Clear Sky Science · en
Assessment of lipid mediators in the urine of patients with Lyme disease, tick-borne encephalitis and human granulocytic anaplasmosis
Why pee can reveal hidden tick infections
Tick bites are often shrugged off as a minor nuisance, yet the germs they carry can quietly trigger serious brain and nerve problems. Diagnosing these illnesses early is tricky because symptoms are vague and blood or spinal fluid tests are invasive and slow. This study explores a simple but powerful idea: could ordinary urine, rich in chemical traces from the whole body, reveal telltale “smoke signals” of tick-borne infections and help doctors tell different diseases apart?
Tick bites and puzzling illnesses
Ticks can transmit several pathogens at once, including the Lyme disease bacterium Borrelia burgdorferi, the tick-borne encephalitis (TBE) virus, and the bacterium that causes human granulocytic anaplasmosis (HGA). These infections often start with flu-like complaints, fatigue, or a rash that may be missed, making it hard to pinpoint the cause. If untreated, Lyme disease can spread from a skin rash called erythema migrans (EM) to the nervous system, causing neuroborreliosis (NB). TBE can progress from a mild fever to brain inflammation. Because these conditions overlap in symptoms and can occur together in one patient, clinicians urgently need better tools to untangle which infection is present and how the body is responding.
Fats, stress, and chemical messengers
When germs invade, the body’s defense system ramps up production of highly reactive molecules called reactive oxygen species. In moderation, they help fight infection; in excess, they damage cell components, especially membrane fats. This process, known as oxidative stress, chops long-chain fats into smaller, reactive fragments and also activates enzymes that remodel membrane lipids. The result is a burst of fatty messengers, including endocannabinoids and eicosanoids, which help steer inflammation and immunity. Because these molecules and their breakdown products are eventually filtered by the kidneys, changes in their levels can be detected in urine, offering a non-invasive snapshot of what is happening inside tissues like the brain, skin, and blood.
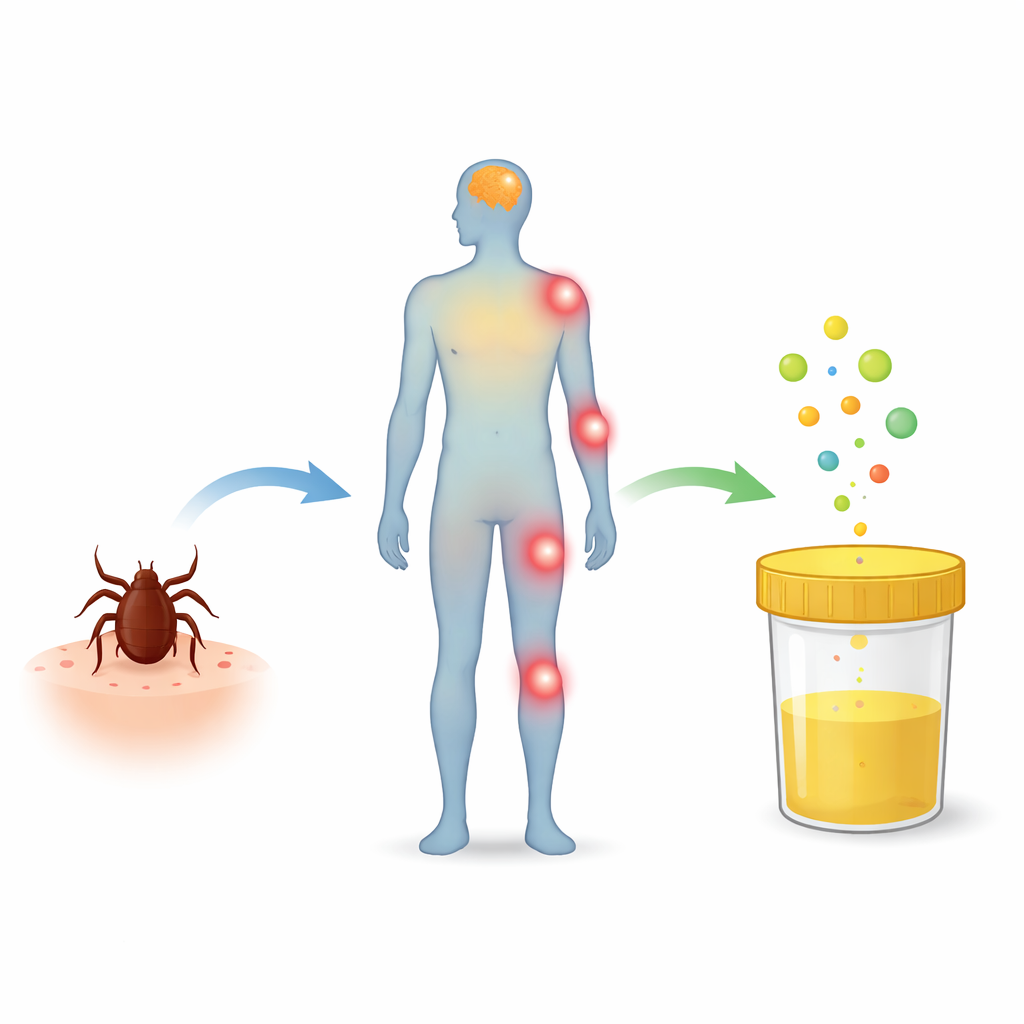
How the study was done
The researchers collected urine from adults with several confirmed tick-borne conditions: early Lyme disease with EM, Lyme neuroborreliosis, TBE, HGA, and co-infection with TBE and Lyme, as well as from healthy volunteers. Samples were taken before treatment and again after therapy. Using highly sensitive mass spectrometry methods, they measured markers of fat damage (such as 8-isoprostanes and 4-hydroxy-2-nonenal) and a panel of lipid messengers: endocannabinoids like oleoylethanolamide (OEA) and palmitoylethanolamide (PEA), and eicosanoids such as prostaglandins, leukotrienes, and hydroxyeicosatetraenoic acids (HETEs). They then looked for consistent differences between patient groups and healthy controls, and whether treatment shifted these chemical profiles.
Distinct chemical fingerprints in urine
Before treatment, patients with early Lyme, neuroborreliosis, or TBE all showed higher urinary levels of 8-isoprostanes than healthy people, signalling increased oxidative stress and fat peroxidation. Among the endocannabinoids, OEA stood out: its level was significantly higher in patients with TBE than in healthy controls, hinting that the body may be boosting anti-inflammatory and antioxidant defenses in this viral brain infection. For the eicosanoids, the team saw a rise in the pro-inflammatory prostaglandin PGE2 in TBE patients, and shifts in other mediators such as leukotriene LTD4 and 5-HETE between different Lyme-related groups, as well as changes in anti-inflammatory molecules like PGD2 and 15-HETE. These patterns suggest that each infection – and even different stages of Lyme disease – leaves a subtly different “lipid signature” in urine.
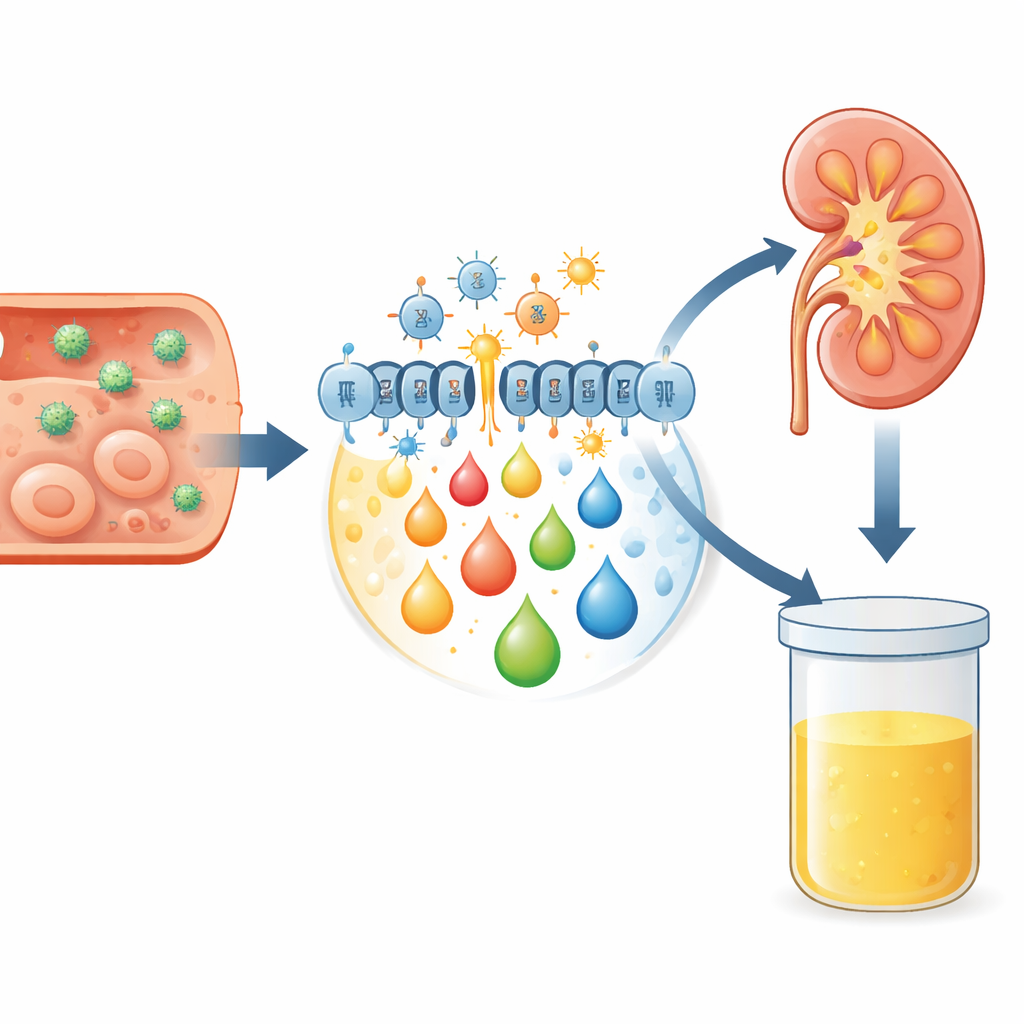
What the patterns do – and don’t – show
Interestingly, when the same patients were tested again after treatment, the overall profiles of these lipid mediators did not change in a statistically clear way, either compared with their own pre-treatment levels or between disease groups. That stability may reflect the small number of participants, especially for HGA, or lingering metabolic changes that outlast symptoms. It also underscores that these molecules are part of complex, overlapping pathways that respond not only to the pathogen itself but also to the body’s attempt to contain damage and restore balance. Nonetheless, the consistent elevation of specific markers like 8-isoprostanes and OEA in certain infections supports their role as indicators of oxidative stress and immune activation.
Why this matters for patients
For people facing confusing tick-borne symptoms, this research offers a glimpse of a future in which a simple urine test could help distinguish between Lyme disease, TBE, anaplasmosis, or co-infections, and perhaps flag disease stage or severity. The study shows that tick-borne infections disturb fat metabolism in ways that can be detected outside the body, without needles or spinal taps. At the same time, the authors stress that their patient group was small and that treatments varied, so these results are a promising first step rather than a ready-made diagnostic tool. Larger studies will be needed to confirm whether urinary lipid mediators can reliably guide diagnosis and monitoring, but the work highlights urine as a surprisingly informative window into how the body fights back against tick-borne threats.
Citation: Biernacki, M., Skrzydlewska, E., Dobrzyńska, M. et al. Assessment of lipid mediators in the urine of patients with Lyme disease, tick-borne encephalitis and human granulocytic anaplasmosis. Sci Rep 16, 11418 (2026). https://doi.org/10.1038/s41598-026-40464-z
Keywords: tick-borne diseases, Lyme disease, tick-borne encephalitis, lipid mediators, oxidative stress