Clear Sky Science · en
Operando observation of dissolution kinetics and alteration layer formation of heavy ion irradiated borosilicate glass
Why Safer Nuclear Waste Storage Depends on Glass
Nuclear power plants leave behind highly radioactive leftovers that must be locked away safely for tens of thousands of years. One of the leading strategies is to trap these leftovers inside blocks of specially formulated glass. But over such long times, the glass is bombarded from within by radiation and bathed by underground water. This study asks a critical question: how does radiation damage change the way this glass slowly dissolves and forms a protective skin when water eventually reaches it?
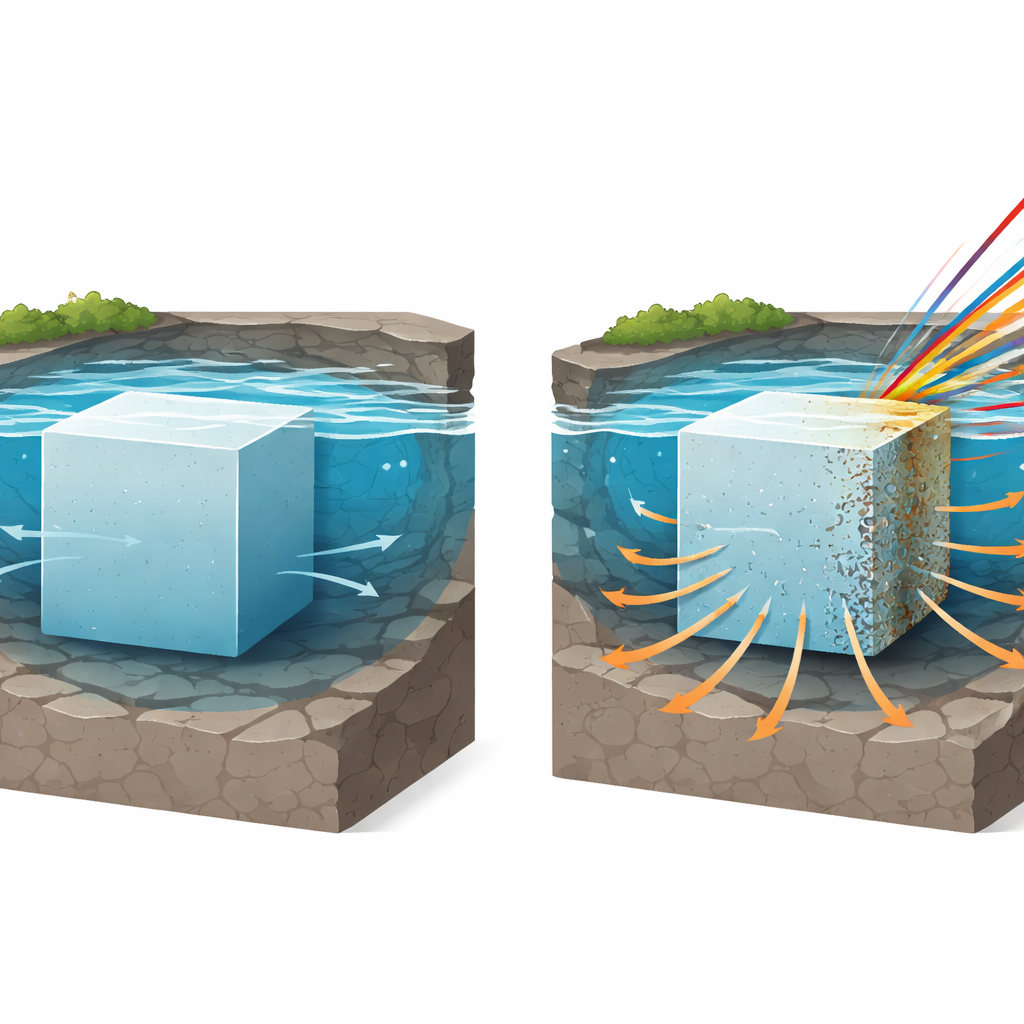
How Waste Glass Meets Water Deep Underground
Borosilicate glass, the material studied here, is already used or planned for many nuclear waste programs because it can hold many different radioactive elements and resists attack by water. When water finally reaches a glass block in a geological repository, it does not simply eat the glass away like sugar in tea. Instead, a complex thin, silica-rich “surface alteration layer” grows on the outside. This layer can slow further attack by acting as a filter and barrier. At the same time, the glass is constantly damaged from the inside by radiation from the trapped waste. That damage rearranges the glass structure on a microscopic scale, but its impact on long‑term water resistance has remained uncertain and hotly debated.
Watching Glass Corrode in Real Time
The researchers worked with a simple, well‑characterized sodium borosilicate glass. To mimic extreme self‑irradiation, they bombarded one face of a glass block with very energetic gold ions, creating a severely damaged zone about 50 micrometers thick. They then mounted the block in a heated flow cell containing a sodium bicarbonate solution, chosen to resemble mildly alkaline groundwater. Using a laser‑based method called Raman spectroscopy, they repeatedly scanned across the same microscopic line through glass, water, and the growing surface layer for almost two weeks. This operando approach allowed them to track, in real time, how fast the glass surface retreated, how the alteration layer thickened, and how the internal ring‑like building blocks of the silica network evolved.
Radiation Makes Glass More Eager to Dissolve
Comparing the irradiated side with an undamaged side of the same sample and with earlier experiments, the team found that radiation damage substantially speeds up glass dissolution. Initially, the irradiated glass dissolved about two and a half times faster than non‑irradiated glass under nearly identical conditions. As the attack progressed and the dissolution front reached the transition between heavily damaged and undamaged regions, the rate spiked again, temporarily becoming even higher. Only after the entire damaged zone had been replaced by a silica‑rich layer did the rate drop, but even the later “residual” rates stayed above those measured on the non‑irradiated side. Throughout, the Raman data showed that the radiation had broken up the original network of silicon–oxygen and boron–oxygen units, leaving more weakly connected structures that react more readily with water.
How the Protective Skin Grows and Changes
The surface alteration layer that formed over the irradiated region was roughly twice as thick as the one grown over the undamaged side. High‑resolution imaging revealed that it was not uniform: there was an outer smooth zone, an intermediate band with different internal packing, and an inner zone made of fine lamellae or stripes. Raman measurements translated these textures into differences in silica ring sizes and connectivity. Larger, more polymerized rings dominated certain zones, while smaller rings and water‑rich structures dominated others. By partially replacing the solution with heavy water (D₂O) partway through the experiment, the team could follow how water moved through this layered skin. They found that the middle zone acted as a partial bottleneck for diffusion, while the outer zone gradually became more restrictive as it matured.
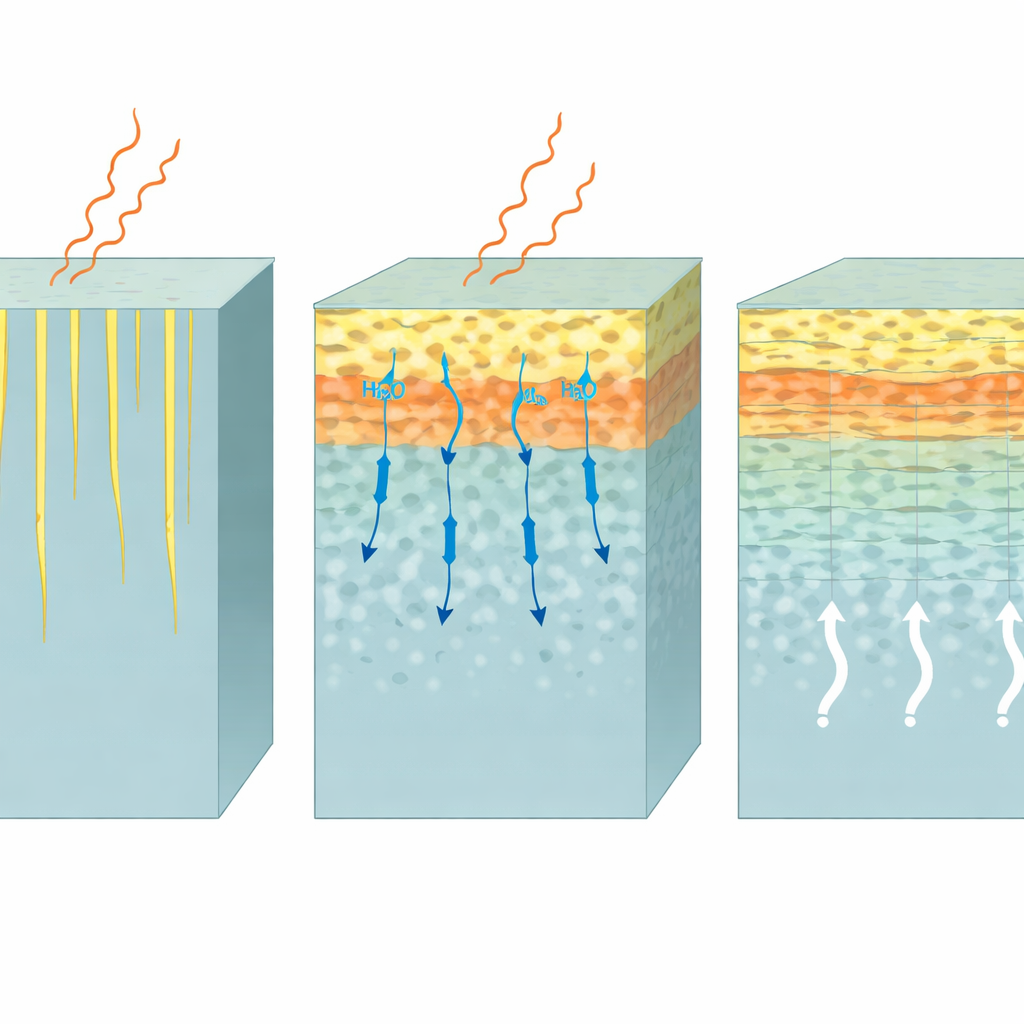
What This Means for Nuclear Waste Safety
For non‑specialists, the key message is that radiation damage makes nuclear waste glass more chemically “reactive”: it dissolves faster in water and grows a thicker but structurally more complex protective skin. The internal architecture of this skin—its bands and lamellae—strongly influences how easily water and dissolved species can move through it, and these features evolve over time. The findings support a picture in which glass dissolves and silica re‑precipitates in a tightly coupled way at a moving reaction front, rather than by simple leaching alone. For long‑term safety assessments, this means that both radiation damage and the changing structure of the surface layer must be considered when predicting how quickly radionuclides could be released from vitrified waste deep underground.
Citation: Lönartz, M.I., Stausberg, L., Fritzsche, M.B.K. et al. Operando observation of dissolution kinetics and alteration layer formation of heavy ion irradiated borosilicate glass. npj Mater Degrad 10, 45 (2026). https://doi.org/10.1038/s41529-026-00754-3
Keywords: nuclear waste glass, radiation damage, glass corrosion, surface alteration layer, borosilicate glass