Clear Sky Science · en
Electronic origin of reorganization energy in interfacial electron transfer
Why the Way Surfaces Move Electrons Matters
From the batteries in phones to the proteins that power our cells, many technologies and life processes rely on moving electrons across the boundary between a solid surface and a liquid. For decades, scientists have assumed that the liquid side of this boundary does almost all the work in controlling how fast these electrons hop, while the solid side simply supplies electrons when needed. This paper turns that assumption on its head by showing that the electronic makeup of the solid surface itself can strongly control the cost of moving an electron—and therefore how quickly energy and chemistry flow.
Revisiting a Classic Picture of Electron Flow
Traditional theories of electron transfer describe the process like rolling a ball over a hill. The height of that hill, called the activation barrier, depends largely on how much the atoms and surrounding liquid must shift to accommodate the new charge—an energy cost known as reorganization energy. In the standard view, this cost comes almost entirely from the liquid and the dissolved molecule, while the solid electrode acts mainly as a reservoir of electrons whose available states set how many pathways the electron can take. The authors ask a simple but far-reaching question: what if the electrode’s own electrons, and how easily they rearrange, also change the height of that hill?
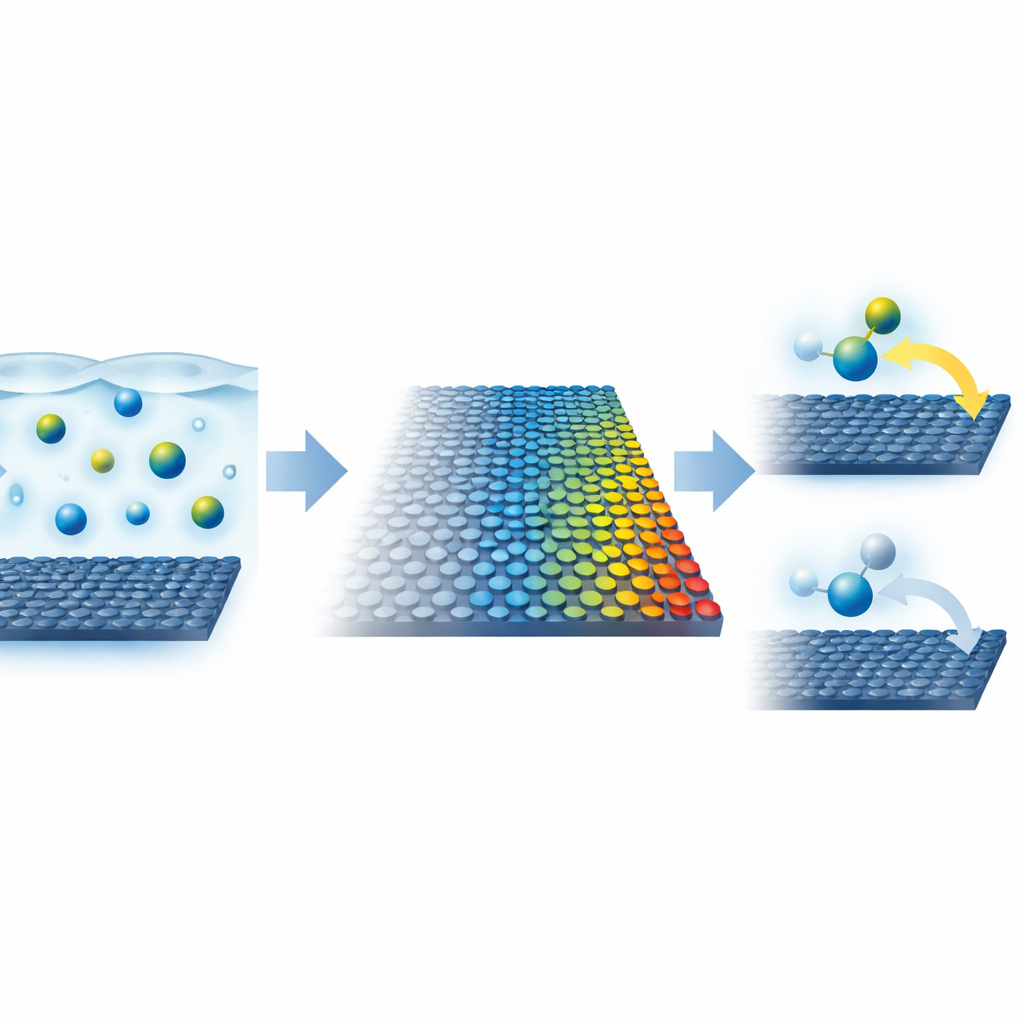
Building Designer Stacks of Atom-Thin Materials
To probe this idea, the team built carefully layered structures from single-atom-thick crystals. A sheet of graphene served as the electrode whose electronic richness could be tuned. On the other side of an ultrathin insulating layer made of hexagonal boron nitride, they placed crystals that either pull electrons from graphene or donate electrons to it. By changing the thickness of this spacer, they could finely control how many extra charge carriers ended up in the graphene, and thus how metallic it behaved. They then used a nanopipette filled with a well‑studied redox molecule to create tiny, well-defined liquid cells on the graphene surface and measured how quickly electrons hopped between the molecule in solution and the solid sheet.
Watching Electron Speeds Change with Electronic Richness
As the electronic richness, or density of states, in graphene increased, the electron transfer rate rose dramatically—far more than classic theory would predict from simply having more available electronic pathways. Even when the dopant crystal was separated from graphene by tens of nanometres of boron nitride, the surface still showed faster electron exchange than undoped graphene. Independent measurements using Raman spectroscopy and Hall transport confirmed how the carrier concentration in graphene changed with spacer thickness, and fluorescence studies suggested that defects in the boron nitride helped mediate extra charge transfer at very small separations. Altogether, these experiments established a clear, quantitative link between the electrode’s electronic character and the speed of interfacial electron flow.
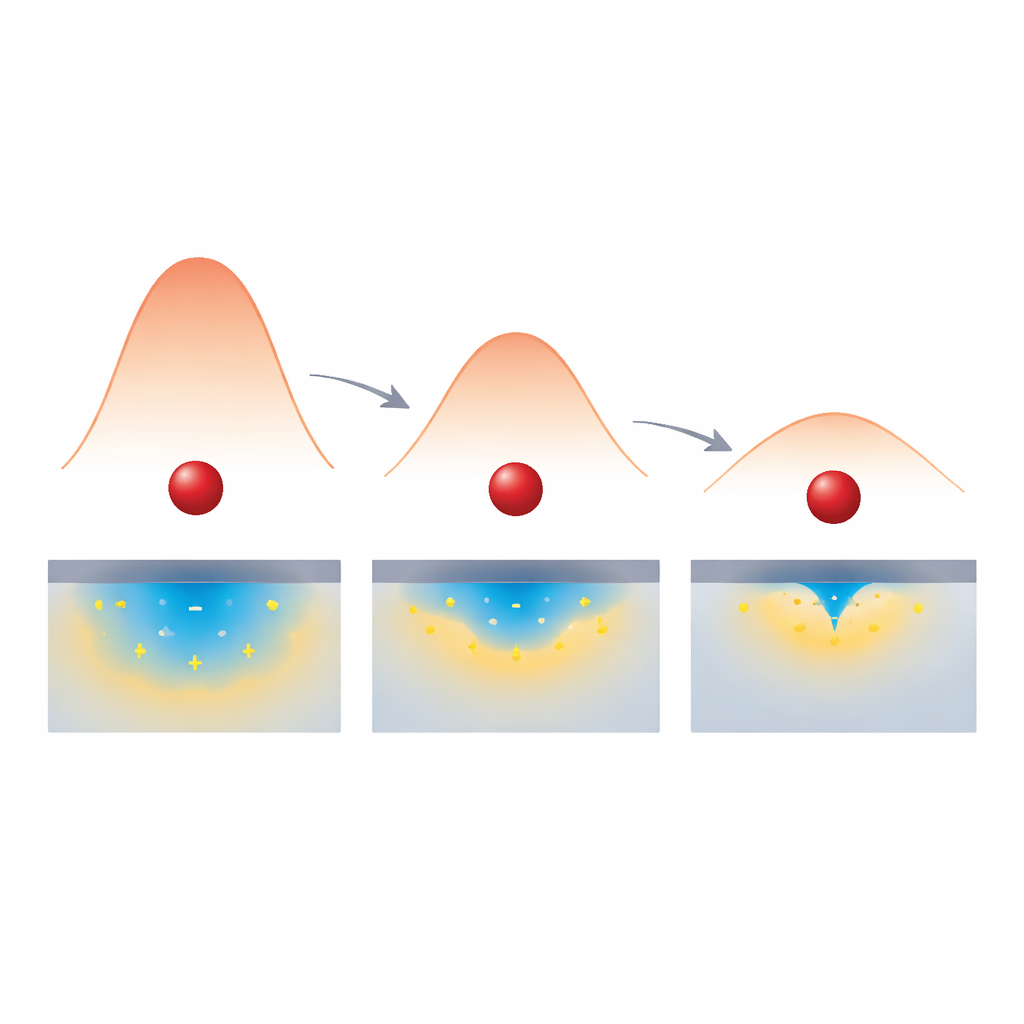
How Charge Screening Lowers the Energy Cost
To understand why the effect was so strong, the authors turned to theory and computer simulations. They modelled how the electrode’s electrons rearrange in response to a charged molecule hovering just above the surface. In a poorly metallic electrode with few carriers, the induced charge spreads out over a broad region, offering only weak stabilization of the transition state for electron transfer. As the density of carriers grows and the material becomes more metallic, the induced charge sharpens and gathers directly under the molecule, like a focused mirror charge. This stronger, more localized screening lowers the reorganization energy: the environment needs to shift less to move an electron, so the energy hill becomes lower. When they built this density‑dependent reorganization energy into an extended Marcus‑type model, the calculations matched the experimental rate changes across the full range of doping, whereas models that kept the reorganization energy fixed failed badly.
What This Means for Future Energy and Quantum Devices
The study shows that the electronic structure of the solid electrode does far more than simply supply electrons—it reshapes the very energy landscape that electrons must cross. In systems with low electronic richness, such as many semiconductors and atomically thin materials, the extra reorganization cost from the electrode itself can be as large as the familiar solvent contribution. By explicitly folding the electrode’s electronic properties into the design of interfaces, researchers can better predict and control electron transfer in batteries, solar cells, catalysts and quantum devices. In short, tuning how well an electrode screens charge offers a powerful new lever for speeding up or slowing down key reactions at surfaces.
Citation: Maroo, S., Coello Escalante, L., Wang, Y. et al. Electronic origin of reorganization energy in interfacial electron transfer. Nature 653, 98–103 (2026). https://doi.org/10.1038/s41586-026-10311-2
Keywords: electron transfer, graphene electrodes, reorganization energy, electrochemical interfaces, density of states