Clear Sky Science · en
Long-term stability of moisture-capturing hydrogels by preventing metal-mediated degradation
Turning Air into Drinking Water
Billions of people live in regions where clean water is scarce, yet the air above them holds vast amounts of invisible moisture. New devices promise to pull this water from the sky using sponge-like materials called hydrogels. For this vision to become practical and affordable, those sponges must keep working day after day, year after year. This paper examines why some of the most promising hydrogels fail too quickly—and shows how a simple fix could help deliver ultra-low-cost water from air.

Why Air-Water Sponges Matter
The atmosphere contains an estimated 13,000 trillion liters of fresh water, enough in principle to supply more than a billion people regardless of geography. A class of technologies called sorption-based atmospheric water harvesting uses special materials to soak up moisture at night or in cool conditions and then release liquid water when warmed. Hydrogels loaded with salts stand out because they are cheap, easy to manufacture at scale, and can pull in large amounts of water even from relatively dry air. However, most studies have focused on how much water these materials can grab in a few dozen cycles, not on whether they remain safe and sturdy over the months and years needed for truly low-cost water.
Good Sponges and Bad Sponges
The researchers first asked a basic question: if you strip away all the complexities of a device, how stable are these hydrogels by themselves? They compared two widely used formulations—one based on polyacrylamide (PAM) and another on polyvinyl alcohol (PVA)—each loaded with lithium chloride salt. Samples were stored in hot salt solution at 75 °C, a temperature chosen to speed up any breakdown and to mimic the heat used when releasing water. Over more than eight months, the PAM-based hydrogel softened only modestly and barely shrank, while continuing to absorb nearly the same amount of moisture. In contrast, the PVA version lost stiffness and volume within weeks, yellowed, and visibly shriveled. Thermal analysis confirmed that the PAM-based hydrogel remains stable well beyond typical operating temperatures, marking it as an intrinsically durable choice for long-term water harvesting.
When Metals Turn Helpful Sponges Harmful
Real devices do not use bare hydrogels; they mount them on metal parts that help move heat in and out. The team discovered that this common design choice can secretly destroy even the more durable PAM hydrogels. When a PAM–salt gel was placed on copper—one of the most popular metals for heat spreaders—it discolored and developed tears within months. In hot salt solution with copper or its oxides present, the same hydrogel that had been stable for eight months turned into a runny liquid in just two to three weeks. Measurements showed that copper ions leached into the surrounding solution, and the gels took on a bluish tint, a classic sign of dissolved copper. In contrast, when the hydrogels contacted iron, iron oxides, or aluminum oxide under the same conditions, they largely kept their shape and strength, and far fewer metal ions were detected.
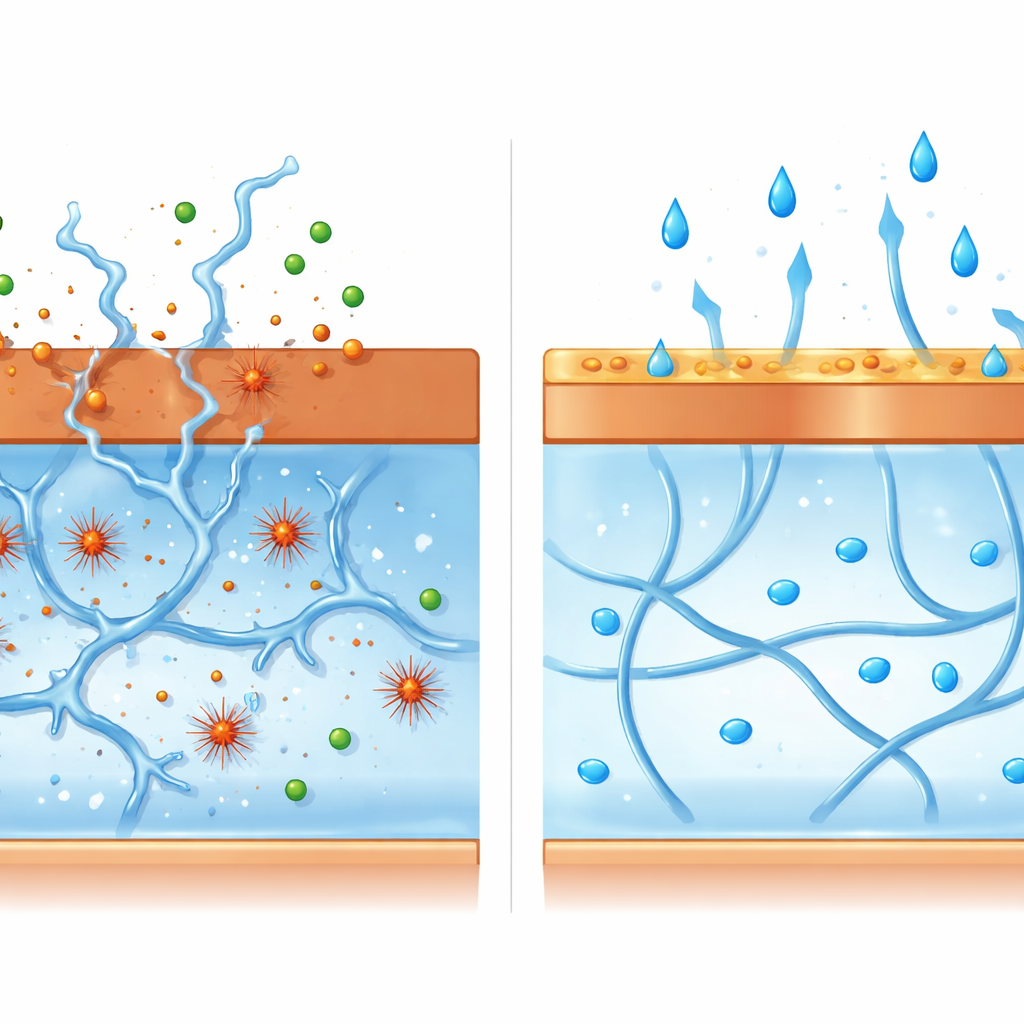
Unseen Chemical Attack and a Simple Shield
To explain these changes, the authors propose a step-by-step degradation pathway. First, copper at the surface slowly corrodes, releasing charged copper species into the salty water pooled within the hydrogel. Next, those metal ions react with dissolved oxygen to generate highly reactive hydroxyl radicals—short-lived chemical sparks that readily chop long polymer chains into shorter pieces. As the network of strands inside the gel is cut apart, it can no longer support its own weight, and the once-solid material collapses. Supporting evidence includes the strong link between copper ion levels and damage, the reduced breakdown when radical scavengers are added, and separate tests showing that dissolved polyacrylamide solutions thin dramatically in the presence of copper. Crucially, this radical-based attack is far weaker with the more stable iron and aluminum oxides, which produce far fewer metal ions under the same conditions.
Keeping the Water Flowing and the Cost Low
Armed with this understanding, the team explored how to protect the hydrogels without redesigning entire devices. They coated copper heaters with a commercial anti-corrosion varnish before applying the PAM–salt gel. The thin coating acts like a transparent raincoat: it blocks copper ions from entering the gel while still allowing heat to flow and water to move in and out. In long-running tests, the protected hydrogel endured more than 190 absorption–desorption cycles over 96 days, steadily harvesting and releasing water equivalent to nearly 500 kilograms per square meter. A simple economic analysis suggests that extending hydrogel lifetimes from days to months can slash the cost of harvested water by more than tenfold, pushing it below one cent per liter—approaching the cost of municipal tap water and far below bottled water prices. By revealing how metals can quietly sabotage these air-water sponges, and by offering a low-cost fix, this work brings the dream of robust, widely accessible water-from-air devices closer to reality.
Citation: Díaz-Marín, C.D., Wilson, C.T., Song, W.J. et al. Long-term stability of moisture-capturing hydrogels by preventing metal-mediated degradation. Nat Commun 17, 3783 (2026). https://doi.org/10.1038/s41467-026-71987-8
Keywords: atmospheric water harvesting, hydrogels, copper corrosion, material durability, water scarcity