Clear Sky Science · en
The vault associates with membranes in situ
Mysterious Cell Cages Get a New Role
Inside our cells sit countless tiny machines, many of which we understand well. One of the biggest, called the vault, has puzzled biologists for decades. It looks like a hollow barrel and is surprisingly common in many organisms, including humans, yet its job has remained unclear. This study peeks at vaults directly inside living cells and uncovers two surprising behaviors: some vaults clasp onto internal membranes, and some carefully pack ribosomes, the cell’s protein factories, inside their hollow interior.
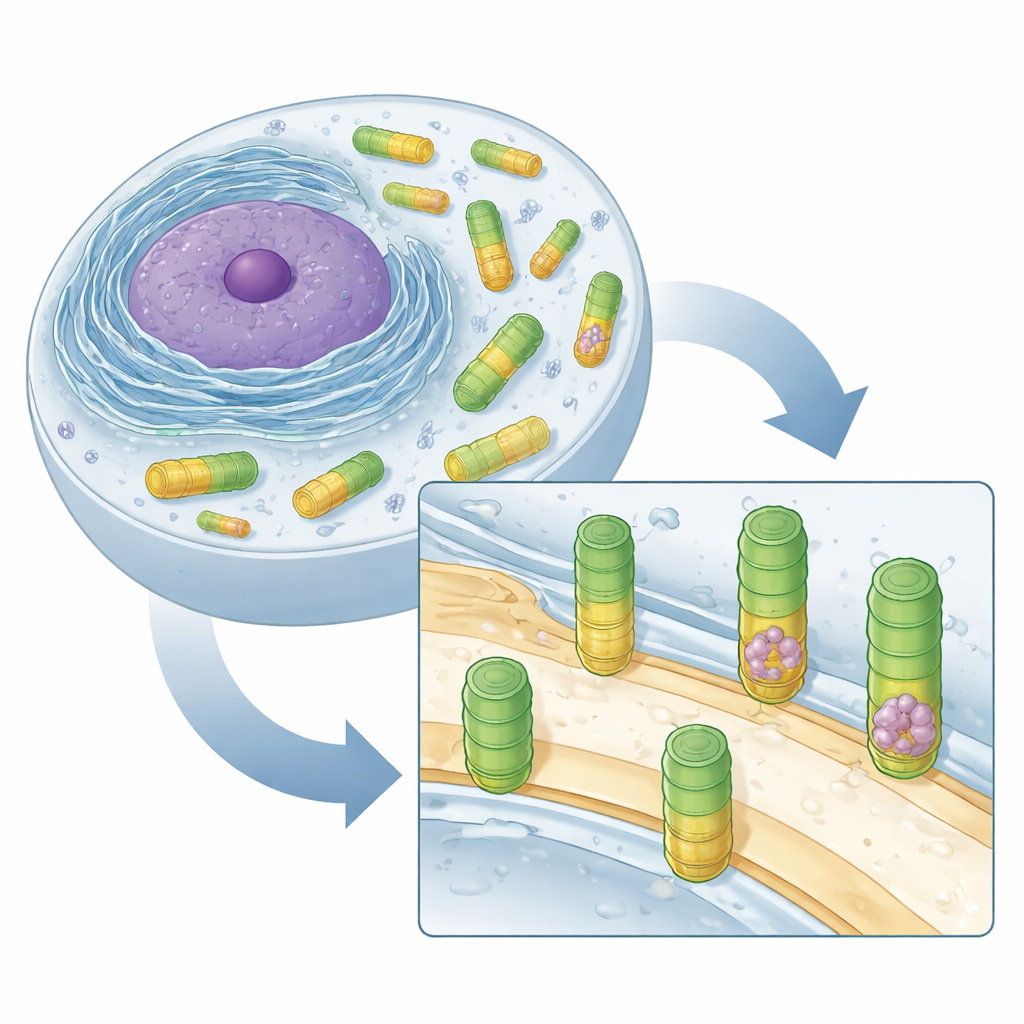
Hidden Barrels in the Cell Interior
The researchers focused on amoeba-like cells of Dictyostelium discoideum, a well-established model organism whose vaults closely resemble those found in animals. Using cryo–electron tomography, a technique that freezes cells and images them in 3D at nanometer resolution, they scanned hundreds of regions around the nucleus. Automated pattern recognition and detailed averaging of many images showed vaults as elongated, football-shaped barrels in the cytosol, the fluid that fills the cell. These barrels matched in size and shape to vaults previously described in rats and humans, suggesting that the structure of vaults has been conserved over evolution, hinting at an important but still unknown function.
A Surprise: Vaults Latching onto Membranes
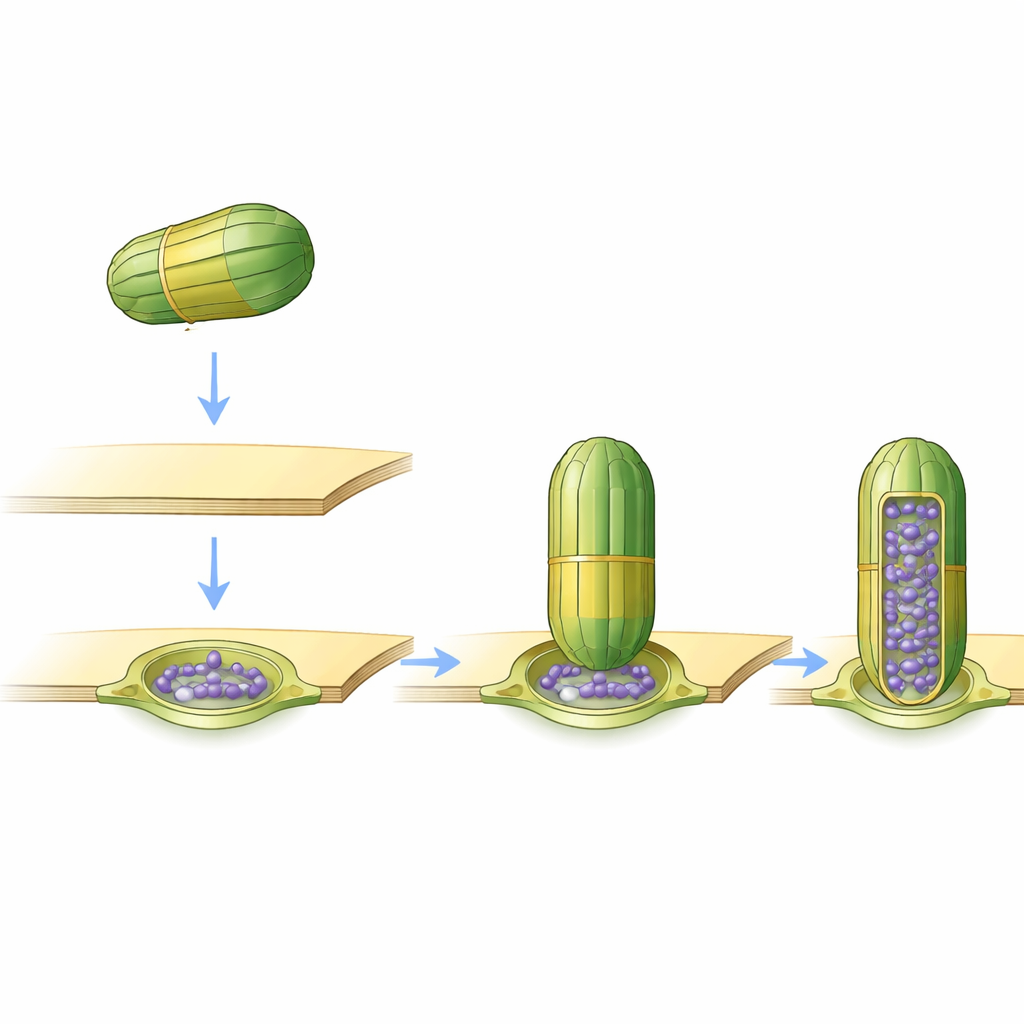
While most vaults were freely floating, a small but striking minority—about 1 to 2 percent—were found attached to membranes of the endoplasmic reticulum (ER) and the nuclear envelope, the two major membrane systems surrounding and neighboring the cell’s nucleus. These attached vaults were always seen standing upright, contacting the membrane at a specific “belt” along the barrel rather than at their ends. The upper two thirds of the barrel stuck out into the cytosol, while the lower third appeared to be rearranged or less visible in the images, suggesting that part of the vault may change shape when it binds to the membrane. This consistent docking height implies a carefully defined contact site rather than random sticking.
Softening and Shaping the Membrane
Membranes are not just passive backdrops; their thickness and curvature influence how proteins behave. The team measured these properties exactly where vaults attached. They found that the patch of membrane directly beneath the vault was thinner than the surrounding area, forming a circular zone of reduced thickness. Around the edge where the vault touched, the membrane formed a thicker “ring,” and its surface bent inward, as if slightly indented by the barrel. These features resemble special membrane microdomains formed by other, related proteins that are known to remodel membranes and help manage their quality. The observations raise the possibility that vaults may help recognize or generate unusual membrane patches for cellular housekeeping.
Vaults as Ribosome Carriers
Another surprise lay inside the vaults themselves. Many vaults contained ribosomes—large complexes that build proteins—tucked neatly within the barrel. By overlaying vault positions with previously mapped ribosomes in the same cells, the authors found 84 ribosomes fully enclosed by vaults. Nearly all of these were complete 80S ribosomes, the working form in eukaryotic cells, rather than partial subunits. In free-floating vaults, the ribosomes were packed in a highly ordered orientation, with the channel that releases new proteins facing the inner wall of the vault. In the few vaults that were both membrane-bound and ribosome-filled, some ribosomes sat in an orientation reminiscent of ribosomes engaged with the ER membrane, suggesting a potential connection to protein production near the ER.
Tracing a Network of Partners
To test whether these structural snapshots reflected real, recurring partnerships, the researchers used a proximity-labeling approach. They fused a vault protein to an enzyme that tags nearby proteins with biotin, then fished out all tagged partners and identified them by mass spectrometry. Hundreds of proteins were enriched near vaults, including many ribosomal proteins and several residents of the ER membrane or its interior space. Known vault-associated factors, such as certain enzymes and a protein linked to telomerase, also appeared. Together with the imaging data, this biochemical map strengthens the view that at least a subset of vaults spends time at the ER and in close contact with ribosomes.
Clues to a Long-Standing Cellular Mystery
Although the work does not yet reveal a single, definitive function for vaults, it provides important clues. Vaults can bind selectively to patches of altered membrane, rearrange part of their shell in the process, and encapsulate ribosomes in precise orientations. These behaviors suggest that vaults might help monitor or tidy up the protein-making machinery at the ER, or assist in clearing damaged components from specific membrane regions. Since vaults are highly conserved and have been linked to cancer drug resistance and stress responses, understanding this newly uncovered membrane-bound and ribosome-carrying behavior could eventually illuminate how cells cope with damage and change—and how vaults may be harnessed or targeted in medicine.
Citation: Geißler, K., Kreysing, J.P., Wang, Y. et al. The vault associates with membranes in situ. Nat Commun 17, 3659 (2026). https://doi.org/10.1038/s41467-026-71837-7
Keywords: vault particles, cell membranes, ribosomes, cryo-electron tomography, endoplasmic reticulum