Clear Sky Science · en
Structural insights into coffee bitter taste perception by TAS2R43 receptor
Why Your Morning Coffee Tastes Bitter
Many people love the rich bitterness of coffee, but that sharp edge in each sip is also your body’s early warning system against toxins. This study uncovers, atom by atom, how a specific taste receptor in your mouth, called TAS2R43, detects bitter chemicals from coffee and from a powerful plant poison. By visualizing this receptor in unprecedented detail, the researchers show how bitterness is turned into an electrical signal to the brain and hint at ways to fine‑tune bitter taste in foods and even tap these receptors for new medicines.
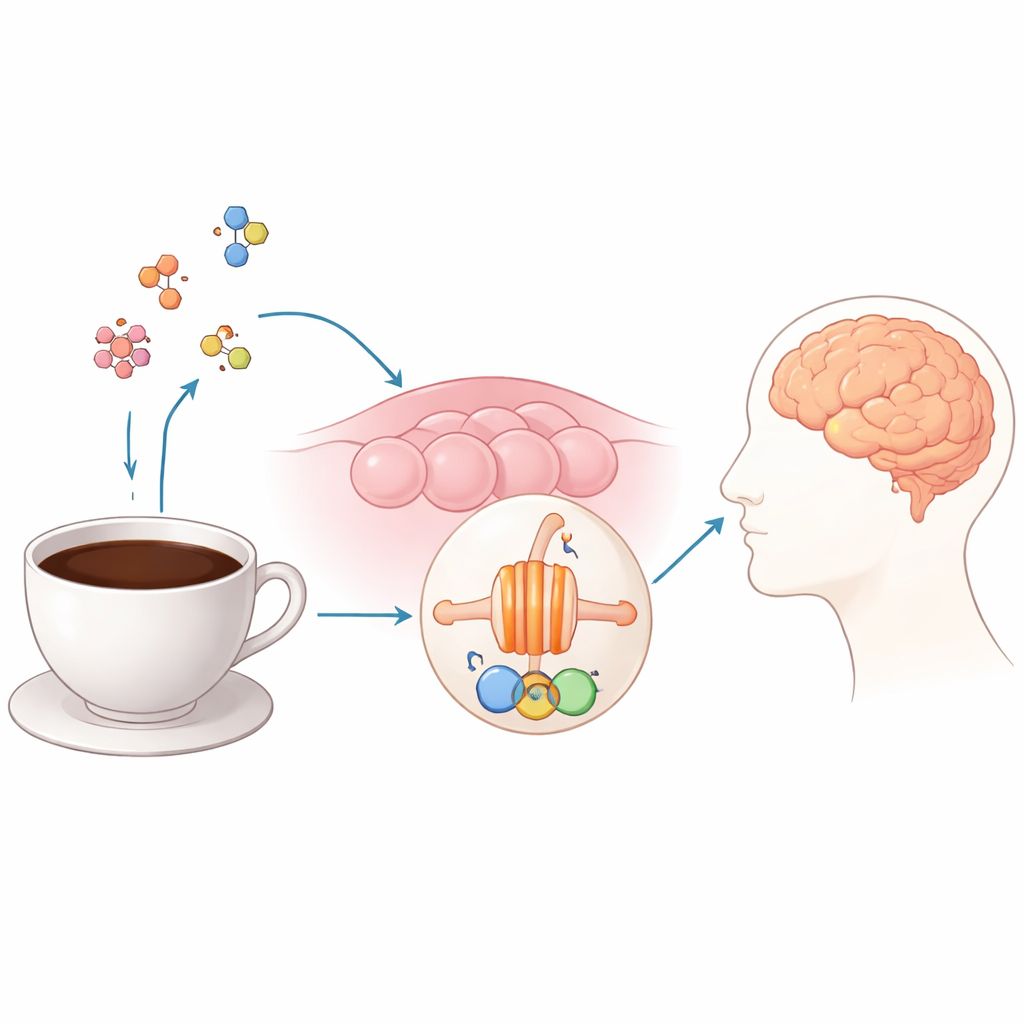
The Body’s Bitter Alarm System
Humans sense five basic tastes: sweet, sour, salty, umami and bitter. While sweet and umami encourage us to eat sugars and proteins, bitter is more like a built‑in hazard sign, helping us avoid spoiled or toxic substances. Bitter taste is detected not by simple pores or channels, but by a family of specialized receptors on taste cells, known as TAS2Rs. One member of this family, TAS2R43, is especially important for recognizing the bitterness of coffee compounds, including caffeine and several lesser‑known molecules formed during roasting. Intriguingly, TAS2R43 is also found outside the tongue—in the gut, airways and other tissues—where its activation has been linked to hormone release, airway relaxation, immune responses and metabolism.
Capturing the Receptor in the Act
To see exactly how TAS2R43 works, the authors used cryo‑electron microscopy, a technique that images flash‑frozen proteins at near‑atomic resolution. They trapped the human TAS2R43 receptor in an active state using aristolochic acid I, a highly bitter plant toxin that binds this receptor extremely tightly, and coupled it to two different cellular switches, known as G proteins: one typical of taste cells (gustducin) and one more common elsewhere in the body (Gi). The resulting structures—sharp enough to trace individual side chains—showed that aristolochic acid nestles into a deep pocket in the outer half of the receptor, a site shared with other bitter receptors yet shaped in a way that gives TAS2R43 its own chemical preferences.
How Bitter Molecules Fit and Flip the Switch
Within this pocket, the toxin is hugged by a ring‑shaped amino acid that stacks against its aromatic rings and by several nearby greasy residues that cradle it from the sides. A positively charged residue reaches in like a fingertip to grip the toxin’s negatively charged groups, while a small patch of water molecules forms a bridge between the toxin and the receptor, stabilizing the fit. When the researchers altered these key residues one by one in human cells, the receptor’s ability to react to the toxin dropped sharply, confirming their importance. They also showed that coffee compounds such as caffeine, cafestol and kahweol can activate TAS2R43, though generally less potently than the toxin; some roasted‑coffee molecules even triggered stronger signaling through the taste‑linked G protein than aristolochic acid itself.
From Binding Pocket to Brain Signal
The structures further reveal how TAS2R43 passes the message inward. On the inner face of the receptor, a cluster of helices forms a docking cradle for the G protein. When the bitter molecule binds outside, this inner surface rearranges, allowing the tip of the G protein to slot deeply into the receptor. Specific charged residues in TAS2R43 clasp matching acidic spots on the G protein, while a tiny water molecule helps glue the two together. Mutating a handful of these contact points disrupted signaling without affecting receptor levels at the cell surface—clear evidence that these are the mechanical joints that turn a chemical encounter on the tongue into a downstream signal that the brain interprets as bitterness.
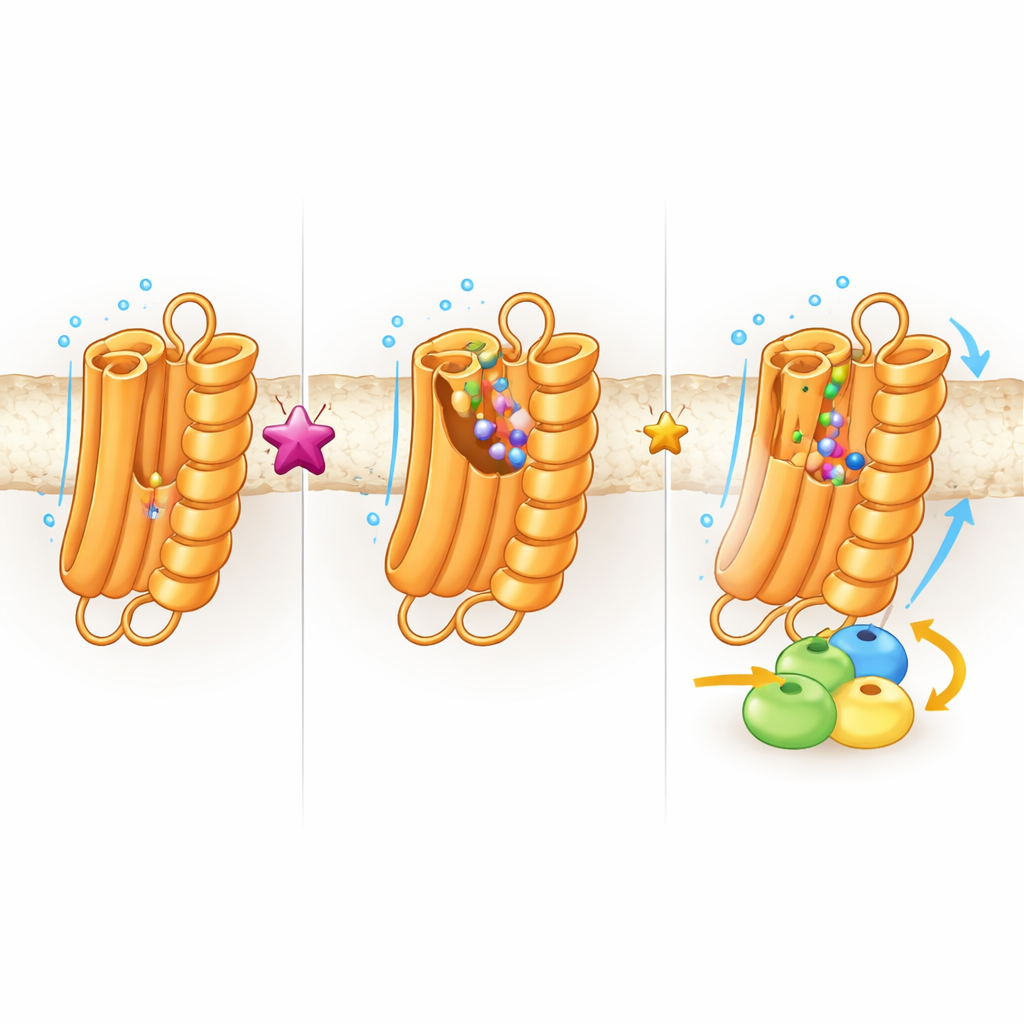
Hidden Pockets and Future Possibilities
To explore how flexible TAS2R43 is, the team ran long computer simulations of the receptor wiggling in a virtual membrane, with and without the toxin bound. These movies revealed that when no ligand is present, parts of the receptor open up, creating temporary inner cavities and enlarging the main pocket; binding of the toxin locks the structure into a more closed, stable state. Such shape‑shifting “cryptic pockets” may allow TAS2R43 to recognize a wide variety of bitter chemicals—or provide new footholds for drugs that dial its activity up or down. The receptor also showed signs of being somewhat active even without any ligand, which could explain why it so readily teams up with G proteins in cells.
What This Means for Taste and Health
In plain terms, this work explains how one of your tongue’s bitter detectors grabs onto both coffee components and a dangerous plant toxin, and how that event pulls an internal lever that ultimately results in the sensation of bitterness. By mapping the exact shape of TAS2R43’s main pocket and its hidden cavities, the study lays a blueprint for designing molecules that soften or block bitterness in foods and drinks or, conversely, selectively activate these receptors in the gut or lungs for therapeutic benefit. Your daily cup of coffee, it turns out, is also a window into a finely tuned molecular alarm system that guards your body while shaping your sense of flavor.
Citation: Kim, Y., Gumpper, R.H., Zhuang, Y. et al. Structural insights into coffee bitter taste perception by TAS2R43 receptor. Nat Struct Mol Biol 33, 701–710 (2026). https://doi.org/10.1038/s41594-026-01776-w
Keywords: bitter taste, coffee, taste receptors, G protein signaling, cryo electron microscopy