Clear Sky Science · en
Atomic-scale quantification of individual oxygen vacancies and structural evolution in valence change memristors
Why tiny gaps in oxides matter for future memory
Modern gadgets depend on memory chips that are fast, dense, and energy-efficient. A leading candidate for next-generation memory is the "memristor," a tiny device whose resistance can be switched on and off like a light dimmer. Many memristors rely on the motion of missing oxygen atoms—called vacancies—inside metal oxides, but until now these vacancies have been nearly impossible to see one by one. This study uses advanced electron microscopes and quantum calculations to watch individual oxygen vacancies move and rearrange inside a promising oxide, revealing how they can make a device either reliable and reversible or unstable and doomed to fail.
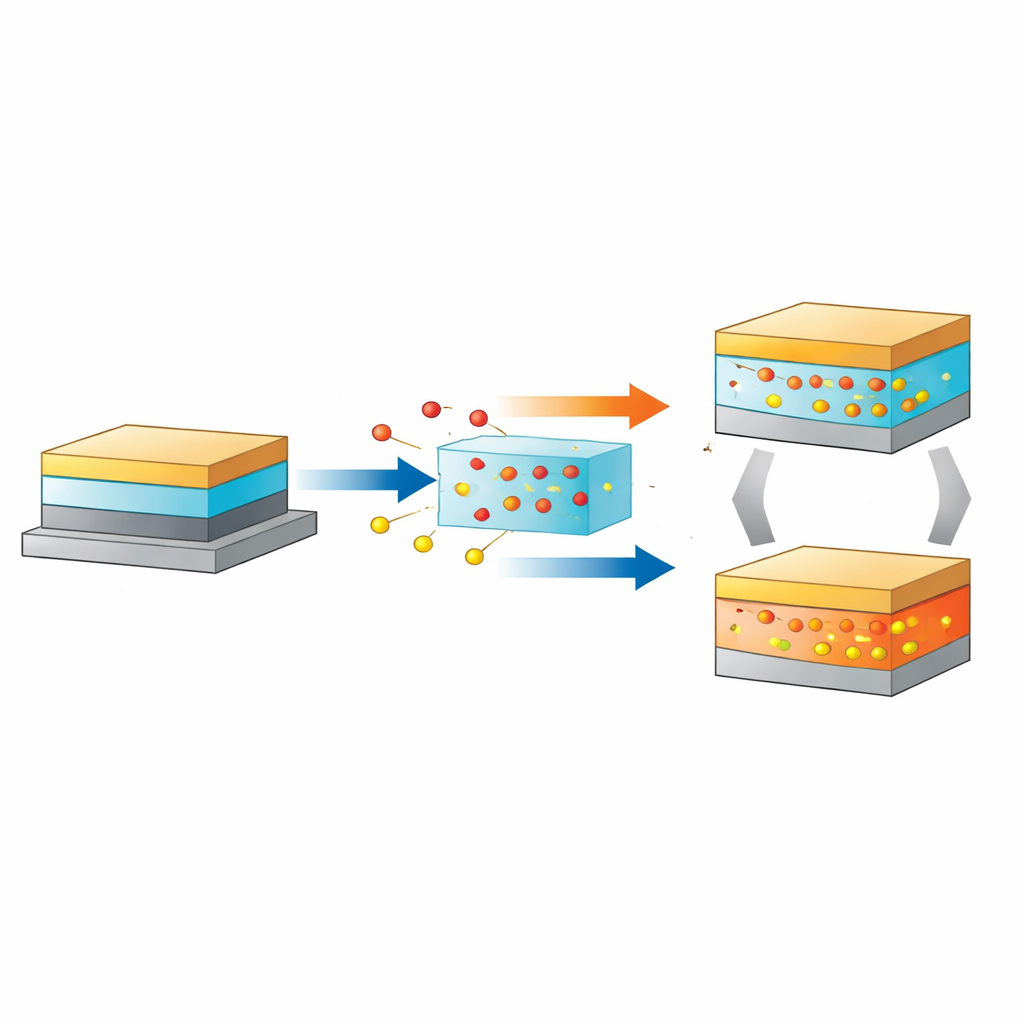
How a new kind of memory device works
The researchers focus on a memristor built from a layered oxide called strontium niobate (SrNbO3.4), sandwiched between a conducting substrate and a metal top contact. When a voltage is applied, oxygen ions are pushed out of or pulled back into the oxide, changing how easily electrons can flow. In the "SET" step, oxygen leaves parts of the crystal, creating vacancies that lower the resistance. In the "RESET" step, reversing the voltage draws oxygen back, restoring a high-resistance state. Ideally this back-and-forth motion would be perfectly repeatable, giving a long-lived memory. But in practice the lowest-resistance state tends to drift and weaken after programming, and repeated cycling eventually damages the device. Understanding exactly where the oxygen goes, and how much the crystal can tolerate, is the core challenge this work addresses.
Seeing individual missing atoms
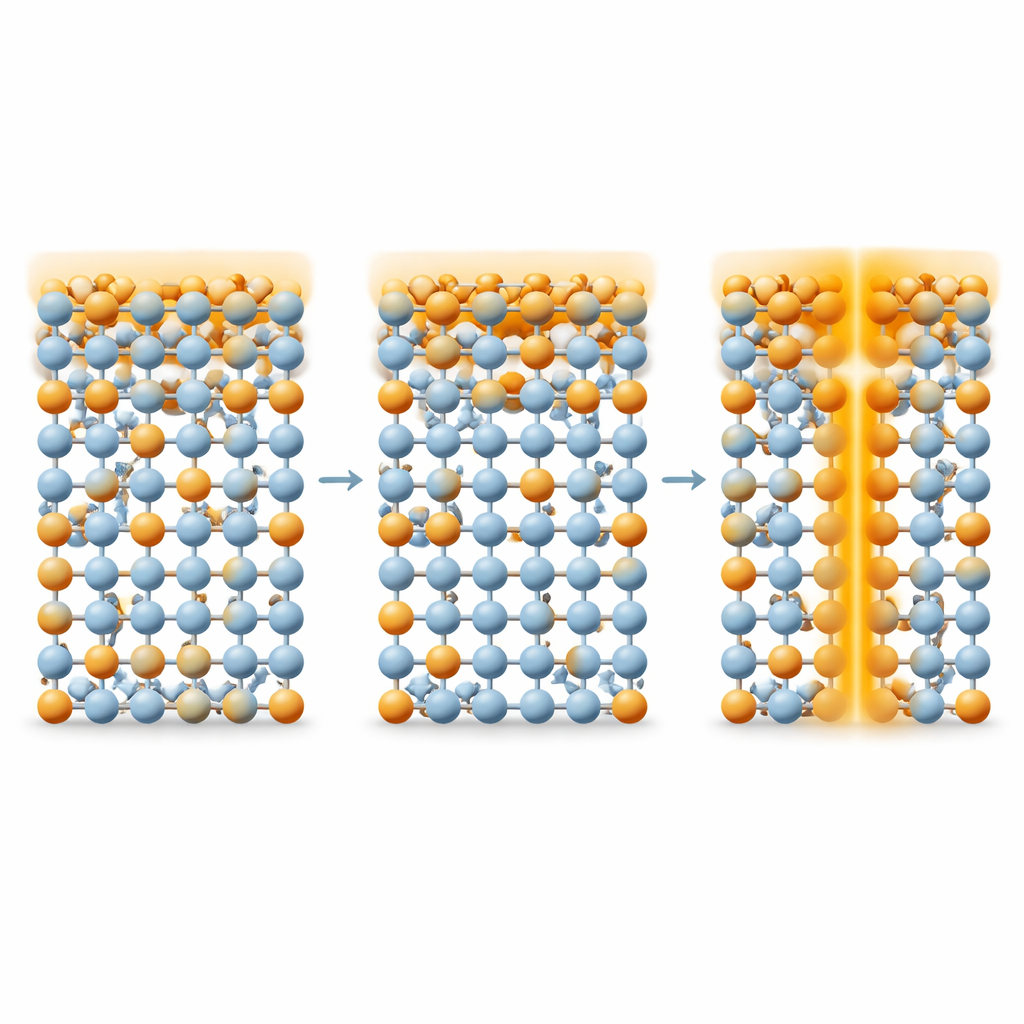
To tackle this problem, the team combines in-situ scanning transmission electron microscopy with detailed computer simulations. They record atomic-resolution images of the oxide while they apply voltage, and monitor how columns of heavier atoms such as strontium shift when nearby oxygen atoms disappear. By calibrating these shifts using quantum mechanical calculations, they show that the sideways displacement of a strontium column increases in discrete steps as one, two, or three oxygen atoms are removed from each structural unit. In effect, the crystal itself becomes a built-in gauge for counting vacancies with atomic precision. Using this method, they map where vacancies appear in real devices during switching and how the surrounding lattice stretches and strains as vacancies accumulate.
When order turns into damage
The maps reveal two very different regimes of behavior. When fewer than three oxygen vacancies form per structural unit, the crystal keeps its original orthorhombic framework, and the vacancies spread uniformly throughout the oxide, like a well-mixed solid solution. In this regime, resistance can be switched repeatedly because oxygen can be added and removed without leaving a lasting scar. However, once the vacancy count exceeds this threshold, vacancies begin to cluster near the interface with the metal electrode, creating a highly distorted, defective region. This damaged zone acts as an easy highway for oxygen ions to rush back into the oxide after SET, making the low-resistance state unstable. Under even higher voltage, oxygen loss becomes so severe that parts of the crystal transform into a different, more metallic cubic phase. If this new phase grows into a continuous filament spanning the film, the device locks into a permanently conducting state and fails.
A simple layer that makes a big difference
Armed with this atomic-scale picture, the authors test a practical fix. Instead of letting the metal contact act as a one-way sink for oxygen, they insert a thin amorphous layer of SrNbO3 between the crystalline oxide and the top electrode. This disordered layer serves as a reversible oxygen reservoir: it can temporarily store oxygen ions driven out of the active crystal during SET and return them during RESET. Electrical tests show that devices with this reservoir maintain a stable gap between high- and low-resistance states over many cycles, and microscopy confirms that the oxide mainly operates in the safe, solid-solution regime rather than slipping into the defective or fully metallic phases. In other words, controlling where excess oxygen goes is just as important as controlling how many vacancies are created.
What this means for future electronics
In conclusion, the study pinpoints a clear atomic-scale boundary between reversible switching and irreversible damage in oxide-based memristors: keep oxygen vacancies below about three per structural unit, and the device can cycle reliably; push beyond that, and defective structures or metallic filaments form that undermine performance or cause permanent failure. By directly counting individual vacancies and linking them to crystal distortions and electrical behavior, the work offers a blueprint for designing more robust memory devices—not only in strontium niobate, but in a wide range of oxides where anion vacancies quietly govern how electrons flow.
Citation: Wang, Z., Lin, W., Li, Y. et al. Atomic-scale quantification of individual oxygen vacancies and structural evolution in valence change memristors. Nat Commun 17, 3588 (2026). https://doi.org/10.1038/s41467-026-71912-z
Keywords: memristor, oxygen vacancies, oxide electronics, resistive switching, strontium niobate