Clear Sky Science · en
PsAvh109 suppresses SA-triggered immunity by mimicking TPL function to disrupt mediator complex assembly
How a Crop Killer Outsmarts Plant Defenses
Plants can’t run from disease, so they rely on a finely tuned immune system written into their DNA. This study reveals how a major soybean pathogen listens in on a plant’s own alarm signal and then strikes at the heart of its genetic control center, shutting down key defense genes at the very moment they are supposed to switch on. Understanding this molecular ambush offers new clues for breeding or engineering crops that can stay one step ahead of devastating diseases.
The Plant Alarm Signal and Its Trade-Offs
When microbes attack, plants quickly reprogram thousands of genes, shifting from growth to defense. A central alarm molecule, salicylic acid (SA), surges in tissues under attack and helps activate so-called pathogenesis-related genes that help stop biotrophic pathogens, such as many fungi and oomycetes. But this powerful response comes with costs: resources diverted to defense cannot be used for growth. To keep this balance, plants rely on flexible genetic switches that can rapidly turn defense genes off or on in response to changing signals.
The Cell’s “Mixer Board” for Immune Genes
At the core of this switching system lies the Mediator complex, a multi-protein hub that connects DNA-bound regulators to the enzyme that reads genes. Two of its components, known as MED21 and MED6, form a critical contact point: when they connect, they help assemble the machinery needed to activate SA-responsive defense genes. Plants also use a built-in brake protein called TOPLESS (TPL), which normally binds the same region of MED21 and blocks MED6 from docking. Under calm conditions this keeps immunity in check so that growth can proceed.
A Pathogen Effector That Imitates the Plant’s Own Brake
The soybean pathogen Phytophthora sojae produces a nuclear protein called PsAvh109 that turns out to be a master saboteur of this system. The researchers found that host-derived SA strongly boosts expression of PsAvh109 in the pathogen, both in culture and during real infections. Once delivered into plant cells, PsAvh109 moves into the nucleus and binds MED21 at exactly the same small N-terminal region that normally accommodates MED6 and TPL. By occupying this site, PsAvh109 blocks MED21–MED6 pairing and prevents proper assembly of the transcriptional machinery on SA-responsive promoters, including several PR1 defense genes. Plants infected with a PsAvh109-deficient pathogen show much stronger early activation of these genes and reduced disease, while versions of PsAvh109 forced out of the nucleus lose their ability to enhance infection.
Hijacking the Timing of the Plant’s Own Signals
The study uncovers a striking twist: the same SA burst that should free the plant’s own brake also triggers the pathogen’s counterattack. In healthy conditions, TPL sits on MED21 and dampens defense-gene activity. When SA rises during infection, TPL’s grip on MED21 is loosened, allowing MED6 to dock and turn on immune genes. But PsAvh109, which binds MED21 even more tightly than either MED6 or TPL, is induced by SA inside the pathogen. It then arrives in the plant nucleus just as TPL is being dislodged, quickly re-occupies MED21, and locks the complex in a repressed state despite the presence of SA. Homologous effectors from related Phytophthora species show similar SA inducibility, suggesting this “sense and respond” strategy is broadly conserved.
Implications for Protecting Crops
By mapping this molecular tug-of-war, the authors reveal a new way pathogens can exploit host signaling rhythms: rather than simply blocking recognition, they time effector release to coincide with immune activation and then directly rewire gene control. For non-specialists, the key message is that disease resistance is not only about having the right defense genes but also about who controls the switches that operate them. Targeting the PsAvh109–MED21 interaction, strengthening MED21–MED6 binding, or altering the MED21 contact surface could offer new avenues to breed soybeans and other crops that maintain strong SA-triggered immunity even in the face of clever pathogens.
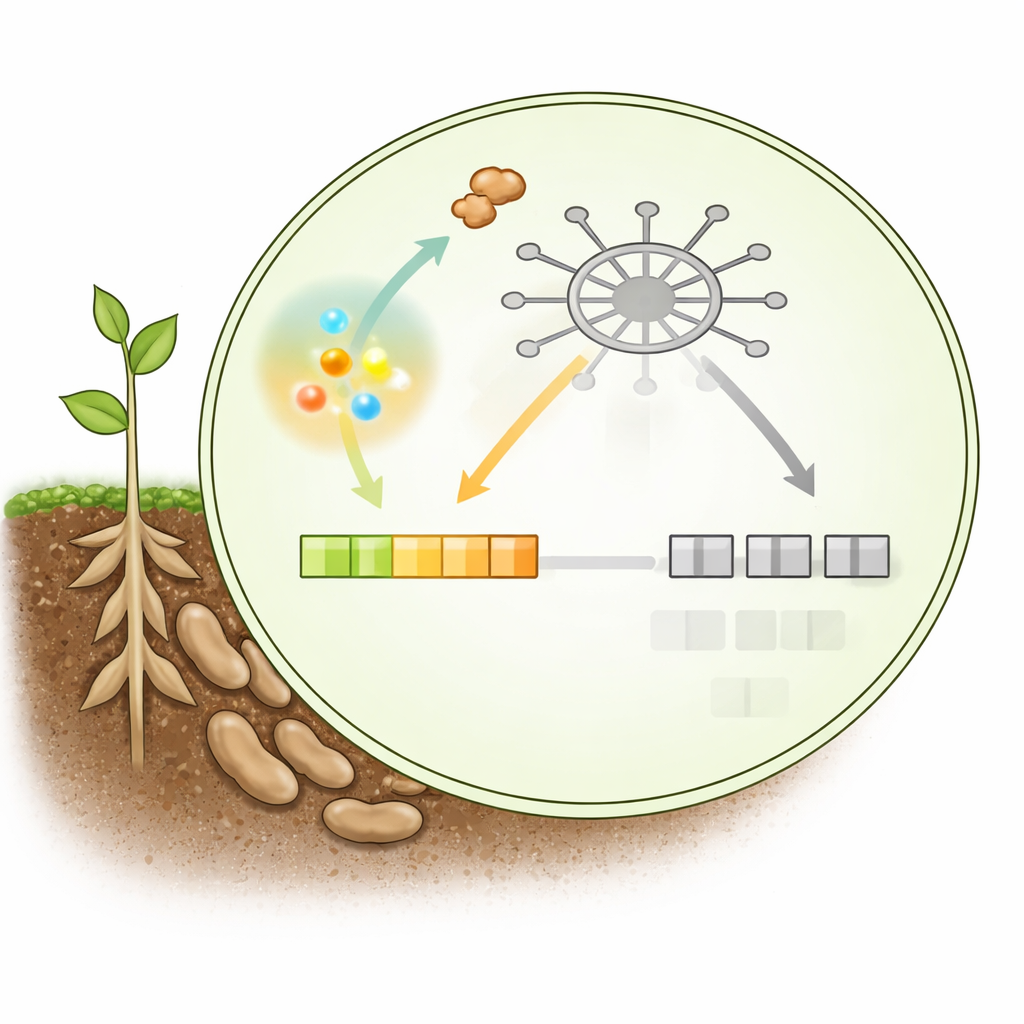
A Visual Snapshot of the Molecular Ambush
In broad strokes, this work shows a three-way contest at a single docking site inside the plant nucleus: growth-favoring repressors, defense-activating partners, and a pathogen effector all compete for control. When the plant is at rest, the repressor holds this spot and keeps immune genes low. When SA spikes, the plant briefly gains the upper hand, displacing the repressor and allowing activation partners to switch on defense genes. But the pathogen, sensing this SA surge, rapidly deploys PsAvh109, which snaps onto the same site with higher affinity and forces those genes back into silence. 
Citation: Tan, X., Sun, Y., Qi, Z. et al. PsAvh109 suppresses SA-triggered immunity by mimicking TPL function to disrupt mediator complex assembly. Nat Commun 17, 3486 (2026). https://doi.org/10.1038/s41467-026-71909-8
Keywords: plant immunity, salicylic acid signaling, Phytophthora sojae, transcriptional repression, Mediator complex