Clear Sky Science · en
Carboxylated wood membranes for selective capture and recovery of critical and commodity metal cations
Turning Trees into Water Cleaners
As the world rushes to build more batteries, mine more metals, and recycle more industrial waste, our rivers and lakes are quietly collecting the leftovers: metal ions such as lithium and iron that can harm ecosystems but are also too valuable to throw away. This study explores an unexpected helper in this challenge—ordinary spruce wood, gently “tuned” with simple chemicals so that it can pull useful metals out of water, potentially turning a low-cost, renewable material into a smart filter for both cleaning and resource recovery. 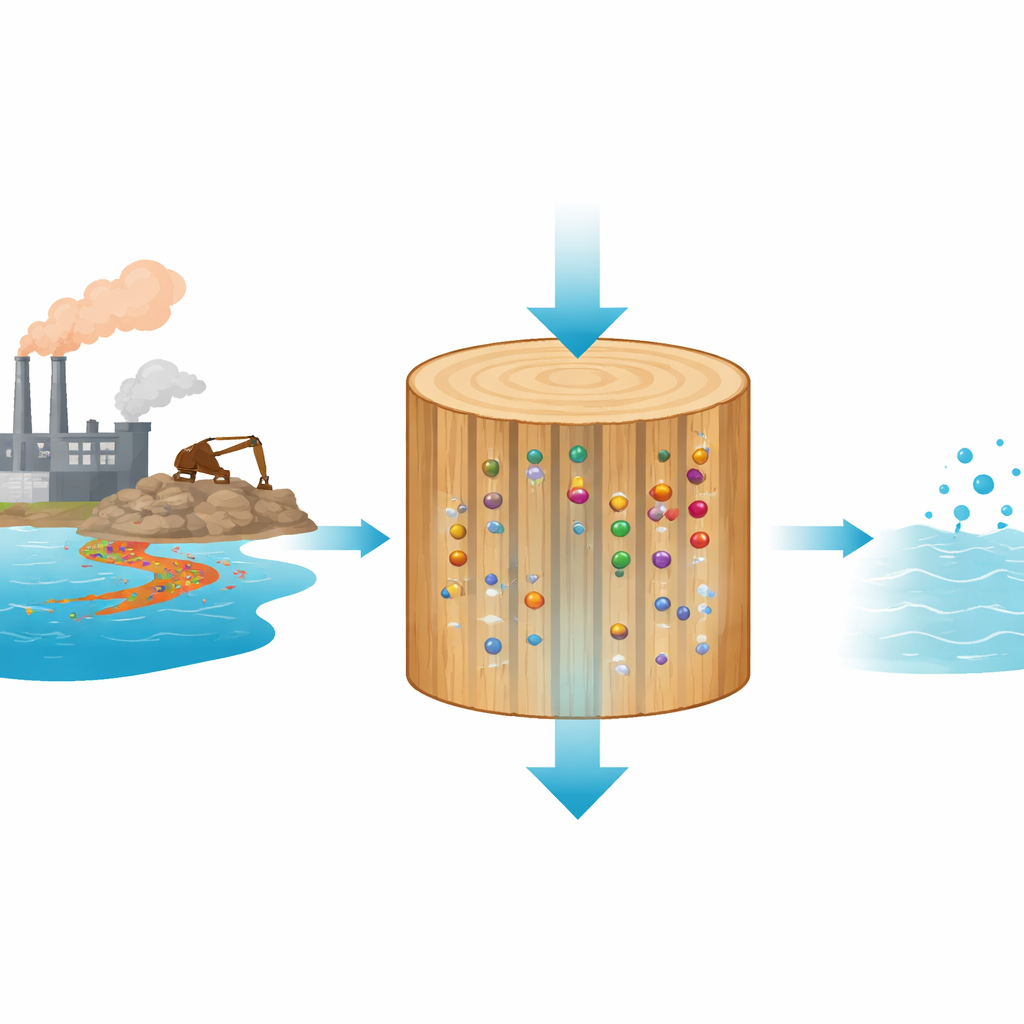
Why Metals in Water Are a Hidden Problem
Metals like copper, manganese, lead, iron, cobalt, and lithium leak into water from mining, battery production, electroplating, and other industries. Even tiny amounts can be toxic and persist in the environment for years. Today’s treatment methods—such as reverse osmosis, ion-exchange resins, and advanced adsorbents—often work well but can be expensive, energy-hungry, prone to clogging, and based on fossil-derived plastics or ceramics. Researchers are therefore searching for bio-based materials that are cheaper, easier on the planet, and still capable of selectively grabbing metals from flowing water so they can be recovered and reused.
Wood as a Natural Flow-Through Sponge
Wood might seem like an unlikely membrane, but it has a built-in advantage: a forest of tiny, aligned channels originally used by trees to move water. These microchannels allow water to pass through under simple gravity while offering long pathways where contaminants can be trapped. Earlier work showed that wood can filter out particles, bacteria, and some organic pollutants mostly by size exclusion and surface adsorption. The authors of this paper take that idea further: instead of removing the tree’s natural structure, they keep the wood intact and add chemical groups that can specifically swap harmless ions in the wood for metal ions in the water.
Smart Chemical Tweaks to the Wood Surface
The team treated thin discs of spruce with two small molecules—succinic anhydride and maleic anhydride—that react with natural “handle” groups in wood to introduce carboxyl groups, which can hold a sodium ion. After a separate “charging” step with a sodium solution, these sodium-bearing sites act as exchange points: when water containing lithium or iron flows through, the sodium ions are released and the metal ions take their place. Tests using infrared spectroscopy, electron microscopy, X-ray scattering, thermal analysis, and moisture sorption all confirmed that these new sites were successfully installed without destroying the wood’s channel structure. Succinic treatment added more of these groups but made the wood swell, warp, and become more brittle, whereas maleic treatment added slightly fewer groups but preserved rigidity and overall architecture much better.
How Well the Wood Filters Actually Work
Under gravity-driven filtration—no pumps, just water poured on top—the modified wood discs were challenged with dilute lithium and iron solutions over three reuse cycles. Maleic-treated membranes removed almost all lithium (about 99.9%) and kept that performance over repeated use, while succinic-treated ones were more variable and typically captured around 90%. For iron, the difference was even clearer: maleic-treated wood consistently removed roughly 72% of dissolved iron, far better than the succinic version, and vastly better than unmodified wood, which barely retained any metal. Separate equilibrium tests showed that both metals bind to a uniform layer of sites that behave much like a monolayer coating, with iron binding more strongly than lithium—consistent with its higher charge and ability to clasp onto several carboxyl groups at once. 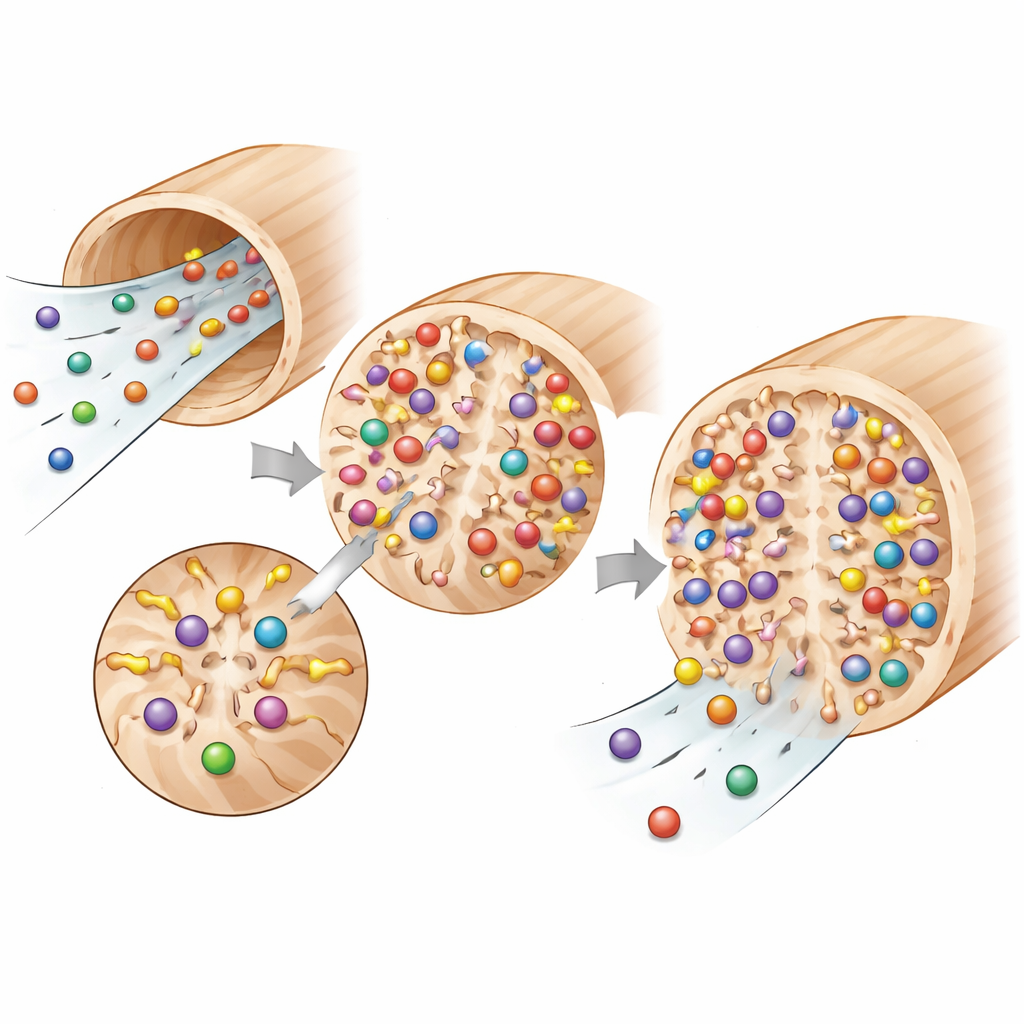
What Makes Maleic-Modified Wood Special
The superior performance of maleic-treated wood comes down to how its added groups are arranged and how easily they stay ionized in water. Maleic anhydride tends to place two carboxyl groups close together, allowing multidentate binding where one iron ion can be held by several “arms” at once. It also makes the surface more acidic, so these sites remain active over a wide pH range typical of real water streams. Succinic anhydride, in contrast, spreads the carboxyl groups further apart and is more sensitive to small pH changes, so not all sites are available at all times. Even though succinic treatment loads more groups into the wood overall, many of them are less effective in practice, and the heavier modification compromises mechanical stability.
From Lab Concept to Greener Metal Recovery
In plain terms, this study shows that a simple chemical upgrade can turn a slice of spruce into a reusable filter that both cleans water and captures metals worth recovering. The maleic-treated wood, in particular, strikes a promising balance between strength, water flow, and metal uptake, removing nearly all lithium and a large share of iron while still allowing practical throughputs under gravity alone. Although future work must test these membranes in more complex mixtures—where common ions like sodium, calcium, and magnesium will compete for space—the concept points toward low-cost, bio-based filters that help close loops in metal use, reduce waste from emerging industries like battery manufacturing, and tap into the filtering power of a material as familiar and renewable as wood.
Citation: Sánchez-Ferrer, A., Upadhye, D., Ahmed, M.J. et al. Carboxylated wood membranes for selective capture and recovery of critical and commodity metal cations. npj Clean Water 9, 36 (2026). https://doi.org/10.1038/s41545-026-00577-4
Keywords: wood membranes, metal ion removal, lithium recovery, sustainable water treatment, bio-based filtration