Clear Sky Science · en
Vagal blood volume receptors compensate for haemorrhage and posture change
Why staying upright does not make you faint
Every time you stand up, gravity suddenly pulls blood toward your legs. Yet most of the time you do not black out. This paper uncovers a hidden safety system of nerve cells that constantly feel how full the heart is with blood and tell the brain to adjust blood pressure on the fly. Understanding this system helps explain why some people get dizzy when they stand and why severe blood loss can quickly become life‑threatening.
Hidden sensors that feel blood volume
The heart is laced with sensory nerves that send a running commentary to the brain about what is happening inside the chest. For decades, scientists have understood one major group, the baroreceptors, which sense blood pressure in large arteries. But another group of sensors that react to how much blood is actually filling the heart had remained mysterious. The authors focused on nerve cells in the vagus nerve that carry a mechanical sensor protein called PIEZO2. Because PIEZO2 opens when cells are stretched, these neurons were prime candidates to act as volume meters for the beating heart.
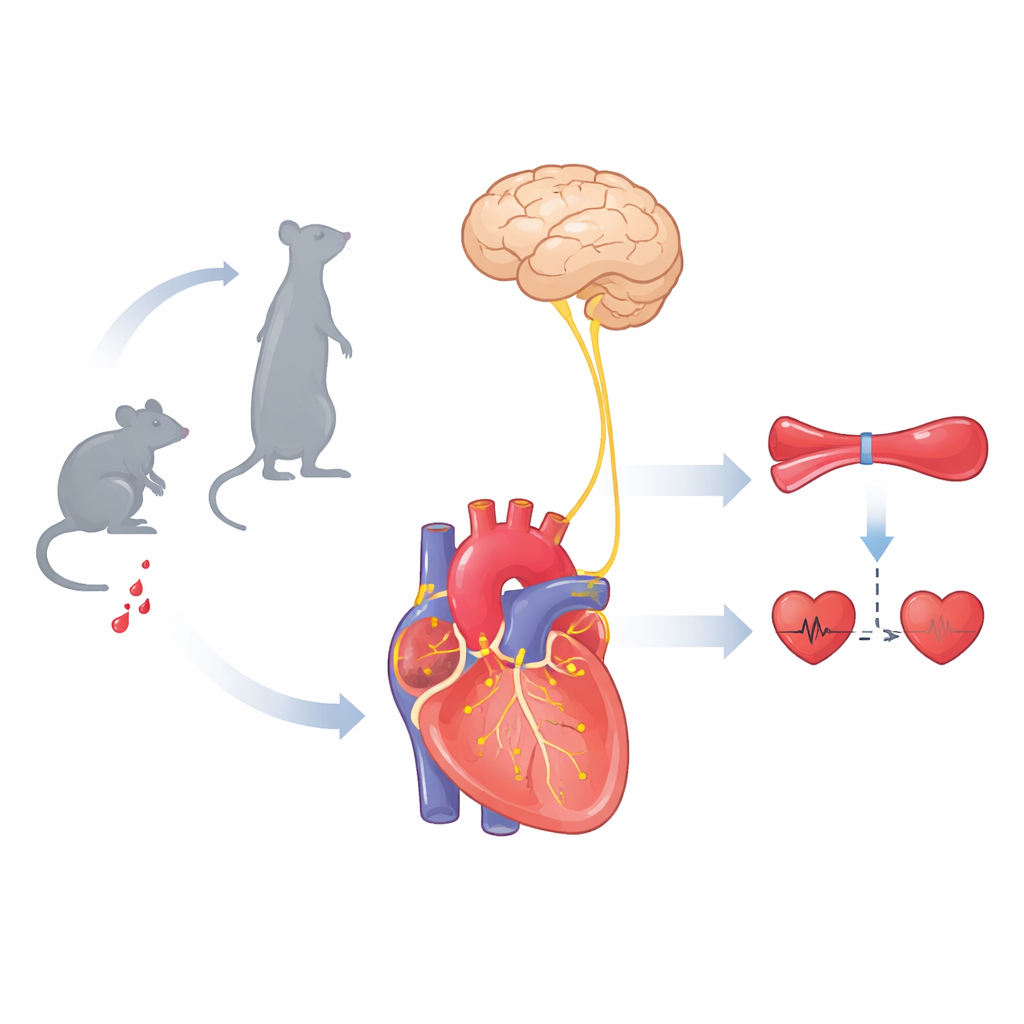
A mouse version of the tilt‑table test
To see how different sensors help the body cope with posture change, the team adapted the clinical tilt‑table test for mice. Anaesthetized animals were rotated from lying down to upright while their blood pressure and heart rate were monitored. Normal mice showed a sharp drop in blood pressure as they were tilted up, followed within seconds by a recovery and a lasting increase in heart rate, preserving blood flow to the brain. Cutting key neck nerves, including the vagus, wiped out this compensation, leading to prolonged low pressure. Strikingly, mice genetically engineered to lack PIEZO2 only in specific vagal sensory neurons also failed to recover properly, even though their classic baroreceptor reflex was intact. This showed that a second, PIEZO2‑dependent pathway is essential for stabilizing blood pressure when gravity suddenly shifts blood downward.
Mapping the special nerve endings in the heart
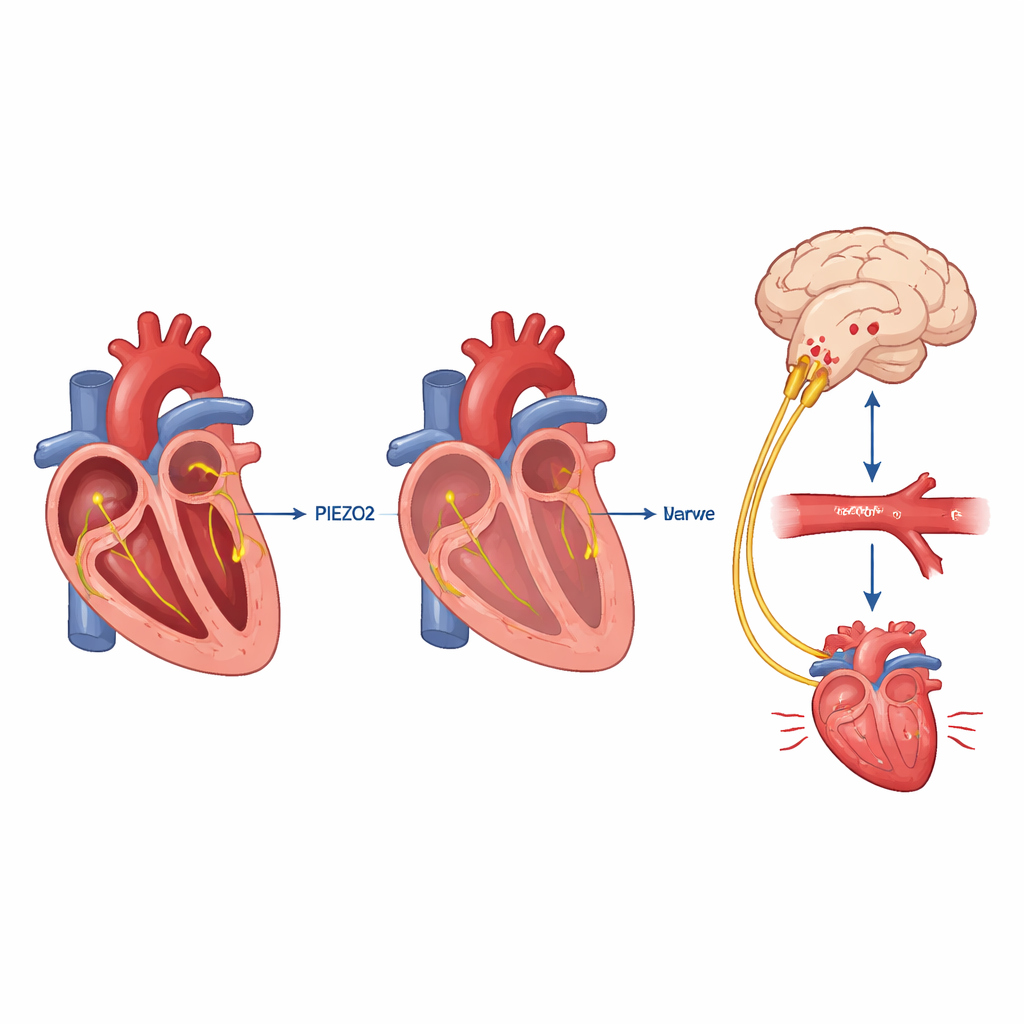
The researchers then traced where PIEZO2‑bearing vagal neurons go. By using viruses that turn these cells fluorescent, they made three‑dimensional images of nerve endings across the mouse heart. They found that PIEZO2 neurons formed distinctive web‑like "end‑net" endings on the atria and ventricles, clustered particularly where blood from the large veins enters the heart. Another major vagal group, marked by a receptor called NPY2R, formed both end‑nets and more bushy "flower spray" endings. When the team selectively killed PIEZO2 neurons in the vagus, mice again lost their ability to correct the blood pressure drop during tilting, but eliminating the NPY2R group did not. This pinpointed PIEZO2 end‑nets as the crucial sensors for posture‑related control, distinguishing them from other heart‑feeling nerves.
Listening to the nerve traffic with each heartbeat
Next, the authors directly recorded electrical activity from the vagus nerve just above the heart while also tracking the electrocardiogram and pressures inside the heart chambers. In healthy mice they saw bursts of nerve spikes locked to two precise moments in every heartbeat: during contraction of the atria and during contraction of the ventricles. When blood volume was slowly withdrawn, these heartbeat‑linked signals weakened; when salty fluid was infused to expand volume, the signals grew stronger. Small losses of blood affected the atrial‑timed signals the most, hinting that these sensors are tuned to early signs of central volume loss. In mice lacking PIEZO2 or whose PIEZO2 neurons had been ablated, the heartbeat‑coupled activity almost vanished and no longer changed with volume, proving that PIEZO2 is the key stretch sensor for these receptors.
A safety net during severe bleeding
The heart volume sensors do not only act during posture changes. In a controlled tail‑bleeding model that mimics traumatic blood loss, normal mice initially kept their blood pressure up and raised their heart rate, despite losing about a quarter of their blood within half an hour. Mice missing PIEZO2 in vagal sensory neurons bled at the same rate but could not mount this compensatory response: their blood pressure sagged, heart rate did not rise enough, and they died significantly sooner. Artificially activating PIEZO2 vagal fibers near the heart with light caused a drop in blood pressure when volume was normal, indicating that the brain can push circulation up or down depending on whether these receptors are loaded or unloaded.
What this means for health and disease
Together, the findings reveal a dedicated set of heart‑based volume receptors that fire with every beat, sense how much blood is returning to the heart, and help the brain keep circulation steady during standing and bleeding. Unlike baroreceptors, which mainly report pressure in the arteries, these PIEZO2‑positive vagal neurons report how full the central blood reservoir is. When they work, we can stand up, lose some blood, or face other challenges without immediately fainting. When they fail, the result is orthostatic hypotension and poor survival after major haemorrhage. Understanding this volume‑sensing pathway could ultimately lead to better diagnosis and treatment for people prone to dizziness on standing, or for stabilizing patients after trauma and surgery.
Citation: Liu, Z., Lu, S., Haskell, I.A. et al. Vagal blood volume receptors compensate for haemorrhage and posture change. Nature 651, 1068–1076 (2026). https://doi.org/10.1038/s41586-025-10010-4
Keywords: vagus nerve, blood volume, orthostatic hypotension, PIEZO2, cardiovascular reflexes