Clear Sky Science · en
Waste-to-sensor: high-sensitivity detection of heavy metals in water using Arc-ferrite/N-rGO nanocomposites from industrial and electronic waste
Turning Trash into a Tool for Safe Water
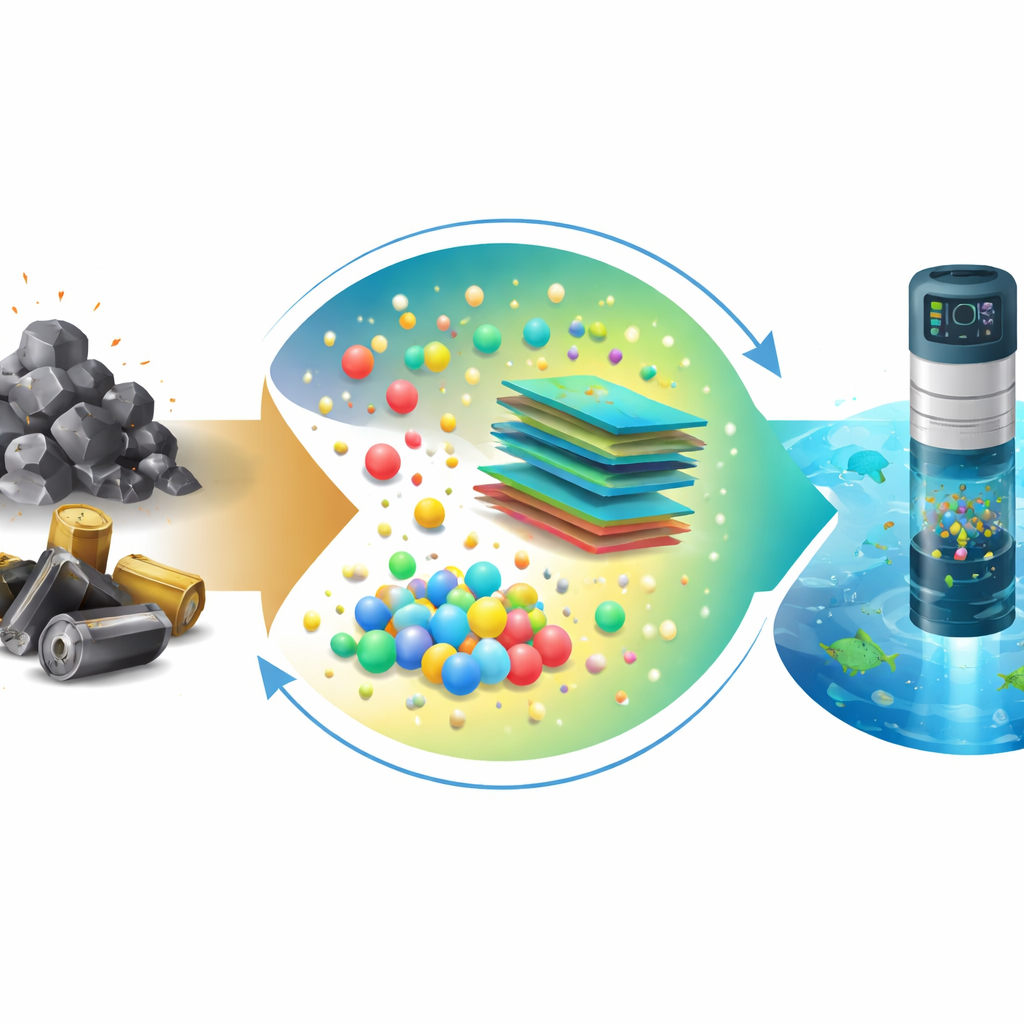
Industrial plants and our growing mountains of electronic gadgets leave behind two big problems: toxic metal pollution in water and hazardous solid waste that is costly to dump. This study offers a way to tackle both at once. The researchers show how to turn steelmaking dust and used batteries into a new type of water sensor that can spot dangerous metals like lead, cadmium, and mercury at extremely low levels—below the limits set for drinking water.
Why Waste and Water Are Deeply Connected
Modern industry generates vast quantities of waste that are rich in metals but poor in treatment options. Steel factories produce fine dust loaded with iron and zinc, which is often labeled hazardous and landfilled at great expense. At the same time, billions of disposable batteries are thrown away, and their graphite cores are usually ignored despite their value. In parallel, toxic metals such as lead, cadmium, and mercury seep into rivers, lakes, and groundwater, where they accumulate in living organisms and pose serious risks to the brain, kidneys, bones, and developing children. Traditional laboratory methods for measuring these metals are precise but bulky, expensive, and slow, making them hard to use where contamination actually occurs.
Building a New Material from Old Scraps
The team set out to design a sensor using only materials recovered from industrial and electronic waste streams. They extracted iron-rich components from electric arc furnace dust and converted them into tiny magnetic particles known as ferrites. At the same time, they purified the graphite from discarded batteries and transformed it into ultra-thin carbon sheets, then gently altered their chemistry to make them more conductive and sprinkled with nitrogen atoms. When these ferrite particles were anchored onto the nitrogen-doped carbon sheets, they formed a nanocomposite—a finely structured blend at the scale of billionths of a meter—with a high surface area and a stable, negatively charged surface that naturally attracts positively charged metal ions in water.
From Nanomaterial to Working Sensor
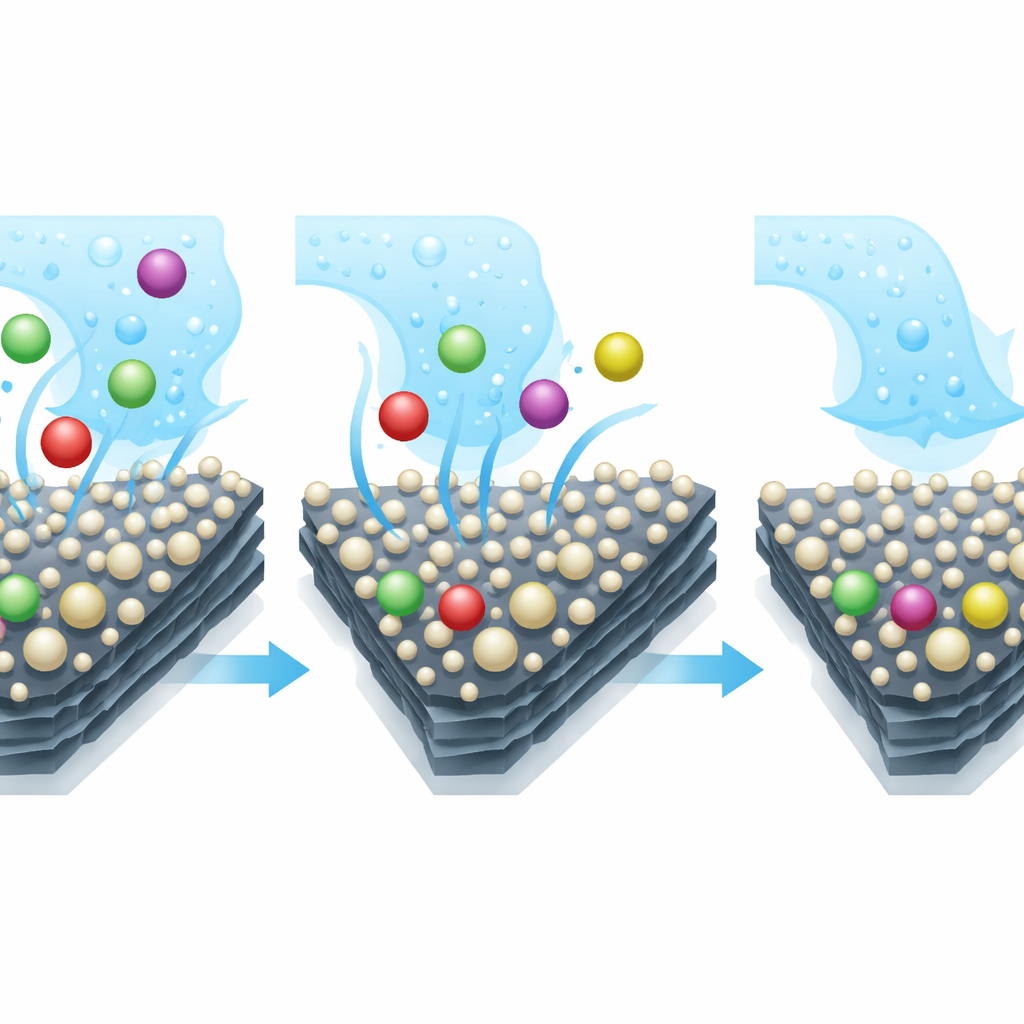
To turn this nanocomposite into a practical device, the researchers mixed it into a simple carbon paste electrode, the core of a small electrochemical sensor. They tested how well this modified surface moved electrons compared with an unmodified carbon paste. The new material boosted the useful sensing current by about three and a half times and lowered the resistance to electron flow, meaning it could respond more strongly and quickly when metal ions were present. The team then carefully tuned the surrounding solution conditions—such as the type of salt solution, its acidity, and how long metal ions were allowed to accumulate on the sensor—so that lead, cadmium, and mercury could all be measured together without interfering with each other.
Seeing Invisible Poisons at Tiny Levels
Under these optimized conditions, the waste-derived sensor could detect all three metals in water at concentrations around one part per billion, far below the safety limits set by the World Health Organization for drinking water. It responded in a straight-line fashion over a broad range of concentrations, which is essential for reliable measurement. Even when the water contained a hundred times higher amounts of other common ions, the signals for lead, cadmium, and mercury barely changed, showing that the surface of the nanocomposite prefers these toxic metals. The researchers also tested real industrial wastewater and compared their sensor’s readings to those from a sophisticated laboratory instrument. The two methods agreed closely, confirming that the simple sensor can perform in demanding, real-world samples.
A New Loop for a Circular Economy
By transforming troublesome steelmaking dust and discarded battery graphite into a high-performance water sensor, this work shows how environmental burdens can become valuable resources. Instead of paying to bury or burn these wastes, industries could feed them into a new loop that produces tools for monitoring the very pollutants they help create. For non-specialists, the key message is that it is now possible to build sensitive, low-cost detectors for harmful metals in water entirely from industrial leftovers. This "waste-to-sensor" approach points toward a more circular, sustainable future in which cleaning up pollution and reusing waste go hand in hand.
Citation: Reda, A., Eldin, N.B., Abdelkareem, S. et al. Waste-to-sensor: high-sensitivity detection of heavy metals in water using Arc-ferrite/N-rGO nanocomposites from industrial and electronic waste. npj Clean Water 9, 32 (2026). https://doi.org/10.1038/s41545-026-00567-6
Keywords: heavy metal sensors, water quality, electronic waste recycling, nanocomposites, circular economy