Clear Sky Science · en
A comprehensive model for the electric double layer of stepped platinum electrodes
Why tiny features on metal matter
Platinum is a workhorse metal in fuel cells and many other clean energy devices, where it helps chemical reactions turn electricity into useful products and back again. At the heart of these processes lies a slim region where the metal touches salty water, known as the electric double layer. In real devices platinum surfaces are not perfectly flat, but full of atomic steps and defects. This study asks a simple but important question: how do these tiny surface features change the way charge is stored at the metal–water interface, and what does that mean for how we understand and improve electrocatalysts?
Looking closely at stepped platinum
To isolate the role of steps, the researchers used carefully prepared single crystals of platinum whose surfaces consist of flat terraces interrupted by regular atomic steps. By changing how often these steps appear, they could tune the surface from nearly flat to strongly stepped, and they examined two common step types with different atomic patterns. All measurements were carried out in very dilute acid, a condition where earlier work had shown that the double layer behaves in a relatively simple way on flat platinum. This provided a clean baseline for comparing the electrical response of stepped and unstepped surfaces.

How the surface stores charge
The team focused on a property called differential capacitance, which reflects how easily extra charge can be added to the interface as the voltage is varied. For flat platinum the capacitance shows a clear minimum at a particular voltage, closely related to the potential of zero free charge, the point where the metal surface carries no net free charge. The stepped surfaces still show a similar minimum, but its depth and position change with the density and type of steps. For one family of steps, the minimum capacitance becomes smaller as more steps are added, while for the other family it becomes larger. This reveals that the way the surface stores charge is highly sensitive to the exact microscopic shape of the metal.
Hidden role of water breaking and surface groups
These opposite trends arise from how easily water molecules split at different step types. On one kind of step, the coverage of hydroxyl groups formed from water splitting is essentially fixed over the voltage range of interest, so the steps behave mainly as static, charged features that lower the local ability to store charge. On the other kind, the amount of hydroxyl on the steps continues to vary with voltage, adding an extra contribution to the capacitance that grows as more such sites are introduced. Additional electrochemical tests and analysis support this picture, showing that only one step family exhibits strong, voltage-dependent adsorption processes in the relevant window.
Connecting measurements and microscopic models
To understand how these surface groups and steps shift the special zero-charge voltage, the authors combined two types of modeling. Atomistic simulations of platinum in water showed that adding hydroxyl at step edges raises the zero-charge potential and weakens the expected link between this potential and the work function, a measure of how tightly the metal holds its electrons. A complementary continuum model treated the surface as a mosaic of step and terrace regions, each with its own interfacial properties. This model indicated that, when the coverage of adsorbed species does not change with voltage, the voltage where the capacitance is minimal remains a good proxy for the overall zero-charge potential even on complex, stepped surfaces.
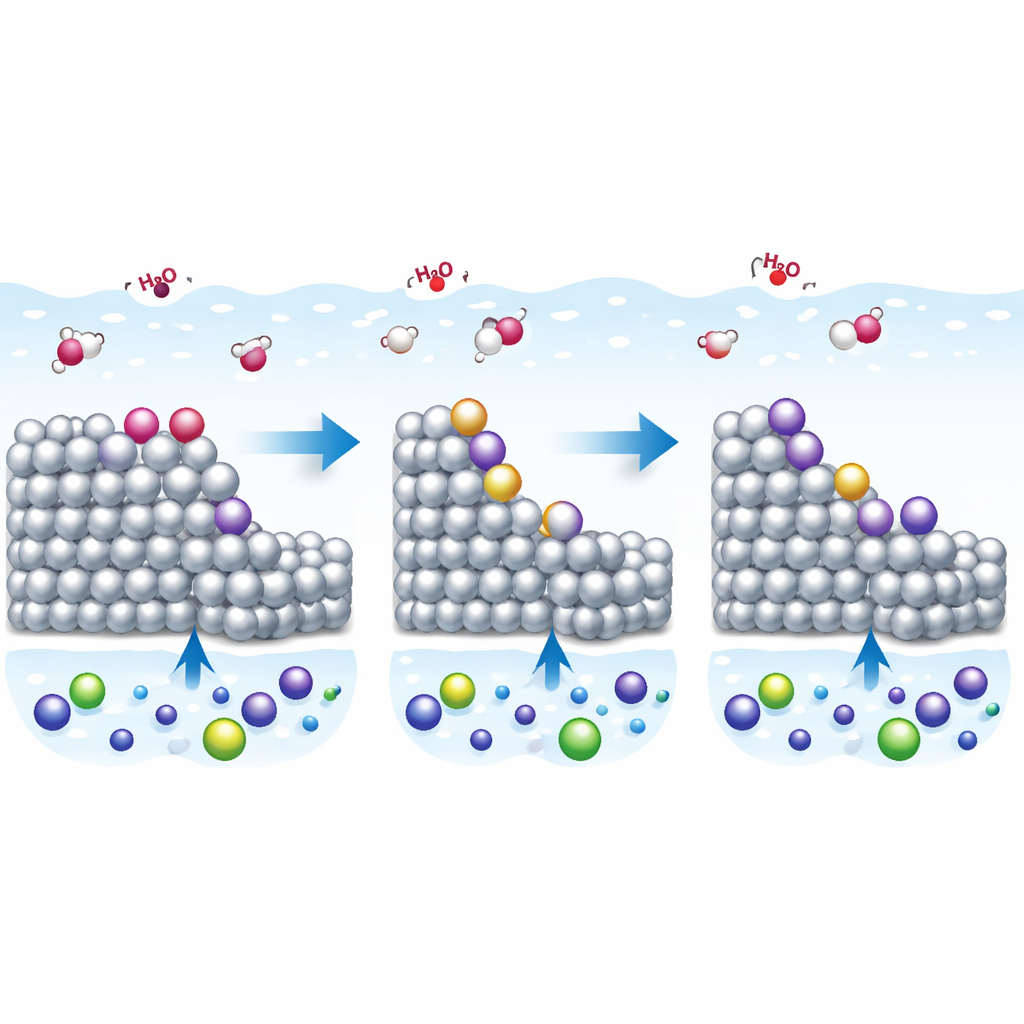
What this means for real catalysts
Taken together, the experiments and simulations provide a coherent picture of how atomic steps and their adsorbed species reshape the electric double layer on platinum. They show that both the density and nature of these steps can significantly alter how charge is stored, and that seemingly subtle surface chemistry, such as hydroxyl coverage, can shift key reference voltages. For designers of fuel cell electrodes and other electrochemical devices, this work highlights that interpreting current–voltage curves requires careful attention to surface structure, not just overall composition. By clarifying these links, the study moves us closer to predicting and tuning the reaction environment on realistic, defect-rich platinum surfaces.
Citation: Fröhlich, N.L., Liu, J., Ojha, K. et al. A comprehensive model for the electric double layer of stepped platinum electrodes. Nat. Chem. 18, 905–912 (2026). https://doi.org/10.1038/s41557-025-02063-9
Keywords: electric double layer, platinum electrodes, electrocatalysis, surface steps, interfacial capacitance