Clear Sky Science · en
Tissue and maturation specific DNA methylation dynamics of gonadotropin genes in chub mackerel (Scomber japonicus) using cost-effective targeted bisulfite sequencing
Why fish fertility matters to all of us
Seafood on our plates and healthy ocean ecosystems both depend on fish that can reproduce reliably, even as waters warm and human-controlled farms expand. This study looks inside the bodies of chub mackerel—a key commercial species—to see how tiny chemical tags on DNA help switch fertility hormones on and off as fish grow up. The work also introduces a faster, cheaper method to track these DNA marks, opening the door to smarter, more sustainable aquaculture.
Tiny switches on DNA
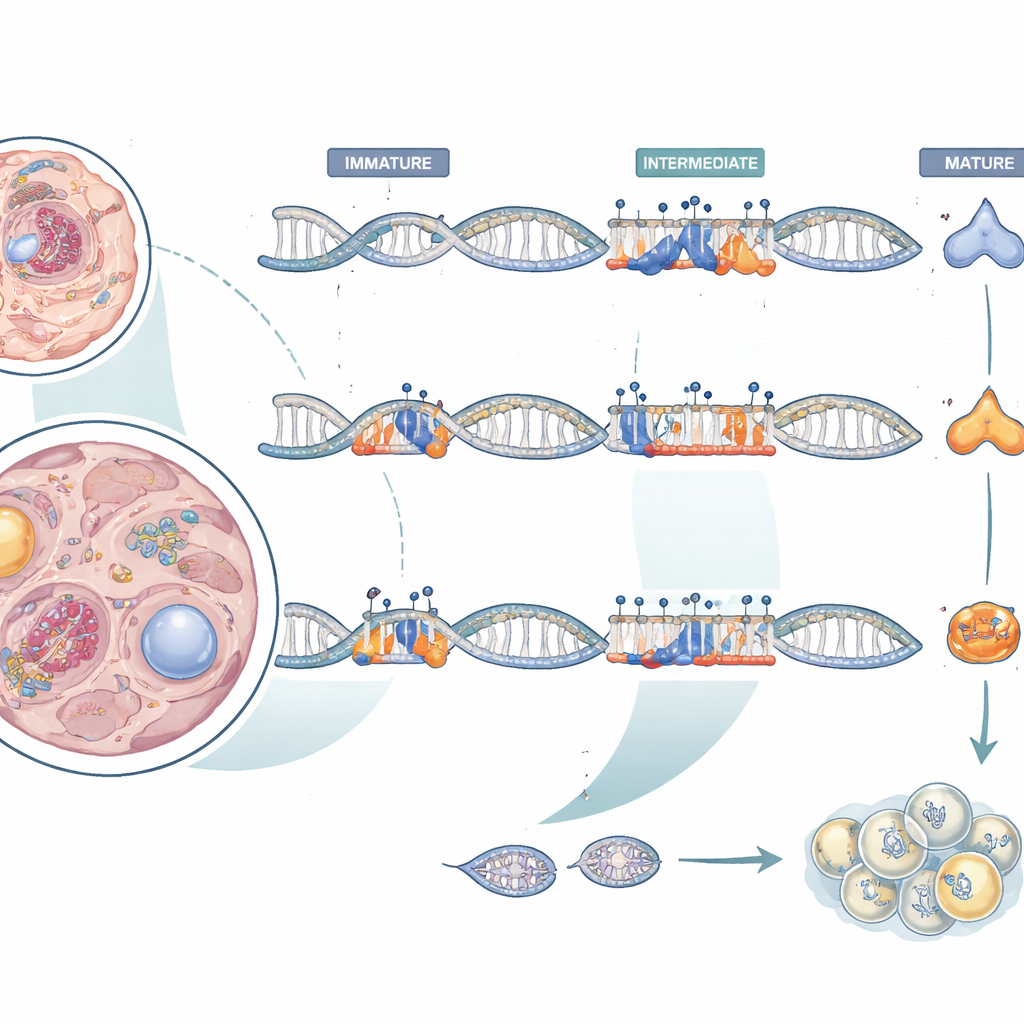
Our genes are wrapped in an extra layer of control made of chemical tags that can turn activity up or down without changing the genetic code itself. One such tag, called DNA methylation, often acts like a dimmer switch: when it is added near a gene, that gene tends to quiet down. The authors focused on two hormones made in the brain’s pituitary gland—follicle-stimulating hormone (FSH) and luteinizing hormone (LH)—which together orchestrate egg and sperm production. By measuring methylation around the DNA for these hormones in many tissues and life stages of farmed chub mackerel, they asked how this chemical code shifts as fish move from youth to sexual maturity.
A faster way to read the chemical code
Reading methylation in detail is usually slow and costly. Traditional approaches sequence one DNA fragment at a time, which limits how many animals or tissues can be analyzed. Here, the team adapted a targeted bisulfite sequencing method, originally developed for plants, to fish. They attached short identifying tags to many DNA fragments and ran them together on a high-throughput sequencer, then used software to sort everything back by sample and target region. This allowed them to analyze 2,880 DNA targets from 96 fish across five tissues—gonad, liver, brain, hypothalamus, and pituitary—at a fraction of the cost and effort of classic methods, yet with enough depth to detect subtle methylation differences.
How hormone genes change as fish mature
The big-picture pattern was that methylation around the FSH and LH genes depended strongly on both tissue type and whether fish were immature or in the breeding season. In most tissues, methylation near the FSH gene stayed high and fairly stable. But in immature gonads it was lower, and in mature pituitaries specific sites lost methylation, making this tissue the least methylated and the most active in FSH production. The LH gene showed the opposite trend in the pituitary: the region within the gene itself was more methylated in mature fish, even though LH levels were higher. This finding reinforces that methylation is not a simple “off switch”—its effect depends on where it lands and which proteins are trying to bind nearby.
A hidden brake inside the hormone gene
To probe how local DNA features shape LH output, the researchers tested short stretches of the LH gene in cultured cells using a light-producing reporter assay. Removing a tiny ten-base segment inside the first intron—a noncoding stretch within the gene—caused the reporter signal to rise, suggesting that this piece normally acts as a brake on gene activity. This segment overlaps a predicted binding site for a common regulatory protein known as Sp1. Interestingly, the nearby DNA methylation at this spot was generally low and did not change much with maturation, hinting that the brake’s strength may be tuned either by small, hard-to-detect methylation shifts or by other nearby control sites. The team also measured genes that add and remove methylation marks and found their activity changed with maturation in a tissue-specific way, pointing to active reshaping of the chemical code rather than simple aging.
What this means for fish and for farming
Putting these findings together, the study shows that the chemical tags on DNA around key fertility hormones shift in specific tissues as chub mackerel approach breeding, and that a small internal element within the LH gene can act as a built-in brake on hormone production. At the same time, the improved sequencing method proves that it is now practical to survey these epigenetic patterns in hundreds of targets and many individuals. For non-specialists, the takeaway is that fish fertility is controlled not only by genes but also by a flexible chemical layer that can respond to internal state and perhaps to the environment. Understanding and eventually guiding this layer could help breeders produce healthy, reliably spawning fish while easing pressure on wild stocks.
Citation: Galotta, M., Ogino, Y., Nagano, N. et al. Tissue and maturation specific DNA methylation dynamics of gonadotropin genes in chub mackerel (Scomber japonicus) using cost-effective targeted bisulfite sequencing. Sci Rep 16, 12222 (2026). https://doi.org/10.1038/s41598-026-40580-w
Keywords: fish reproduction, DNA methylation, gonadotropin hormones, epigenetics in aquaculture, targeted bisulfite sequencing