Clear Sky Science · en
Effects of feeding strategies on culture performance and product quality in NISTCHO
Why feeding cells matters for modern medicines
Monoclonal antibodies are key drugs for treating cancer and immune diseases, and most are made in large tanks of hamster cells. This study asks a simple but important question: how does “feeding” these cells with sugar and nutrients over time change both how well they grow and the fine details of the antibodies they make? Using a shared community cell line called NISTCHO, the researchers show how adjusting when and what the cells eat can tune drug output and subtle sugar patterns that affect safety and activity.
Growing medicine factories in a bottle
The team worked with NISTCHO, a standardized hamster cell line that makes a model antibody called cNISTmAb. Because many labs can use the same cells, NISTCHO is becoming a reference system for comparing production methods. Here, the authors ran small fed-batch cultures, where cells grow for about eleven days while fresh nutrients are added on a schedule. They tested seven different feeding plans that varied in how much glucose was supplied, how often fresh feed was added, and whether an extra mix containing galactose and manganese, called Gal+, was included.
How sugar supply shapes growth and yield
Glucose, the main energy source, turned out to be the strongest driver of cell growth and antibody yield. When enough glucose was supplied every other day, cells stayed healthy, reached high cell densities, and produced the highest antibody titers. When glucose additions were cut in half, cultures ran out of fuel earlier, cell numbers dropped quickly, and antibody production stalled. An alternative plan that added smaller glucose amounts more frequently limited waste buildup but also reduced productivity, likely because the sugar level spent too much time near the lower limit that cells can efficiently use.
Fine-tuning the sugars on antibodies
Antibodies carry small sugar chains that influence how long they last in the body and how strongly they engage the immune system. The researchers carefully measured these sugars and used statistical tools usually applied to big data sets. They found that time in culture and the feeding strategy both shaped the sugar patterns. Across all conditions, later samples showed more of certain sugars that include fucose and fewer versions lacking this sugar, changes that may reduce some immune functions. Adding the Gal+ mix consistently increased galactose on the antibody sugars, a change known to boost one type of immune activity, without harming cell growth.
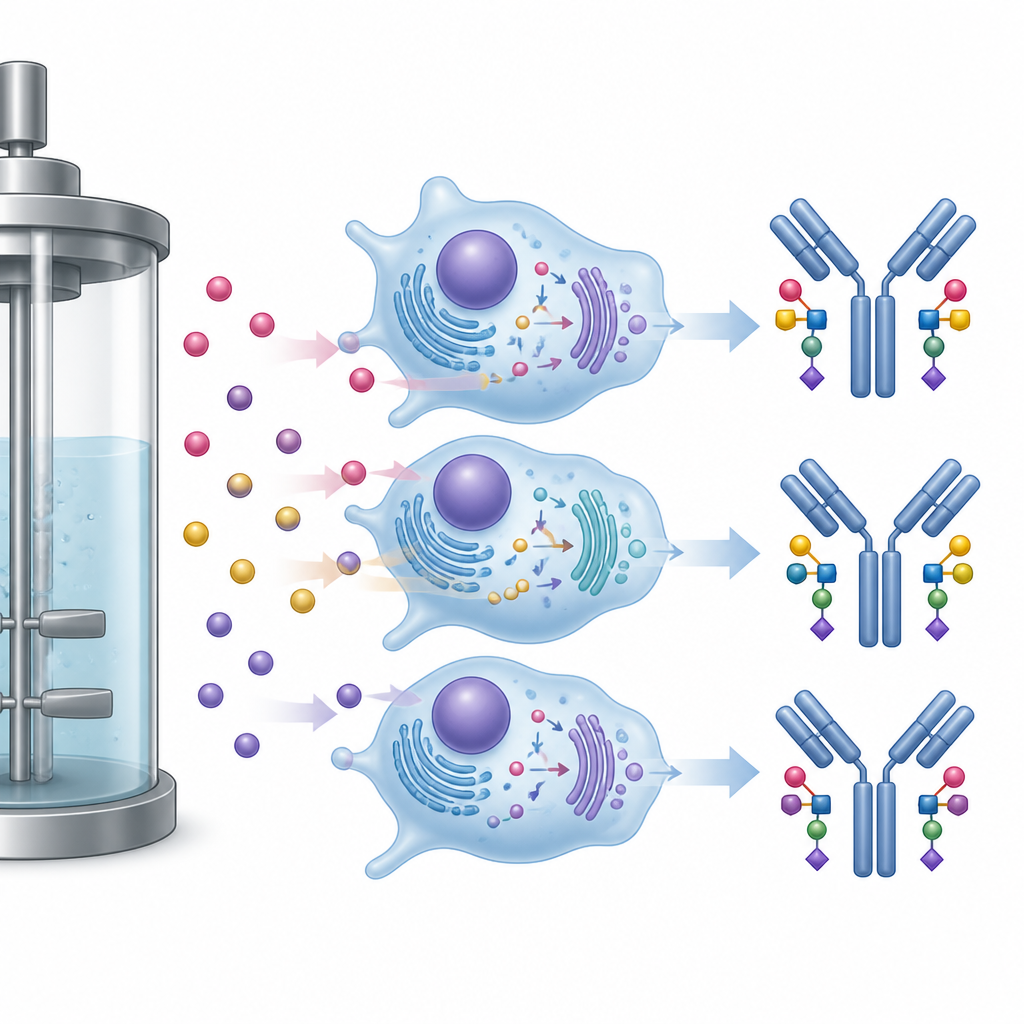
Hidden effects of sugar spikes on side reactions
Glucose in the liquid can also attach directly to the antibody in a non-enzymatic reaction called glycation, which can subtly change its properties and complicate analysis. The study showed that feeding frequency and residual glucose levels influenced how much glycation occurred during the rapid growth phase. Strategies with daily glucose additions tended to have more glycated antibody early on, likely because more free sugar remained in the medium around fewer cells. By the end of the culture, though, these differences narrowed, and overall glycation stayed low, around five percent, across all strategies.
What this means for making better biologic drugs
Together, the results show that simply changing how and when nutrients are added can steer both the amount and quality of antibodies made by NISTCHO cells. High but controlled glucose supply supports strong productivity, while the Gal+ supplement reliably raises galactose on antibody sugars and helps cells stay alive under leaner conditions. Because NISTCHO is a public reference system, these findings give drug makers and researchers a shared roadmap for adjusting feeding strategies to fine-tune antibody sugar patterns, while keeping undesirable changes like excess fucosylation and glycation in check.
Citation: Schäpertöns, V., Hofer, L., Berger, T. et al. Effects of feeding strategies on culture performance and product quality in NISTCHO. npj Syst Biol Appl 12, 67 (2026). https://doi.org/10.1038/s41540-026-00686-3
Keywords: monoclonal antibody production, CHO cell culture, glycosylation, bioprocess feeding strategy, NISTCHO