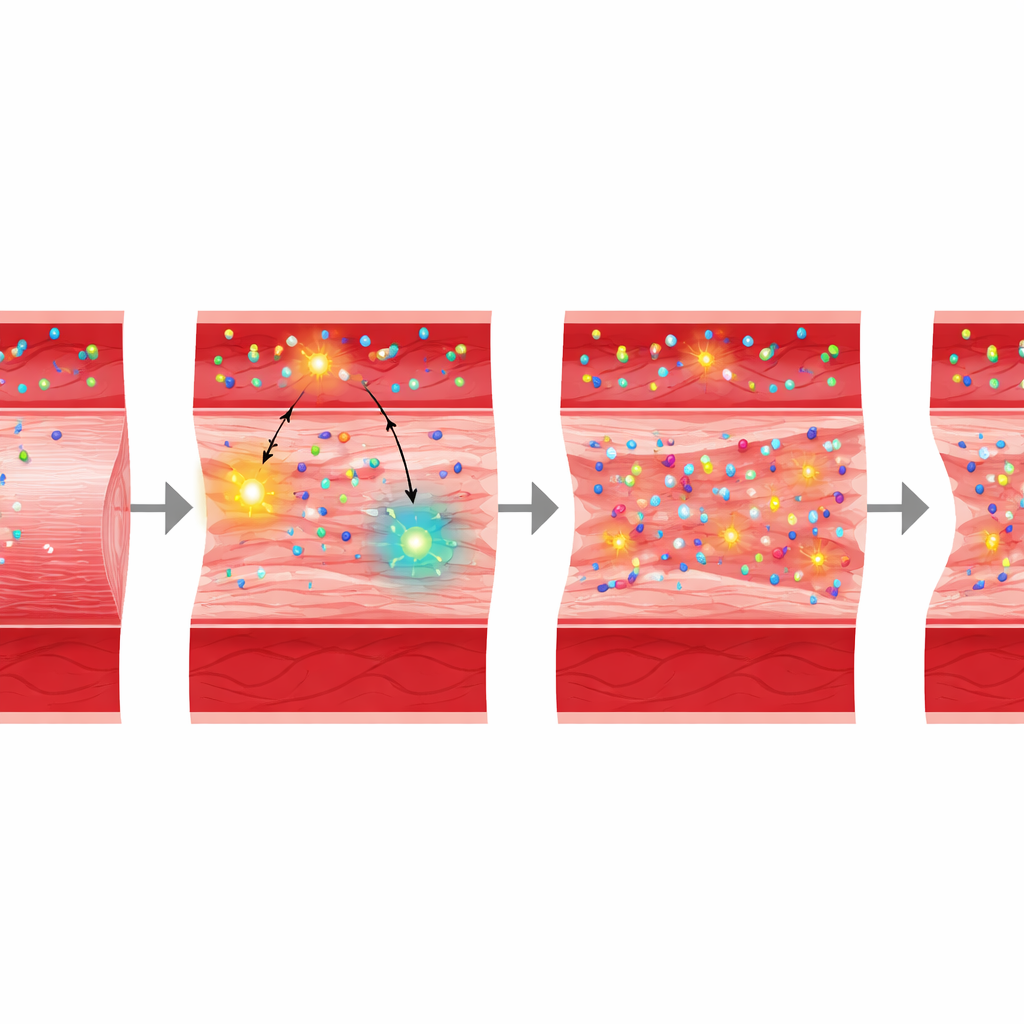
The team then examined Nox4, the vessel-wall enzyme that generates hydrogen peroxide. Angiotensin II or calcium chloride treatment boosted Nox4 levels and hydrogen peroxide in the abdominal aorta, precisely where aneurysms develop. When mice lacked Nox4, angiotensin II–treated aortas no longer showed the heightened uptake of MPO, and these animals were largely protected from aneurysm formation, despite still having other oxidant-producing enzymes. In the calcium chloride model, regions of the aorta that failed to increase Nox4 also failed to accumulate MPO or develop aneurysms. Together, these findings indicate that Nox4 activity in the vessel wall helps draw circulating MPO into the tissue and sets the stage for structural damage.
Proving circulating enzyme alone can weaken the artery
To test whether blood-borne MPO by itself can worsen aneurysms, the researchers turned to mice genetically unable to make their own MPO, which are normally resistant to aneurysm formation. They used a liver-targeted gene-delivery virus to make these mice produce human MPO that is released into the bloodstream, without changing levels of Nox4 in the aorta. This maneuver raised MPO in the liver, blood, and aorta, and restored susceptibility to aneurysms: treated mice showed larger aortic diameters, more clots, more breakdown of the elastic fibers that give the aorta its spring, and early signs of mitochondrial damage in aortic cells. Notably, these changes occurred without major shifts in blood pressure or broad inflammatory signals, underscoring a direct role for circulating MPO protein in damaging the vessel wall.
What this means for protecting the aorta
This work reveals a harmful partnership between a vessel-wall oxidant source and an inflammatory enzyme circulating in the blood. Nox4 in the aortic wall generates hydrogen peroxide that both fuels MPO chemistry and appears to promote the physical uptake of MPO into the tissue. Once inside, MPO intensifies oxidative stress, degrades the supporting matrix, and contributes to the weakening and ballooning of the abdominal aorta. For patients, the findings highlight circulating MPO as more than a passive blood marker—it may be an active driver of disease—and point to Nox4 and MPO together as potential dual targets for drugs aimed at preventing or slowing abdominal aortic aneurysms.
Citation: Coffey, P., Horimatsu, T., Kim, D. et al. Nox4 and circulating free MPO synergistically promote aortic aneurysm formation. Sci Rep 16, 10320 (2026). https://doi.org/10.1038/s41598-026-40599-z
Keywords: abdominal aortic aneurysm, oxidative stress, myeloperoxidase, Nox4, vascular inflammation