Clear Sky Science · en
SNCA triplication disrupts proteostasis and extracellular architecture prior to neurodegeneration in human midbrain organoids
Why this research matters to everyday life
Parkinson’s disease is best known for its tremors and movement problems, but long before symptoms appear, brain cells are already struggling to keep their internal and external environments in balance. This study uses tiny lab-grown models of the human midbrain to watch those earliest changes unfold in real time. By capturing how extra copies of a key Parkinson’s gene disturb both the proteins inside nerve cells and the supportive matrix around them, the work points to new ways to detect and possibly slow the disease well before neurons start dying.
Building mini human midbrains in the lab
To explore these early events, the researchers started with human skin or blood cells that had been reprogrammed into induced pluripotent stem cells. Some of these cells carried three copies of the SNCA gene, which encodes the protein alpha-synuclein and is known to cause aggressive, early-onset Parkinson’s when duplicated. Others were carefully corrected versions from the same patient, serving as a genetic control, along with a line in which SNCA was deleted. By guiding these cells through a stepwise recipe, the team grew three-dimensional midbrain organoids—spherical clusters that mimic the human midbrain’s architecture and contain dopamine-producing neurons, the very cells that degenerate in Parkinson’s. 
Early stress inside neurons without a protein shutdown
The scientists examined these organoids at 50 and 100 days of growth, a window when neurons are maturing but large-scale cell death has not yet set in. Organoids with SNCA triplication showed strongly elevated alpha-synuclein and its disease-linked modified form, along with naturally formed neuromelanin, a pigment found in human midbrain neurons. Inside these cells, several major control switches that regulate protein production and stress responses became overactive, especially the mTOR, AKT, ERK, and eIF2α pathways. Surprisingly, despite this surge in “growth and stress” signals, the overall rate of new protein synthesis remained steady. Detailed ribosome profiling revealed that instead of a blanket slowdown or speedup, cells were fine-tuning how specific messages were translated into proteins—a buffering strategy that appears to keep protein output in check even as gene activity changes.
Shifts in nerve cell wiring and the surrounding scaffold
To understand the consequences of this altered signalling, the team surveyed thousands of proteins and RNA molecules at once. They found that organoids with extra SNCA showed a coordinated rise in many proteins involved in nerve communication—such as those related to synapses, signal release, and axon growth—suggesting that neuronal circuits are being rewired. At the same time, there was a broad decline in components of the extracellular matrix, the mesh-like scaffold that stabilizes cells and shapes their connections. Intriguingly, certain matrix elements that form dense coats around neurons, called perineuronal nets, were increased at the RNA and protein levels even as other matrix components fell, hinting at a complex, uneven remodeling of the brain’s structural environment.
Thickening nets around vulnerable dopamine neurons
Using high-resolution confocal imaging, the researchers visualized this remodeling directly. In organoids with SNCA triplication, they observed stronger staining for pericellular and interstitial matrix structures, particularly those rich in chondroitin sulfate proteoglycans and the protein aggrecan. Around general neuronal populations, these changes became clear by day 100. However, around dopamine-producing neurons marked by tyrosine hydroxylase, the build-up of perineuronal nets appeared earlier, already evident at day 50 and persisting with maturation. Even organoids lacking SNCA entirely showed altered matrix around these neurons, indicating that both excess and absence of alpha-synuclein can disturb the delicate balance of the extracellular environment. 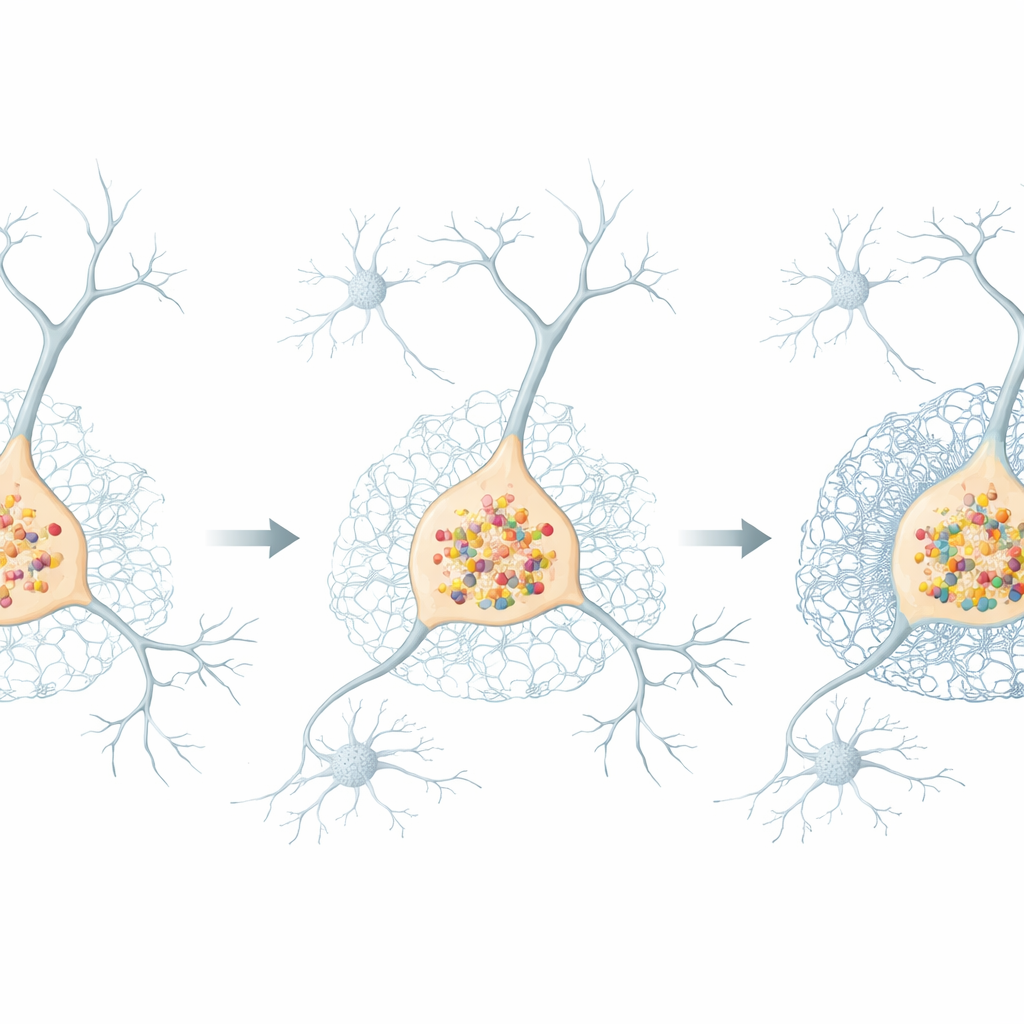
What this means for understanding Parkinson’s
Taken together, the findings show that in this human midbrain model, extra copies of SNCA disturb the cell’s protein-management systems and reshape the extracellular scaffold well before dopamine neurons begin to die. Rather than a simple overload that shuts down protein production, the cells respond with targeted adjustments in translation and with early, cell type–specific thickening of perineuronal nets. For a layperson, this suggests that Parkinson’s may start as a subtle failure of housekeeping both inside neurons and in the tissue that surrounds them, creating a stiff, altered neighborhood that gradually makes these cells more fragile. By identifying this early “proteostasis–matrix” axis, the study highlights new opportunities to detect Parkinson’s earlier and to design treatments that protect nerve cells by restoring balance to both their internal protein economy and their external support network.
Citation: Statoulla, E., Zafeiri, M., Chalkiadaki, K. et al. SNCA triplication disrupts proteostasis and extracellular architecture prior to neurodegeneration in human midbrain organoids. npj Parkinsons Dis. 12, 81 (2026). https://doi.org/10.1038/s41531-026-01292-0
Keywords: Parkinson’s disease, alpha-synuclein, midbrain organoids, perineuronal nets, proteostasis