Clear Sky Science · en
Umami induces pulmonary epithelial senescence via L-glutamic acid-triggered minority MOMP
Why the taste of "savory" may matter to your lungs
Many people know monosodium glutamate (MSG) as the ingredient that gives foods a rich, savory "umami" taste. This study suggests that when MSG and other glutamate-heavy diets are consumed in large amounts over long periods, they may quietly affect lung health. Using mice and lung cells, the researchers traced how excess dietary glutamate can push lung cells into an aged, worn-out state, potentially worsening chronic lung diseases. Their work points to a new link between what we eat and how our lungs age.
From dinner plate to breathing problems
The team began by feeding mice water laced with high levels of MSG for several weeks. They measured how the animals’ lungs worked using a specialized ventilation system. Over time, the mice showed stiffer lungs and reduced ability to expand and take in air—signs of impaired breathing. When the researchers examined lung tissue under the microscope, they saw increasing areas of injury and early scarring. Measurements of lung chemistry revealed that MSG raised levels of glutamate, the amino acid that carries umami flavor, and that higher glutamate strongly tracked with worse lung function and more damage.
Different rich diets, same problem
MSG is not the only way to raise glutamate in the body, so the scientists tested two other eating patterns known to boost glutamate: a long-term high-fat diet and a high-protein diet. Although these diets had very different effects on body weight, both led to more lung injury and fibrous tissue in mice, along with higher glutamate levels in the lungs. Again, the more glutamate present, the more damage the researchers saw. This suggested that glutamate itself, rather than some unique property of MSG, was a common driver linking rich diets to lung harm.
How lung cells grow old before their time
To understand what glutamate was doing inside the lungs, the team focused on the thin layer of cells lining the air sacs, which are critical for gas exchange. They looked for signs of cellular senescence, a state in which cells stop dividing, secrete inflammatory substances, and gradually degrade nearby tissue. In mice on a high-MSG diet, a larger fraction of these epithelial cells carried a key senescence marker. In cultured human lung cells exposed to glutamate, the same markers rose, and the cells showed classic aging-associated staining and increased release of inflammatory molecules such as IL-6 and IL-8. When the researchers treated MSG-fed mice with a combination of senescent-cell–targeting drugs, lung function improved and tissue damage lessened, even though glutamate levels stayed high. This indicated that the build-up of aged cells was a major reason the lungs were failing.
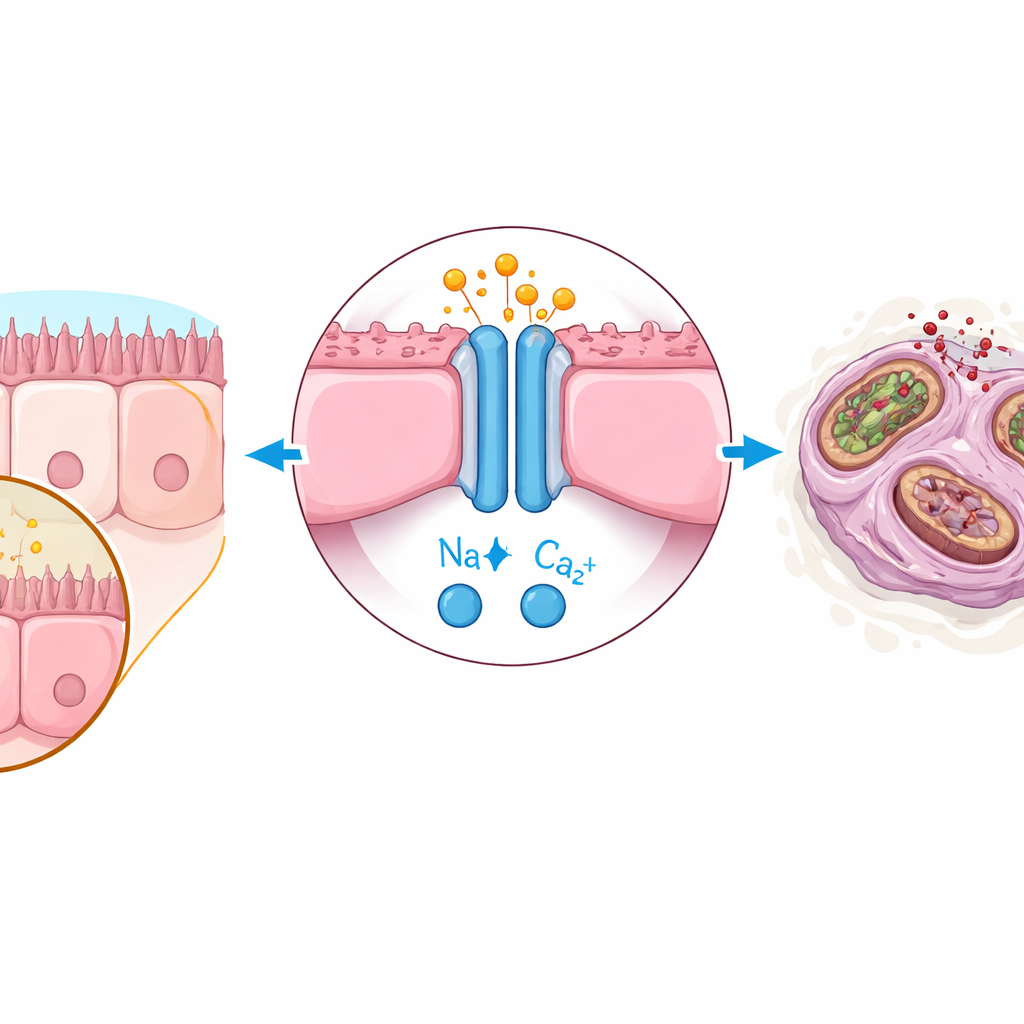
A hidden chain reaction inside lung cells
Digging deeper, the study uncovered the internal chain reaction that links glutamate to cell aging. Lung cells carry a receptor called NMDAR, best known from brain cells where it responds to glutamate. In the lungs, excess glutamate overactivated this receptor, which in turn disturbed mitochondria, the cell’s energy producers. Instead of a full-scale breakdown, the mitochondria underwent a limited leak of contents, a process called minority mitochondrial outer membrane permeabilization (miMOMP). This subtle injury was enough to switch on a normally death-related enzyme, caspase-3, at low levels that did not kill the cell but nudged it into a senescent state. Blocking NMDAR with drugs such as memantine, or inhibiting caspases, reduced mitochondrial stress, dampened senescence markers, and protected lung tissue in mice.
When scar-forming lung disease meets a rich diet
The team then asked what happens if a high-glutamate diet is layered on top of an existing lung injury. They used bleomycin, a drug that provokes lung scarring resembling pulmonary fibrosis in mice. As expected, bleomycin alone caused injury, collagen build-up, and more senescent epithelial cells. But when combined with MSG, a high-fat diet, or a high-protein diet, the damage and fibrosis were significantly worse, and lung glutamate levels climbed even higher. These findings imply that glutamate-heavy diets may not only harm otherwise healthy lungs but can also accelerate the course of established fibrotic lung disease.
What this means for everyday life
For non-specialists, the message is not that any single serving of savory food will damage the lungs, but that long-term, heavy exposure to glutamate—from MSG and from rich, fat- or protein-heavy diets—may contribute to lung aging and scarring. The work identifies dietary glutamate as a potentially adjustable risk factor for chronic lung disease and highlights a specific pathway—the NMDAR–miMOMP–senescence axis—that future drugs might target. While more work in humans and at more typical intake levels is needed, this study opens the door to thinking about lung health not only in terms of air quality and smoking, but also in terms of how our daily food choices shape the aging of the organs that help us breathe.
Citation: Zhang, J., Zhao, Y., Yang, B. et al. Umami induces pulmonary epithelial senescence via L-glutamic acid-triggered minority MOMP. npj Sci Food 10, 125 (2026). https://doi.org/10.1038/s41538-026-00783-x
Keywords: monosodium glutamate, lung aging, cellular senescence, diet and lung disease, pulmonary fibrosis