Clear Sky Science · en
Adventitious carbon breaks symmetry in oxide contact electrification
Why tiny bits of grime on rocks matter
Anyone who has shuffled across a carpet and felt a zap, or seen lightning flash in a volcanic ash cloud, has met the strange world of static electricity. This study tackles a long-standing puzzle in that world: why do two pieces of the very same rock material charge each other when they touch and separate? The answer turns out to hinge on a surprisingly humble culprit—ultra-thin, naturally occurring films of carbon-based grime that coat almost every surface we encounter.
A quiet force shaping dust, storms and worlds
Silicon dioxide and related oxides make up much of Earth’s crust and the surfaces of the Moon, Mars and many asteroids. When grains of these materials collide in desert dust storms, volcanic plumes or swirling disks of rock around young stars, they exchange electric charge. That charging can help sand grains stay aloft for long distances, spark volcanic lightning and even help tiny “pebbles” stick together as planets begin to form. Yet for decades, scientists have struggled to explain why two pieces of the same insulating material—say, two bits of glass—do not remain electrically neutral after repeated contact.
Levitation, bouncing spheres and controlled grime
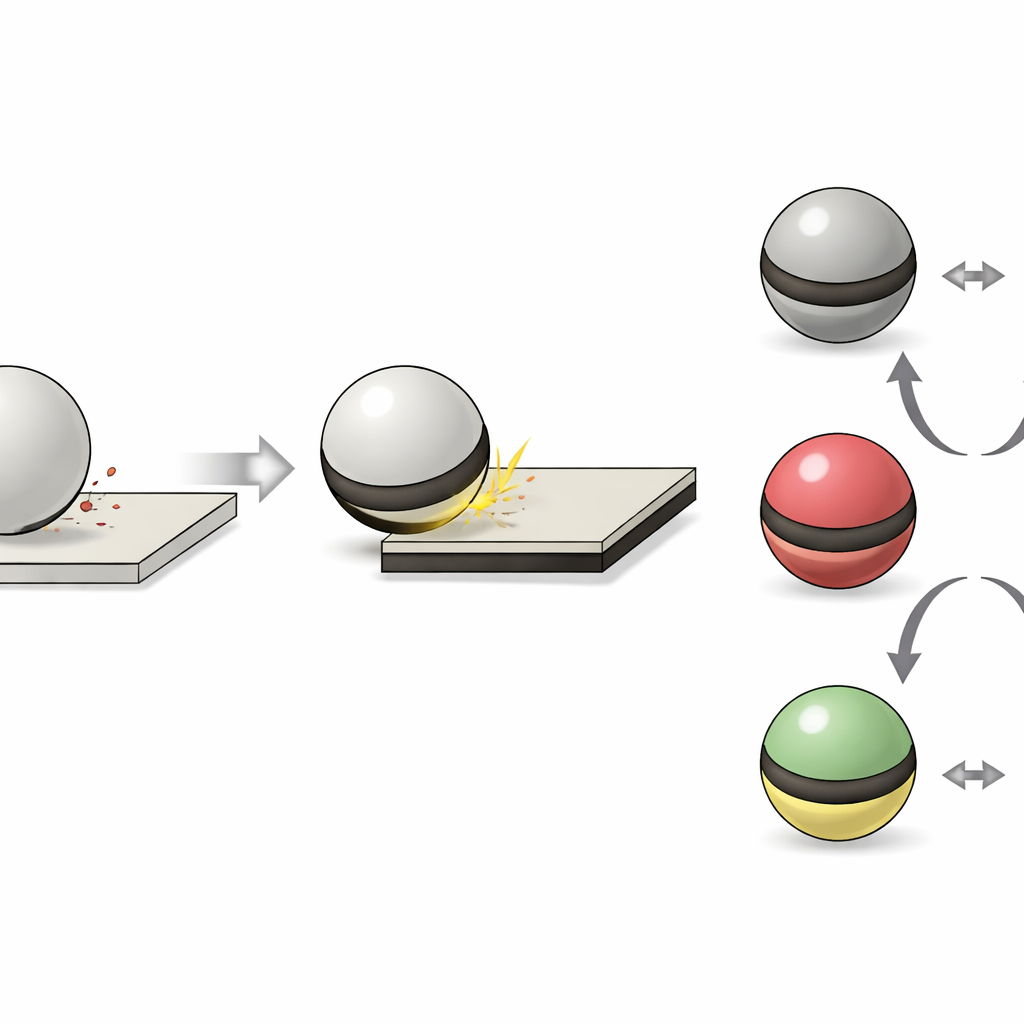
To probe this mystery, the researchers designed an experiment where a tiny sphere of pure fused silica is suspended in mid-air by sound waves above a matching silica plate. By briefly turning off the acoustic trap, they let the sphere drop, bounce off the plate and then recapture it, one collision at a time. A carefully tuned electric field causes the charged sphere to wobble; from its motion, the team can measure how much charge it has gained or lost after each bounce. Initially, different sphere–plate pairs showed consistent charging in one direction or the other, but across many pairs the “winner” was random—as if each supposedly identical piece of silica were its own distinct material.
Stripping surfaces to reveal the hidden player
The team then asked whether molecules that naturally settle onto surfaces from air could be tilting the balance. Instead of adding custom coatings, they removed whatever was already there by gently baking samples or exposing them to a low-power plasma—standard steps in high-tech cleaning. This simple change flipped how the objects charged: a sphere that once became positive could be driven negative, and treating the plate could boost the sphere’s positive charge. Even mild heating nudged the effect, and repeating the treatment made it stronger. These results clashed with the common view that absorbed water alone explains such behavior, because the treated, more “water-friendly” surfaces did not charge the way water-based ideas would predict.
Carbon films that come and go, and take charge with them
To see what was really on the surfaces, the researchers used several surface-sensitive techniques. Time-of-flight mass spectrometry revealed a rich mix of organic fragments—small carbon-and-hydrogen pieces—spread across silica that had only been cleaned and left in normal air. After baking or plasma treatment, these carbon signals dropped sharply. Other measurements that probe just the top atomic layer showed that, once cleaned, carbon slowly crept back over many hours. Strikingly, the rate at which charge behavior drifted back towards its original state matched the rate at which carbon returned. Infrared spectroscopy, which tracks vibrations of carbon–hydrogen bonds, confirmed the same hours-long regrowth of a carbon-rich layer. Together, the parallel time scales for carbon “recoating” and changing electrical behavior point squarely to these adventitious carbon films as the key symmetry breaker.
From one material to many: when carbon outruns the rock
Finally, the team asked whether this hidden carbon layer matters only when identical materials touch, or also when different oxides meet. They tested pairs of silica, alumina, spinel and zirconia with varied roughness and crystal structure. After standard cleaning, these materials lined up into a tidy “triboelectric series”: one end tended to become positive, the other negative, in a consistent order. But when they selectively baked the member of each pair that had been charging positive, the direction of charge transfer reversed in every case—effectively turning the series upside down. Similar flips appeared with other oxide and glass combinations. This shows that the underlying material still influences charging, but if one surface is largely stripped of carbon while the other is not, the carbon imbalance can overwhelm those intrinsic differences.
What this means for dust, devices and future work
For a non-specialist, the message is that the tiniest, most easily overlooked layer on a rock or glass surface can dominate how it behaves electrically. The study makes a strong case that natural, carbon-based films—picked up from air and constantly drifting in and out of balance—are what break the supposed symmetry between “identical” oxide surfaces and help decide which way charge flows. In nature, where conditions are far from pristine, this subtle grime likely shapes how dust moves, how lightning is born in ash clouds and how particles clump in space. For engineers and scientists, it means that any theory of contact charging in oxides must reckon with these trace carbon coatings, and that controlling or even just tracking them could be crucial in technologies that depend on—or seek to avoid—static electricity.
Citation: Grosjean, G., Ostermann, M., Sauer, M. et al. Adventitious carbon breaks symmetry in oxide contact electrification. Nature 651, 626–631 (2026). https://doi.org/10.1038/s41586-025-10088-w
Keywords: triboelectric charging, oxide surfaces, surface contamination, adventitious carbon, static electricity