Clear Sky Science · en
Experimental and theoretical assessments of an innovative star-shaped polyamine surfactant designed for X-65 steel corrosion mitigation in acidic environment
Protecting Metal from Hidden Damage
From oil pipelines deep underground to the steelwork inside cars and bridges, modern life depends on carbon steel. Yet this common material quietly dissolves whenever it meets strong acids, such as those used to clean and descale equipment in the petroleum industry. The study reported here explores a new way to shield steel from such attack: a custom-built, star‑shaped molecule that clings to metal and forms a microscopic raincoat, dramatically slowing corrosion in harsh acidic conditions.
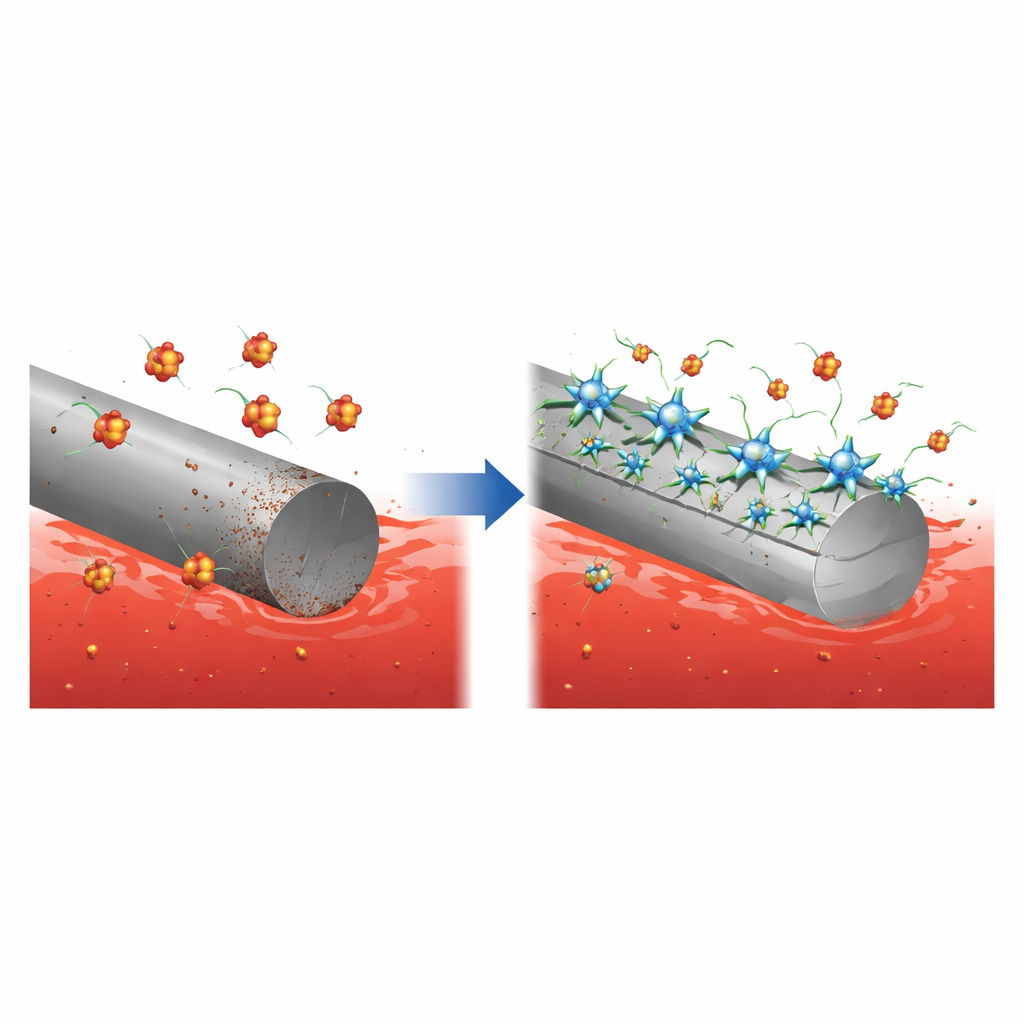
A Big Problem in Industrial Pipes
Corrosion costs industry billions each year and can account for several percent of a nation’s economic output when failures, maintenance, and downtime are added up. One vulnerable material is X‑65 carbon steel, widely used in oil and gas pipelines because it is strong and affordable. During routine cleaning, these pipelines are flushed with hydrochloric acid to strip away scale and deposits. While effective, this acid also gnaws at the steel itself, thinning pipe walls and creating rough, weakened surfaces. To tame this trade‑off, companies inject small amounts of special chemicals called corrosion inhibitors, which are designed to coat steel and block contact with the aggressive liquid.
A Star-Shaped Molecular Shield
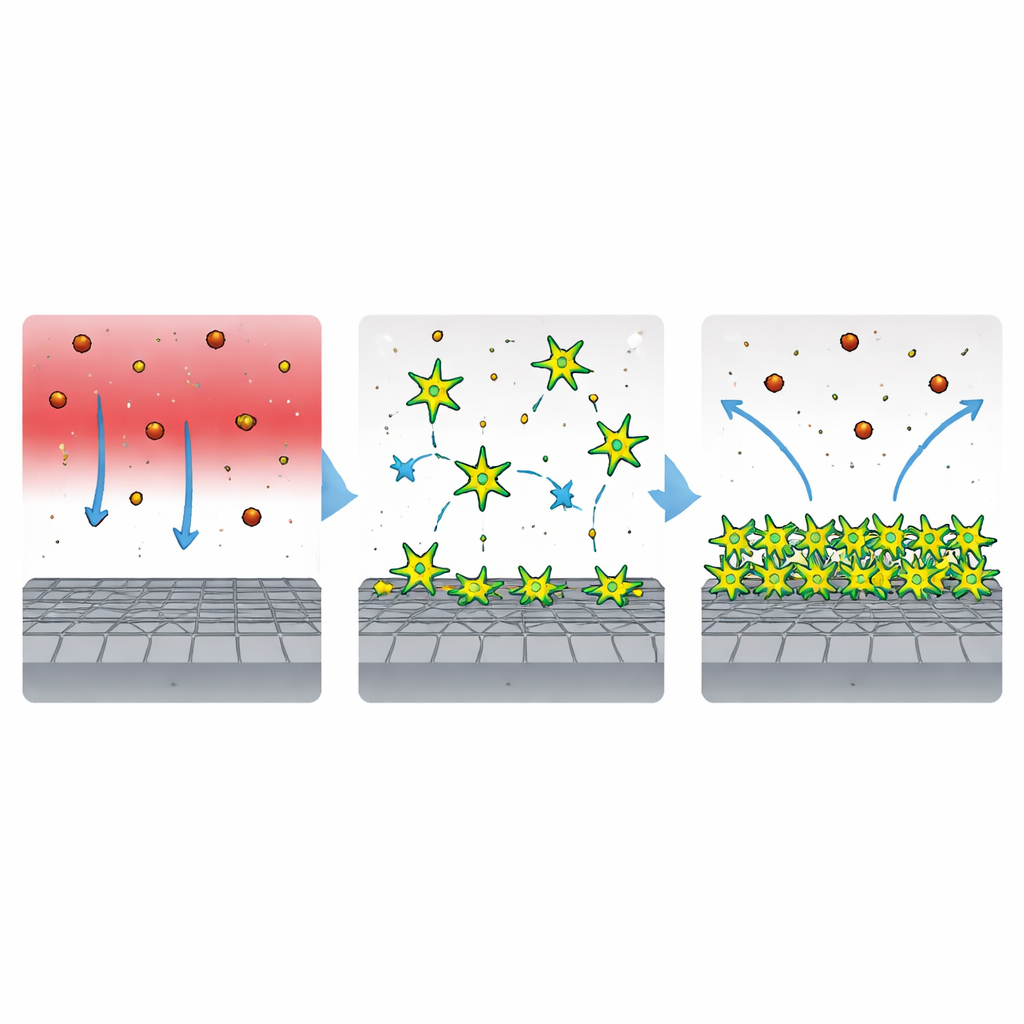
The researchers designed a new inhibitor called PAS, a polyamine surfactant with a distinctive star‑like architecture. Each molecule has a central hub connected to several flexible arms. The tips of these arms carry nitrogen- and oxygen-rich groups that are strongly attracted to steel, while long hydrocarbon tails dislike water and prefer to cluster together. This combination encourages PAS to leave the bulk solution and spread out over solid surfaces. Laboratory measurements of surface tension confirmed that even modest amounts of PAS strongly prefer interfaces, a sign that the molecules are well suited to forming tight, protective films on metal in water‑based environments.
Putting the Coating to the Test
To see whether this molecular design actually protects steel, the team immersed X‑65 samples in acidic solution with and without PAS. They tracked how quickly metal dissolved by weighing the samples over time and by using electrochemical techniques that sense how easily charges move during corrosion. Across a wide range of conditions—different temperatures, immersion times, and inhibitor doses—the presence of PAS cut metal loss dramatically. At an optimal concentration, the protective efficiency reached about 96 percent, and the resistance to charge transfer at the steel surface increased more than twentyfold. Other measurements showed that PAS slows both sides of the corrosion reaction, acting on the pathways where iron atoms dissolve and where hydrogen gas is released.
Seeing the Invisible Film
Microscopic imaging gave a visual before‑and‑after picture of the steel surface. Without PAS, the acid left the metal severely scarred, with pits, cracks, and corrosion products rich in iron and chloride. When PAS was added, the surface appeared far smoother and cleaner, and the amount of corrosive chloride clinging to it dropped sharply. Atomic force microscopy, which scans surfaces in three dimensions, confirmed that the average roughness fell by more than half. Computer simulations and quantum‑level calculations supported this picture: they showed the star‑shaped molecules lying nearly flat on the steel, anchoring themselves through multiple contact points and forming a dense, continuous layer that repels attacking species in the liquid.
What This Means for Everyday Technology
In plain terms, the study shows that carefully tailored, star‑shaped molecules can act like multi‑armed anchors that grab onto steel and spread out to form a tough, waterproof skin. This skin greatly slows the chemical eating-away that would otherwise thin and weaken metal in acidic environments. Because PAS works efficiently at relatively low doses and combines strong attachment sites with water‑repelling tails, it offers a promising route to longer‑lasting pipelines and industrial equipment, potentially cutting costs, reducing leaks, and improving safety wherever steel must endure harsh chemical cleaning.
Citation: Elaraby, A., El-Tabey, A.E., Migahed, M.A. et al. Experimental and theoretical assessments of an innovative star-shaped polyamine surfactant designed for X-65 steel corrosion mitigation in acidic environment. Sci Rep 16, 8499 (2026). https://doi.org/10.1038/s41598-026-38444-4
Keywords: corrosion inhibitor, carbon steel, surfactant coating, acidic environment, pipeline protection