Clear Sky Science · en
StimVision: smartphone video kinematics to optimize DBS programming in Parkinson’s disease
Turning Everyday Phones into Brain-Tuning Tools
For people with Parkinson’s disease, deep brain stimulation (DBS) can be life-changing, but finding the best settings for each person is still a slow, trial‑and‑error process. This study introduces “StimVision,” a way to use nothing more than a standard smartphone camera and smart algorithms to watch how a patient moves and quickly suggest which DBS setting works best. The approach promises to make brain stimulation more precise, more objective, and potentially easier to deliver in ordinary clinics—or even at home.
Why Tweaking Brain Implants Is So Hard
DBS works by sending tiny electrical pulses to deep regions of the brain to ease movement problems such as slowness and stiffness. Today, doctors typically adjust these pulses by eye, relying on short exams and rating scales to judge whether a patient’s movements look a bit better or worse. As modern DBS systems offer more and more adjustable settings, this process becomes increasingly demanding and subjective. Clinicians lack a simple, quantitative way to compare many possible programs within a single visit and to know which one truly gives the strongest benefit.
Watching the Hand to Read the Brain
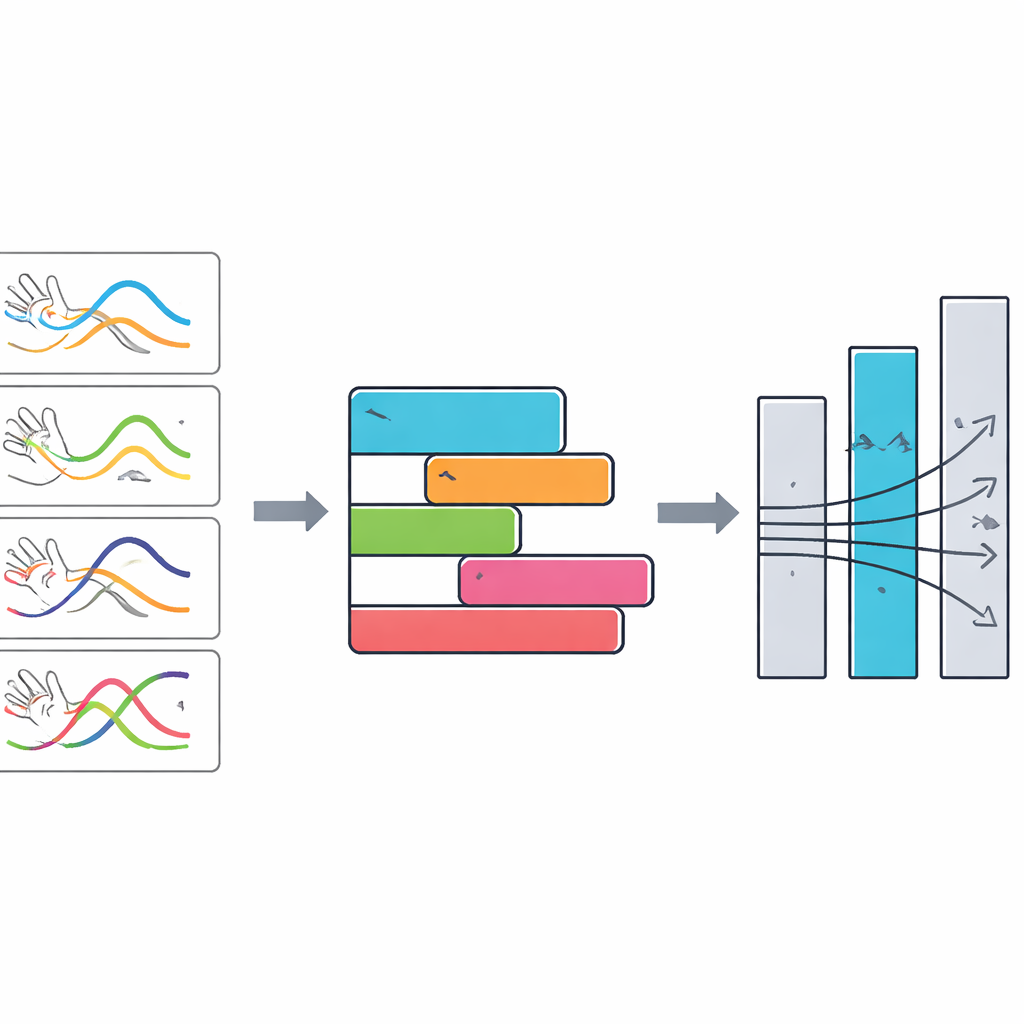
The researchers reframed the problem as a selection task: given several DBS settings, which one produces the best movement for this specific person right now? Fifteen people with Parkinson’s disease and DBS implants performed a simple hand opening–closing task—the same motion already used in clinical exams—while being filmed with a 60‑frame‑per‑second smartphone camera. Using an open‑source computer vision tool, the team tracked the position of the hand without markers and converted the video into a precise signal describing how the fingers opened and closed over time. From this signal they extracted 23 numerical features capturing how big, how fast, how regular, and how steady the movements were, such as average speed, frequency of cycles, and how much the movement slowed down during the short sequence.
Turning Motion into an Objective Score
To compare DBS settings, the team invented a patient‑specific metric called the Dynamically Weighted Improvement Score (DWIS). For each person, the software first calculated how much each of the 23 movement features improved or worsened compared with DBS turned off, always orienting the numbers so that higher meant better movement. It then identified which features varied the most between programs—those were considered the most informative for that patient—and automatically gave them more weight. By summing the weighted improvements, the system produced a single composite score for each DBS setting and ranked all programs within that session. In every patient, StimVision identified one clearly best program, and this ranking remained stable even when the weighting method was altered, suggesting the approach is robust rather than overly sensitive to tuning choices.
What Better Movement Really Looks Like
When the researchers compared each person’s best DBS setting with their DBS‑off state, they found broad gains. Most tested programs helped to some degree, but the top‑ranked ones produced pronounced improvements. The strongest effects were increases in movement speed and rhythm: hands opened and closed faster, with higher average velocity and quicker closing phases, and the movements stayed more consistent across the short trial instead of fading or slowing. Using a statistical technique that groups related features together, the team found that these many measurements could be distilled into three main themes or “domains”: Movement Speed, Movement Consistency, and Rhythm & Timing. They then compared this DBS “signature” with data from an earlier study of the common Parkinson’s drug levodopa. The two treatments showed similar patterns in speed and consistency, but differed in timing‑related features, hinting that electrical and drug therapies act partly through shared pathways yet shape the fine timing of movement in different ways.
Matching the Computer to the Clinician’s Eye
For any new measurement to be useful in medicine, it must agree with what clinicians already recognize as meaningful improvement. The researchers therefore tested how DWIS related to scores on a standard clinical scale for hand movements, rated by neurologists who were unaware of the algorithm’s outputs. Across all tested programs, higher DWIS values were strongly associated with better clinician ratings, even after accounting for how impaired the hand was at baseline. This indicates that the smartphone‑based score is not just capturing mathematical noise; it aligns with expert judgment while providing a more fine‑grained and reproducible readout than can be seen by eye alone.
What This Could Mean for Patients
Although this proof‑of‑concept study involved only 15 people and focused on a single hand task, it points toward a future in which DBS programming is guided by fast, objective measurements captured with everyday devices. StimVision could help clinicians navigate the growing maze of stimulation settings, shorten the time spent on trial‑and‑error adjustments, and eventually support remote assessments outside the hospital. By giving doctors a clear, data‑driven picture of how each DBS program shapes speed, steadiness, and rhythm of movement—and by offering a common language to compare brain stimulation with medications—the approach may ultimately help tailor Parkinson’s treatment more precisely to each individual’s needs.
Citation: Lange, F., Köberle, P., Adaçay, G. et al. StimVision: smartphone video kinematics to optimize DBS programming in Parkinson’s disease. npj Parkinsons Dis. 12, 100 (2026). https://doi.org/10.1038/s41531-026-01335-6
Keywords: Parkinson’s disease, deep brain stimulation, smartphone video, motor kinematics, computer vision