Clear Sky Science · en
Structural modifications in strain-engineered bilayer nickelate thin films
Why changing a crystal’s shape matters
Superconductors can carry electric current without wasting energy as heat, but most only work at very low temperatures. A new family of nickel-based materials has shown surprisingly high superconducting temperatures, raising hopes for more efficient power grids, magnets and electronics. In this study, researchers zoom in on a particular nickel compound, La3Ni2O7, grown as ultra-thin films, to see exactly how squeezing or stretching the crystal changes its atomic structure and, in turn, its ability to become superconducting.
Making thin films feel squeezed or stretched
The team grew very thin layers of La3Ni2O7 on different underlying crystals, or substrates, which naturally force the film to stretch or compress in the plane of the layer. Some substrates pull the film slightly apart, creating tensile strain, while others push it together, creating compressive strain. By carefully choosing these substrates, the researchers produced a series of films ranging from strongly compressed to strongly stretched. They then measured how easily each film carried electric current as the temperature dropped. Only the most strongly compressed films showed signs of superconductivity, while moderately compressed films remained metallic and stretched films turned insulating.

Watching atoms shift by trillionths of a meter
To understand why compression is so important, the researchers used advanced electron microscopy techniques that can pinpoint atom positions with precision of just a few trillionths of a meter. A key method, called multislice electron ptychography, allowed them to see not only the heavy lanthanum and nickel atoms, but also the much lighter oxygen atoms that sit around nickel in tiny octahedral cages. By mapping how the nickel–oxygen bonds bend and stretch across the series of strained films, they found that compressive strain makes these cages more symmetric in the plane of the film, while tensile strain leads to a more uneven pattern of bond angles.
Symmetry in the plane, freedom out of the plane
The measurements revealed an important contrast between the film geometry and that of bulk crystals under high pressure, where superconductivity was first discovered in this material. In both cases, the in-plane distances between atoms shrink in a similar way as superconductivity appears. However, in the thin films the spacing between layers along the vertical direction actually grows under compression, whereas in bulk crystals it shrinks under pressure. Detailed analysis of nickel–oxygen bond lengths showed that the out-of-plane bonds become longer in the superconducting film, even though the overall unit cell volume still decreases. This result suggests that squeezing the planes within the layer is more crucial than pushing the layers closer together, challenging earlier assumptions that vertical compression was the main driver.
Disentangling which distortions matter most
To go beyond visual inspection, the researchers built a theoretical framework that breaks down the complicated distortions of the oxygen cages into simpler building blocks: changes in bond length, changes in internal angles and rigid rotations of the octahedra. Using supercomputer calculations, they asked how each type of distortion affects the electronic bands where the charge carriers live. They found that bond length changes mainly shift the energy levels up or down, while rotations play a special role in “cleaning up” the low-energy bands by reducing unwanted mixing of certain nickel orbitals. In both superconducting films and pressurized bulk crystals, higher octahedral symmetry and specific rotation patterns were linked to a cleaner electronic landscape that is thought to favor superconductivity.
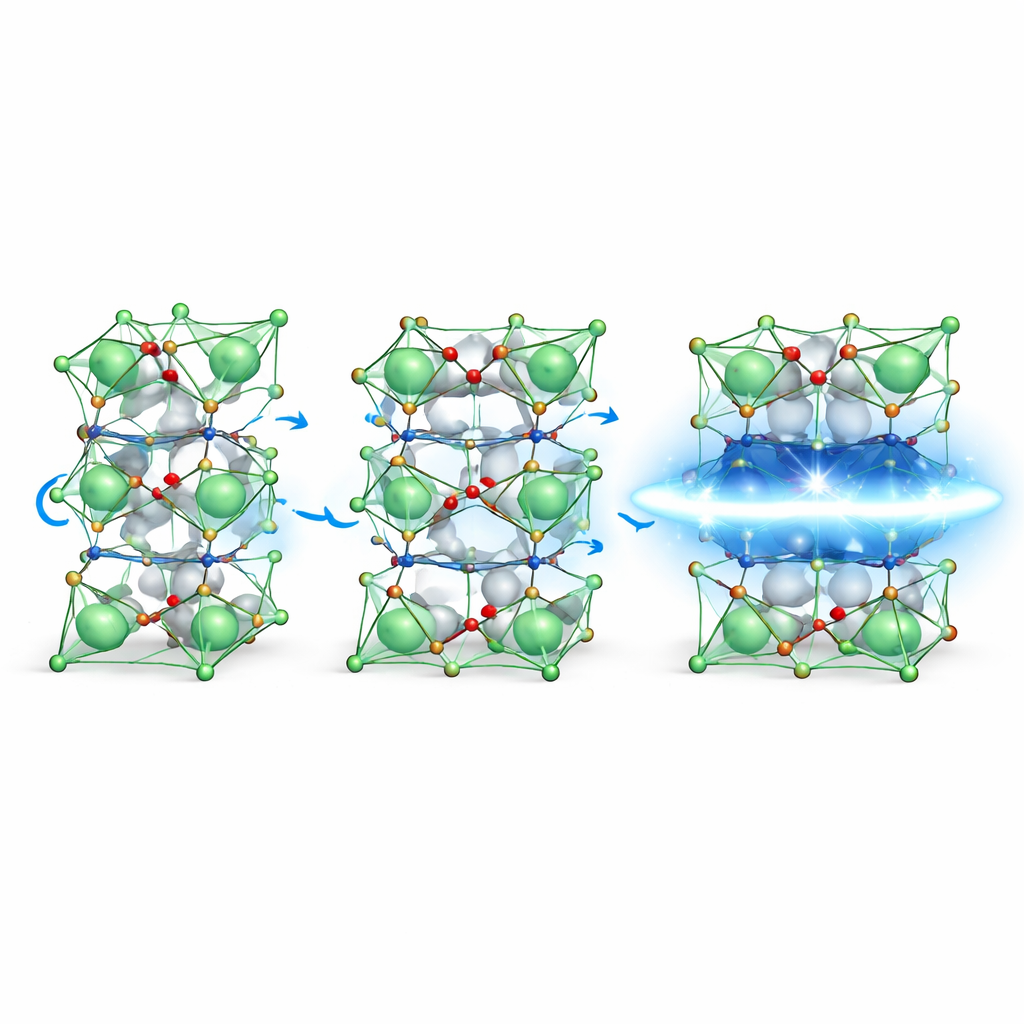
What this means for future superconductors
Taken together, the work shows that not all pressure is equal: carefully engineered in-plane compression and symmetry in the nickel–oxygen network appear to be the shared ingredients that support superconductivity in La3Ni2O7, whether in bulk crystals or in thin films. By directly tying picometer-scale structural tweaks to changes in electronic behavior, the study offers a roadmap for designing and tuning next-generation superconductors, not only in nickelates but across many oxide materials where tiny shifts in atomic arrangement can have outsized effects on how electricity flows.
Citation: Bhatt, L., Abarca Morales, E., Jiang, A.Y. et al. Structural modifications in strain-engineered bilayer nickelate thin films. Nature 653, 76–82 (2026). https://doi.org/10.1038/s41586-026-10446-2
Keywords: nickelate superconductors, strain engineering, thin films, electron microscopy, crystal structure