Clear Sky Science · en
Biodegradable lean Mg-Dy alloy enhances bone repair in senile osteoporotic fractures by modulating macrophage mediated immune microenvironment
Stronger Bones from Smart Metals
Broken bones in older adults with osteoporosis are notoriously slow and difficult to heal. This study explores a new type of temporary metal implant made from magnesium and a rare earth element called dysprosium. Unlike traditional steel or titanium hardware, this metal slowly dissolves inside the body and appears to calm harmful inflammation while nudging the body’s own bone-building cells to work harder, offering a potential new path to better recovery after fractures in the elderly.
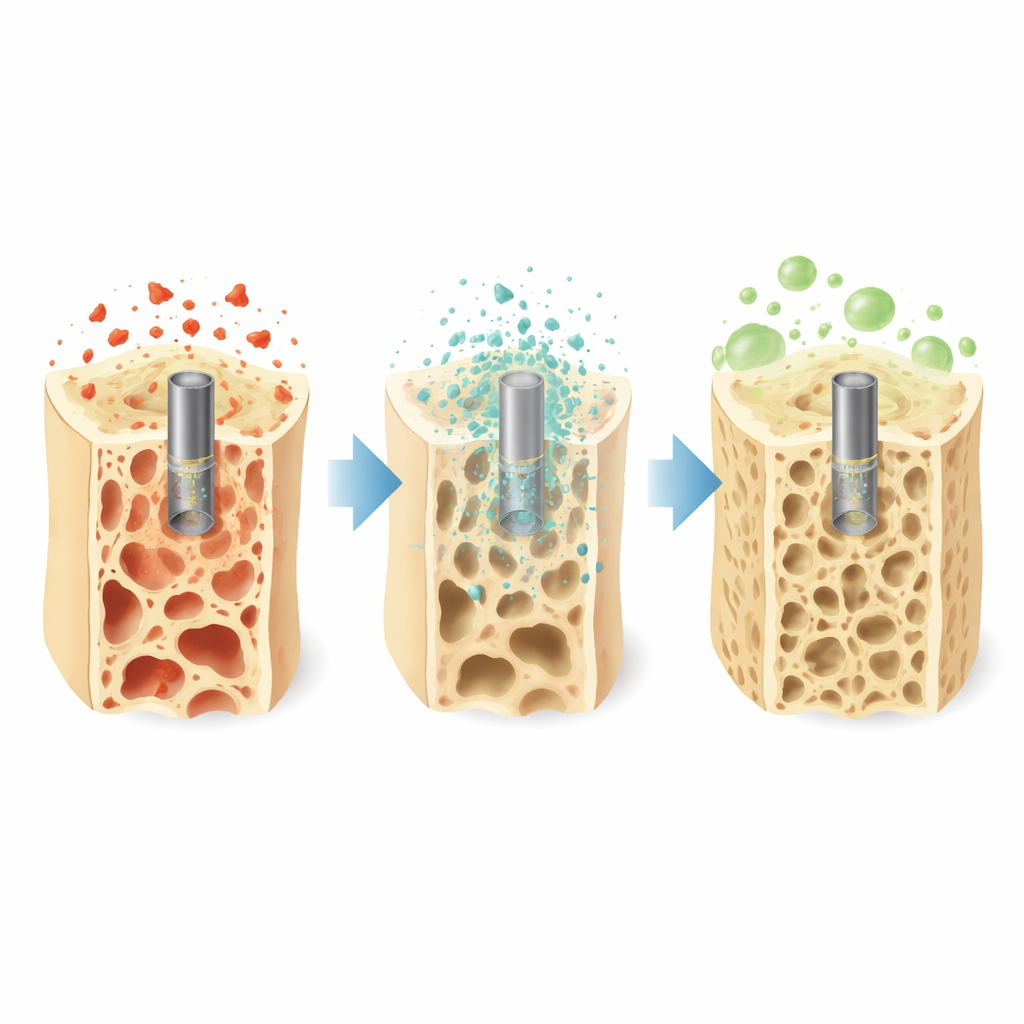
Why Fragile Bones Need a New Kind of Support
Osteoporosis makes bones thin, porous, and easy to break, especially in people over 65. Standard metal plates and screws are very strong, but they can shield the surrounding bone from normal loads, which may actually weaken bone further and sometimes require a second surgery for removal. At the same time, the aging immune system tends to stay stuck in a simmering inflammatory state. Immune cells called macrophages linger in a harsh, “angry” mode that blocks bone repair. The authors argue that ideal implants for elderly patients should not only hold bone together but also gently degrade and actively guide the immune system toward healing.
A Gentle Metal That Works with the Body
The researchers designed a simple alloy made mostly of magnesium with a tiny amount (about 1%) of dysprosium, then shaped it using a low-temperature extrusion process to tune its internal structure. This processing produced an implant strong enough to match or exceed the strength of natural bone and stiff enough to provide support without being overly rigid. When the alloy was soaked in a fluid that mimics body conditions, it corroded at a controlled pace and formed a thin, compact surface layer enriched in dysprosium, magnesium oxide, calcium, and phosphate—ingredients similar to the mineral found in bone. This protective film slowed down further breakdown, limited gas formation, and created a surface that should be friendly to surrounding tissues.
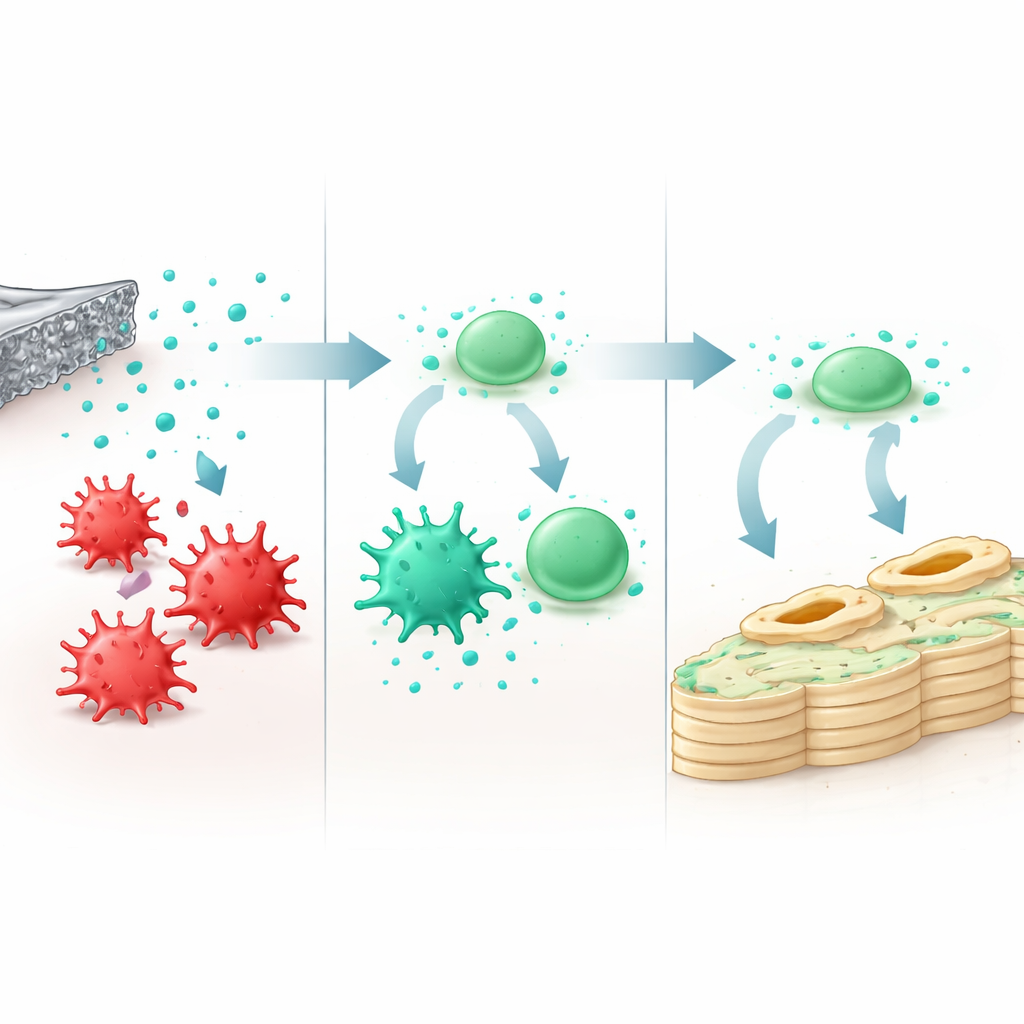
Turning Harmful Inflammation into Healing Help
To see how the material affects the immune system, the team exposed macrophages to liquid that had been in contact with the alloy. Within a safe range of magnesium ion levels (up to about 8 millimoles), the cells stayed healthy but shifted behavior. Markers of the aggressive, pro-inflammatory state dropped, while signs of a calming, pro-healing state rose. These “reprogrammed” macrophages produced fewer inflammatory molecules and less reactive oxygen, and more soothing factors associated with tissue repair. When their secretions were then applied to bone-forming stem cells from bone marrow, those stem cells turned on genes involved in early and late stages of bone building and showed higher activity of enzymes linked to mineral deposition. Molecular tests pointed to a key signaling route, known as NF‑κB, as part of the chain that connects magnesium exposure, immune cell shift, and enhanced bone formation.
Helping Old Bones Heal in Living Animals
The researchers next tested the alloy in a mouse model that mimics senile osteoporosis. They implanted small Mg–Dy rods into bone defects in aged, osteoporotic mice and followed healing over several weeks using high-resolution scans and tissue staining. Compared with control groups, bones treated with the low-temperature–processed alloy showed higher bone volume and mineral density around the implant site. Microscopy revealed richer callus (the temporary repair tissue), better transition to solid bone, and tighter integration between new bone and the degrading metal. Importantly, the alloy degraded steadily rather than falling apart too quickly, keeping mechanical support in place while new bone grew in.
A Temporary Scaffold That Guides Healing
Taken together, the study suggests that this biodegradable magnesium–dysprosium alloy can act as a smart, disappearing scaffold for fragile bones. It is strong enough to stabilize fractures, slowly dissolves into ions that the body can handle, and, crucially, nudges immune cells away from chronic inflammation and toward a healing mode that boosts bone-forming stem cells. For elderly patients with osteoporosis, such implants could one day reduce the need for removal surgeries and improve the chances that a broken bone regains its strength, by working with both the skeleton and the immune system rather than against them.
Citation: Zhou, S., Chen, X., Cai, Z. et al. Biodegradable lean Mg-Dy alloy enhances bone repair in senile osteoporotic fractures by modulating macrophage mediated immune microenvironment. npj Mater Degrad 10, 43 (2026). https://doi.org/10.1038/s41529-026-00757-0
Keywords: osteoporosis, biodegradable implant, magnesium alloy, bone healing, immune modulation