Clear Sky Science · en
Multiscale kinetic model of ethylene oligomerization in Ni-NU-1000 metal-organic framework
Turning a Simple Gas into Tailor-Made Building Blocks
Ethylene, one of the most basic molecules produced in huge quantities by the chemical industry, can be stitched together into longer chains that become ingredients for plastics, detergents, and many everyday products. But industry does not just want “more” product; it wants chains of very specific lengths. This paper shows how computer modeling can predict and tune which chain lengths form inside a porous solid catalyst, potentially guiding the design of cleaner, more efficient chemical processes.
Why the Shape of a Catalyst Matters
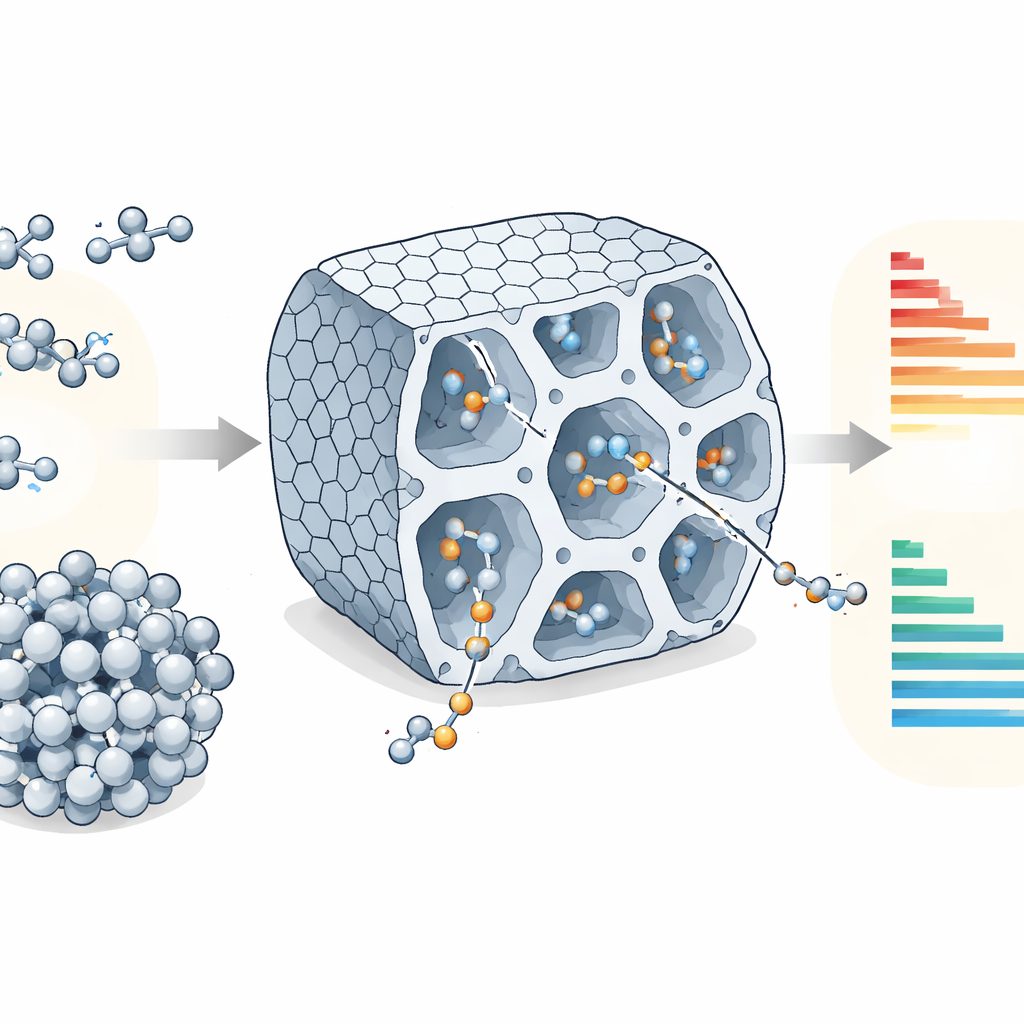
Chemists often focus on the metal atoms that drive reactions, but in porous materials the surrounding scaffold can be just as important. Here, the metal is nickel, anchored as isolated single atoms in a metal–organic framework called NU-1000. This framework is like a sponge made of orderly tunnels and tiny rooms: wide channels allow molecules to move, while smaller cavities host the nickel sites that link ethylene molecules into short chains known as oligomers. Previous studies showed that this material can make valuable products such as butenes and hexenes, but it was unclear how the interplay between intrinsic reaction chemistry and the material’s pore structure controls which products dominate.
Connecting Atomic Events to Reactor Behavior
The authors build a multiscale kinetic model that bridges processes from the atomic level up to the reactor scale. First, quantum mechanical calculations provide energy barriers for each elementary step of the reaction on the nickel site: ethylene attaches, inserts into a growing chain, and eventually lets go as a finished molecule. Second, large-scale molecular simulations describe how ethylene and its products adsorb inside the pores and how quickly they diffuse through the framework. These ingredients feed into a master-equation model that tracks the time-dependent concentrations of all species under realistic temperatures and pressures, for both continuous-flow reactors and closed batch reactors.
How Chains Grow, Stop, and Move
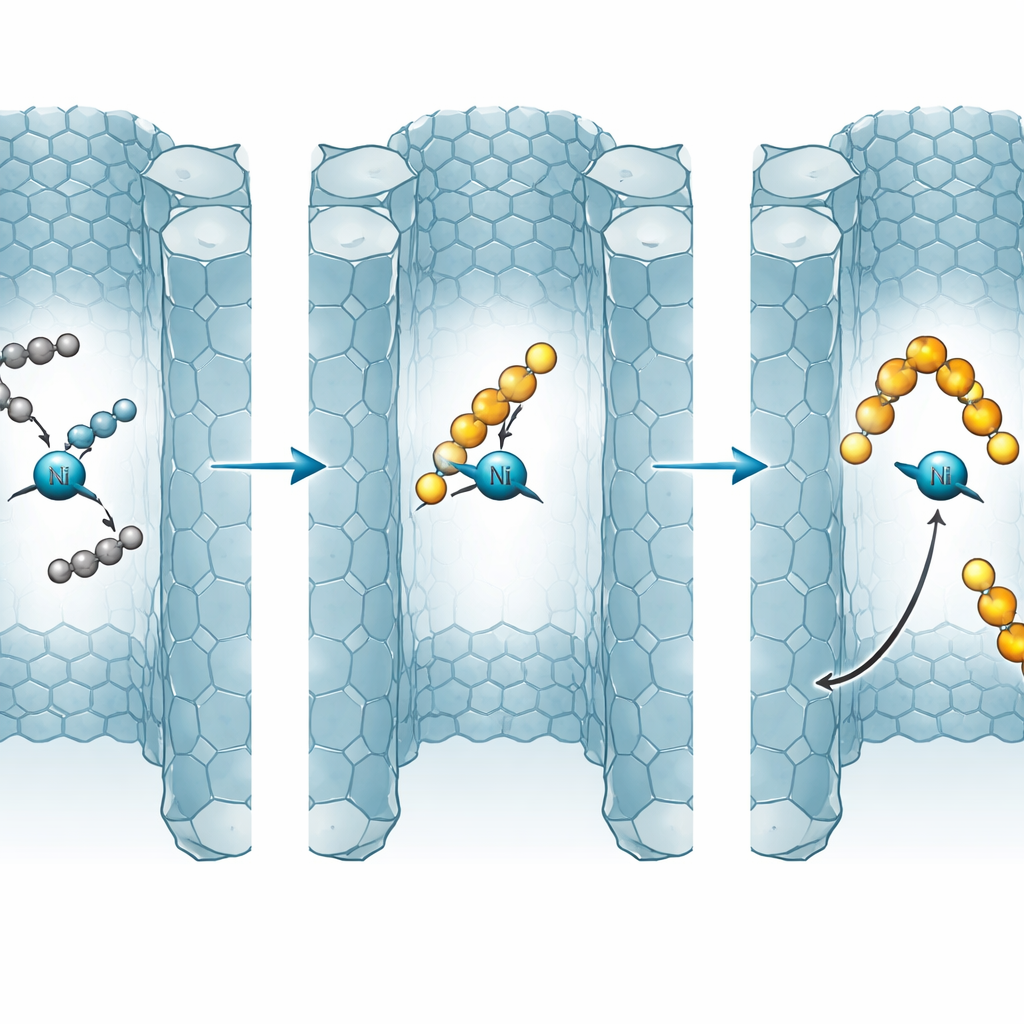
Inside NU-1000, ethylene adds to a nickel–carbon bond in repeated steps, lengthening the chain. At any stage, a competing route can “terminate” growth, releasing an olefin product and regenerating the nickel site. The model shows that the balance between growth and termination is highly sensitive to temperature, pressure, and how easily molecules can diffuse out of the pores. At moderate temperatures, the system favors formation of four-carbon chains, offering a window of high selectivity for butenes. As temperature rises further, both termination and reverse reactions speed up, and longer chains become more stable over time, pushing the distribution toward heavier products that can eventually resemble waxes or polymers.
When Diffusion Becomes a Hidden Lever
A key insight is that “residence time” in the pores acts like an extra control knob. In small particles with short diffusion paths, newly formed products escape quickly, effectively freezing them at short chain lengths and keeping the catalyst open. In larger particles or loosely packed beds, products linger, are more likely to re-adsorb, and can grow into longer chains before leaving. The model predicts that increasing the effective diffusion length or packing in more nickel sites narrows or even erases the selective window for butenes, leading to heavier oligomers and a higher risk of pore blocking and catalyst deactivation, especially in flow operation. Batch operation, where products are not swept away, naturally favors heavier products even more strongly.
Design Rules for Smarter Porous Catalysts
By uniting electronic structure, adsorption, diffusion, and reactor conditions into one framework, this work explains why similar nickel sites can behave very differently in different porous hosts and operating modes. For nickel-based NU-1000, the most promising recipe for selectively making short-chain olefins is a combination of small effective particle sizes, moderate nickel loading, and reactor setups that rapidly remove products. More broadly, the study demonstrates that controlling how molecules move through and compete for space in porous catalysts is just as crucial as tailoring the active metal site itself, offering a transferable strategy for designing next-generation materials that turn simple feedstocks into precisely targeted products.
Citation: Avdoshin, A., Matsokin, N.A., Huynh, TN. et al. Multiscale kinetic model of ethylene oligomerization in Ni-NU-1000 metal-organic framework. npj Comput Mater 12, 124 (2026). https://doi.org/10.1038/s41524-026-02044-7
Keywords: ethylene oligomerization, metal-organic frameworks, single-atom catalysis, diffusion and mass transport, catalyst selectivity modeling