Clear Sky Science · en
Rational design and antibacterial assessment of novel pyranopyrazole derivatives: a combined experimental and in silico study
Why New Germ-Fighting Molecules Matter
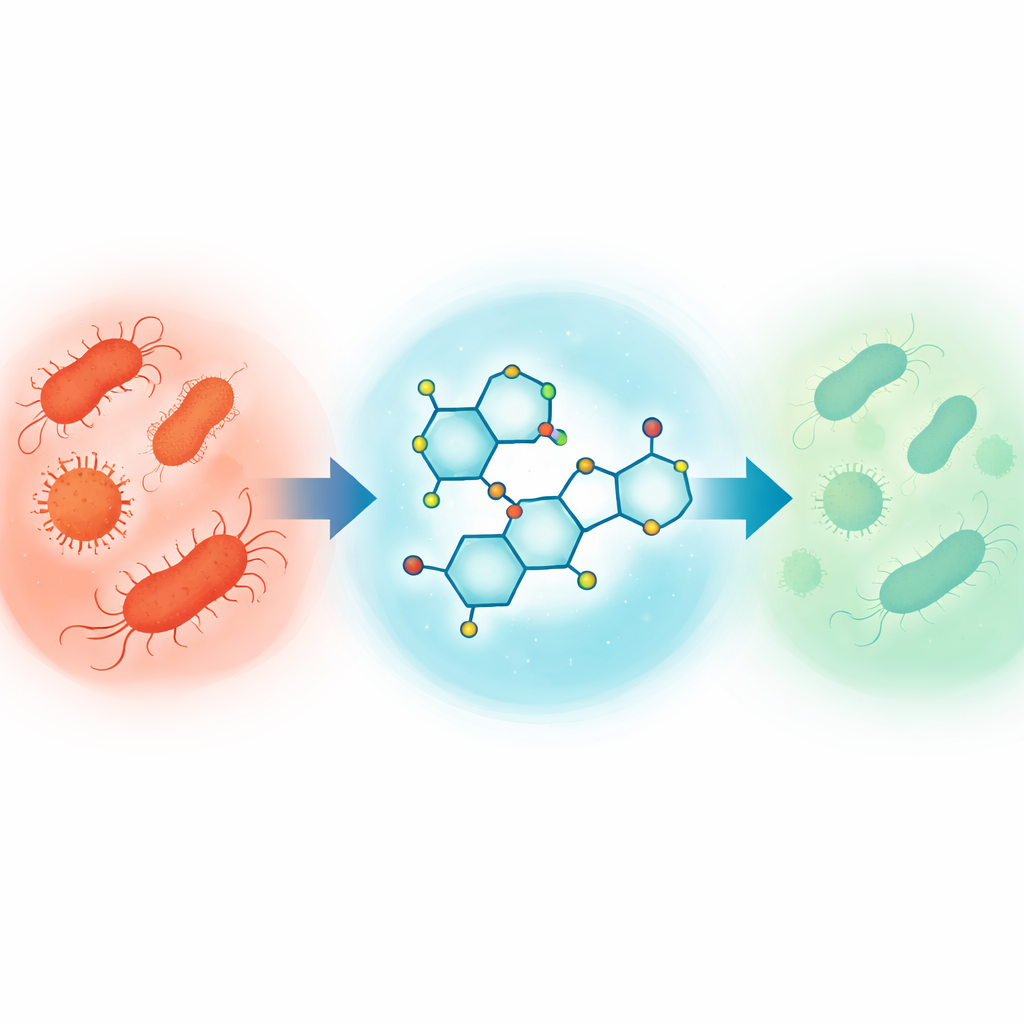
Antibiotic-resistant infections are on the rise worldwide, making once-routine illnesses harder and sometimes impossible to treat. This study explores a fresh class of laboratory-made molecules, called pyranopyrazoles, designed to act as new antibacterial weapons. Using a mix of green chemistry and advanced computer modeling, the researchers asked a pressing question: can we rationally design small molecules that both kill harmful bacteria and behave like real, safe drugs inside the body?
Building New Molecules in a Cleaner Way
The team began by synthesizing a family of pyranopyrazole-based compounds using multicomponent reactions, in which several simple starting materials are combined in a single vessel. This strategy reduces waste and avoids harsh conditions, aligning with greener approaches to chemistry. A central intermediate molecule served as a flexible hub that, when reacted with different small reagents such as organic acids, amines, and nitriles, produced a diverse panel of related structures. Each step in these reaction pathways was confirmed using standard analytical tools that read out how atoms are connected, ensuring that the intended molecular architectures had indeed been built.
Putting the Compounds to the Germ Test
Once in hand, the new molecules were tested in the lab against four medically important bacteria: two Gram-positive species (Staphylococcus aureus and Bacillus subtilis) and two Gram-negative species (Escherichia coli and Pseudomonas aeruginosa). The researchers measured how well each compound prevented bacterial growth by examining the clear “inhibition zones” they produced on agar plates, using the common antibiotic chloramphenicol as a benchmark. Several compounds, including variants labeled 2b, 3a, 5b, 6b, 7b, and 8b, showed strong activity, sometimes approaching or even surpassing the reference drug for particular strains. Notably, some molecules were better suited to Gram-positive bacteria, while others worked best on Gram-negative species, underscoring how small structural tweaks can shift antibacterial strength and selectivity.
Peering Into Electronic Behavior and Enzyme Targeting
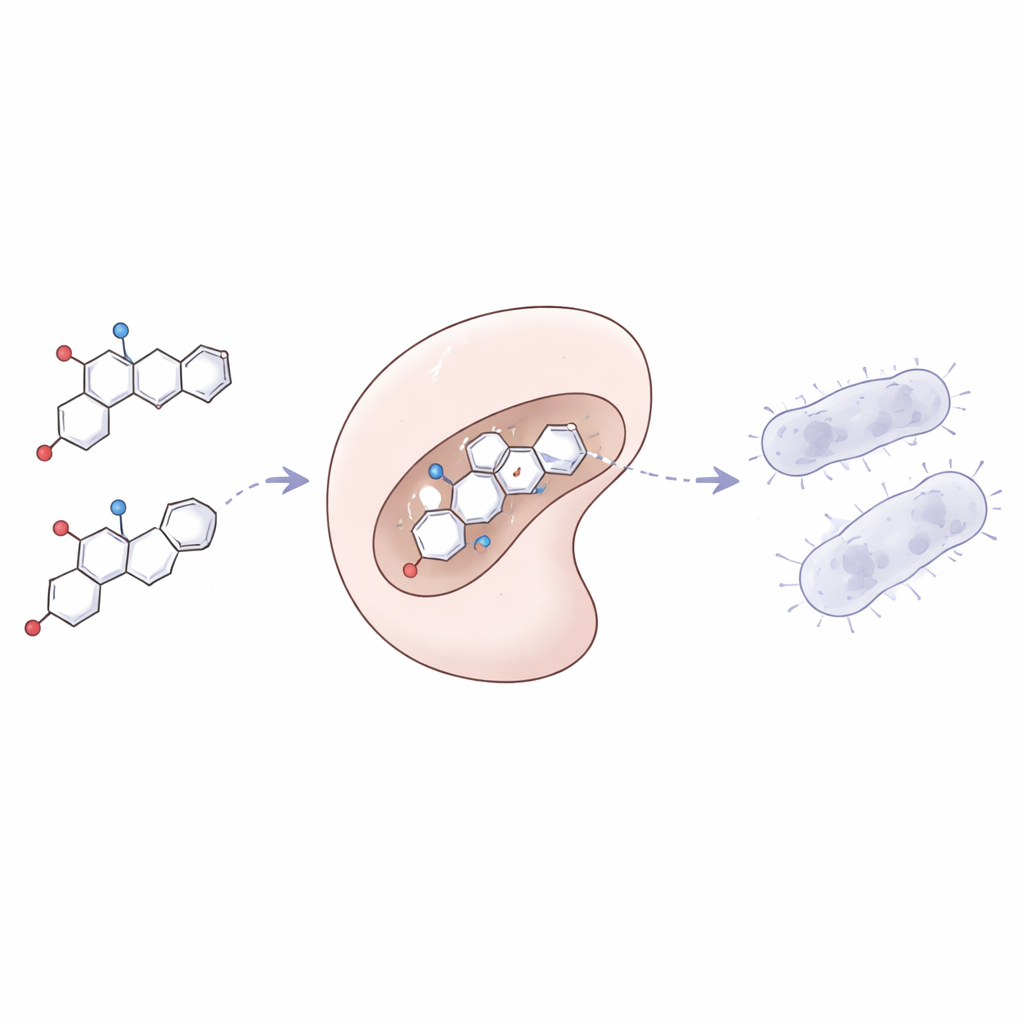
To understand why some pyranopyrazoles outperformed others, the team used quantum chemical calculations to probe how electrons are arranged within the molecules. They focused on two key energy levels that control how easily a molecule can donate or accept electrons, which in turn influence how it reacts with biological targets. Compounds with smaller gaps between these levels tended to be more reactive and correlated with stronger antibacterial effects. Color-coded electrostatic maps further revealed which parts of each molecule were more positively or negatively charged, highlighting the regions most likely to latch onto proteins. The main protein target was tyrosyl-tRNA synthetase, an essential bacterial enzyme involved in building new proteins. Computer docking simulations showed that several pyranopyrazoles fit snugly into this enzyme’s active site, forming multiple stabilizing interactions and achieving very favorable binding energies in the nanomolar range.
From Computer Predictions to Drug-Like Behavior
Designing a good antibiotic is not just about killing bacteria; the molecule must also travel through the body, be absorbed, and avoid excessive toxicity. The researchers therefore ran in silico ADME and toxicity predictions to estimate properties such as absorption in the gut, distribution, and potential side effects. Many of the most promising molecules met widely used “drug-like” criteria, including Lipinski’s Rule of Five, and were predicted to have high intestinal absorption. Overall toxicity risk was modeled as low to moderate, and most compounds were classified as non-mutagenic and non-cytotoxic, although some showed signs of potential liver effects that would need further study. Combined with the docking and electronic data, these results helped identify a subset of pyranopyrazoles with the best balance of potency, selectivity, and predicted safety.
What This Means for Future Antibiotics
In simple terms, this work shows that it is possible to deliberately design and fine-tune new antibacterial molecules by pairing smart synthetic chemistry with powerful computer tools. The pyranopyrazole framework, especially when decorated with aromatic rings, halogen atoms, and cyano groups, can penetrate bacterial defenses, bind tightly to a vital bacterial enzyme, and interfere with protein production. While these compounds are still at an early, preclinical stage, they offer a promising starting point for developing future drugs that could help combat antibiotic-resistant infections. The next steps will involve testing how they behave in living organisms, refining their safety, and exploring their effectiveness against hard-to-treat, multidrug-resistant strains.
Citation: Abdelatty, M.M., Makhlouf, A.A., Moustapha, M.E. et al. Rational design and antibacterial assessment of novel pyranopyrazole derivatives: a combined experimental and in silico study. Sci Rep 16, 11621 (2026). https://doi.org/10.1038/s41598-026-37625-5
Keywords: antibiotic resistance, pyranopyrazole, drug design, molecular docking, antibacterial agents