Clear Sky Science · en
Carbon monoxide oxidation expands the known metabolic capacity in anaerobic methanotrophic consortia
Life in the Dark Ocean Floor
Far below the ocean’s surface, in chilly, oxygen-free mud, tiny partner microbes work together to keep methane—a powerful greenhouse gas—from reaching the water above. This study reveals that these deep-sea partnerships are even more versatile than scientists realized. When their usual fuel, methane, runs low, they can tap into another gas, carbon monoxide, to stay alive and keep carbon cycling under changing seafloor conditions.
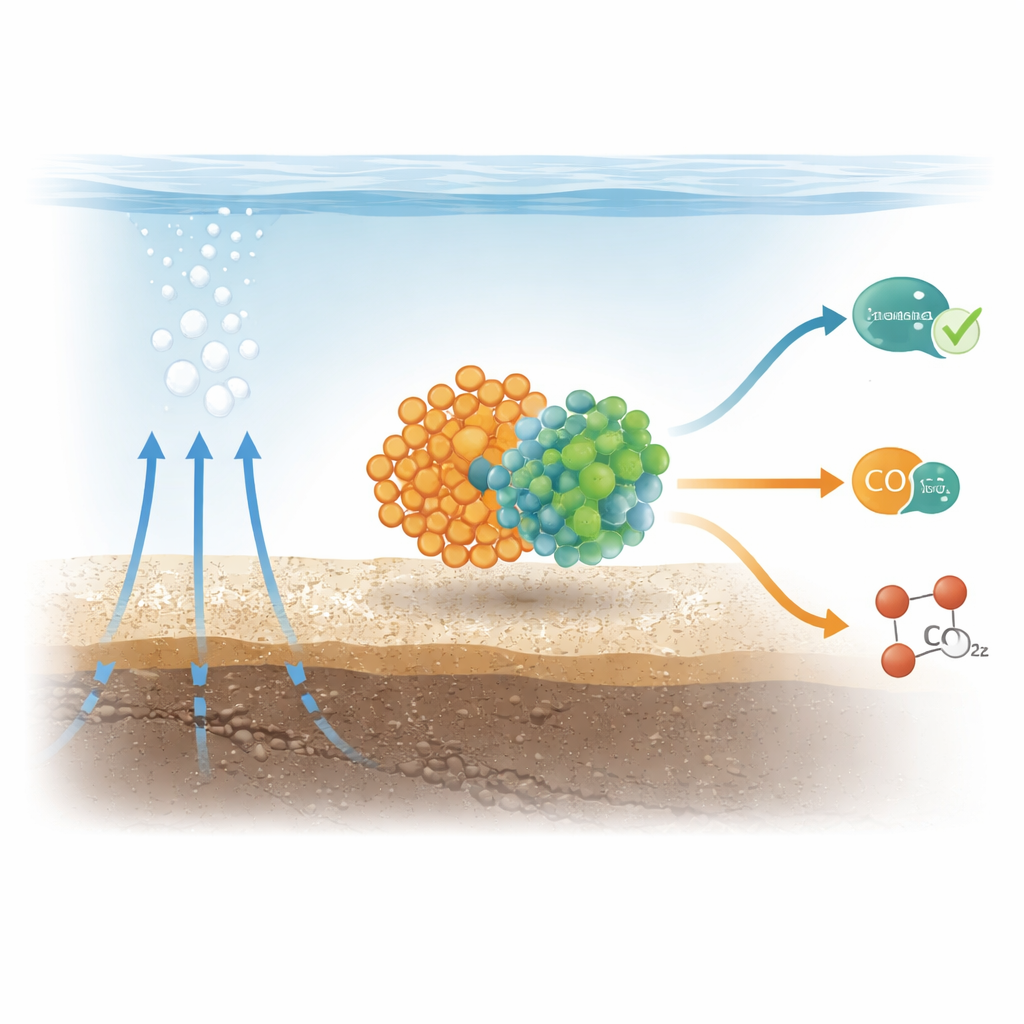
Hidden Partnerships Beneath the Seafloor
In many methane-rich seep sites on the seafloor, two kinds of microbes form tight-knit clusters: methane-eating archaea (called ANME-2b) and sulfate-reducing bacteria. Together, they normally perform a slow but crucial job: the archaea consume methane, pass the released energy and electrons to their bacterial partners, and the bacteria use these electrons to reduce sulfate from seawater. This teamwork locks up much of the methane before it escapes into the ocean and atmosphere. The new work asked whether these same partners could use a different fuel—carbon monoxide, a gas that can be produced in sediments by heat and by other microbes.
A New Fuel for Old Partners
The researchers set up sealed, oxygen-free “microcosms” using mud from a methane seep off Costa Rica and supplied them with carbon monoxide under controlled conditions. By tracking isotopic labels in carbon and sulfur, they showed that the consortia oxidized carbon monoxide and, at the same time, reduced sulfate to sulfide. When sulfate was absent, carbon monoxide oxidation instead drove the production of methane from carbon dioxide. The rate of methane production from carbon monoxide was modest—about one-ninth as fast as their usual methane-eating metabolism—but clearly measurable, showing that the same communities can both consume and generate methane depending on the available chemicals.
Zooming In on Single Cells
To find out which microbes were actually active on this new fuel, the team used a powerful combination of fluorescence imaging and nanoscale mass spectrometry. They fed the communities nitrogen labeled with a heavy isotope and then measured how much of this label ended up inside individual cells. Even in bottles where only carbon monoxide was provided as the energy source, many ANME-2b cells, and some of their bacterial partners, incorporated the labeled nitrogen—evidence that they were carrying out basic cellular maintenance. At higher carbon monoxide levels, however, activity declined, indicating that too much of this gas can be toxic, and that the consortia likely experience gentler, more favorable conditions in nature.
Reading the Microbial “Activity Log”
Beyond single-cell measurements, the scientists examined which genes were switched on when the communities used methane versus carbon monoxide. In the archaea, genes for the key methane-handling enzyme stayed highly active under both fuels, but genes tied to their main energy-conserving steps were dialed down when carbon monoxide was supplied. This pattern suggests that carbon monoxide use yields enough energy to keep cells running, but not enough to support strong growth. Meanwhile, the partner bacteria turned on genes associated with both direct electron sharing between cells and their own carbon monoxide-processing enzymes, hinting that they can receive energy from the archaea and also burn carbon monoxide themselves.
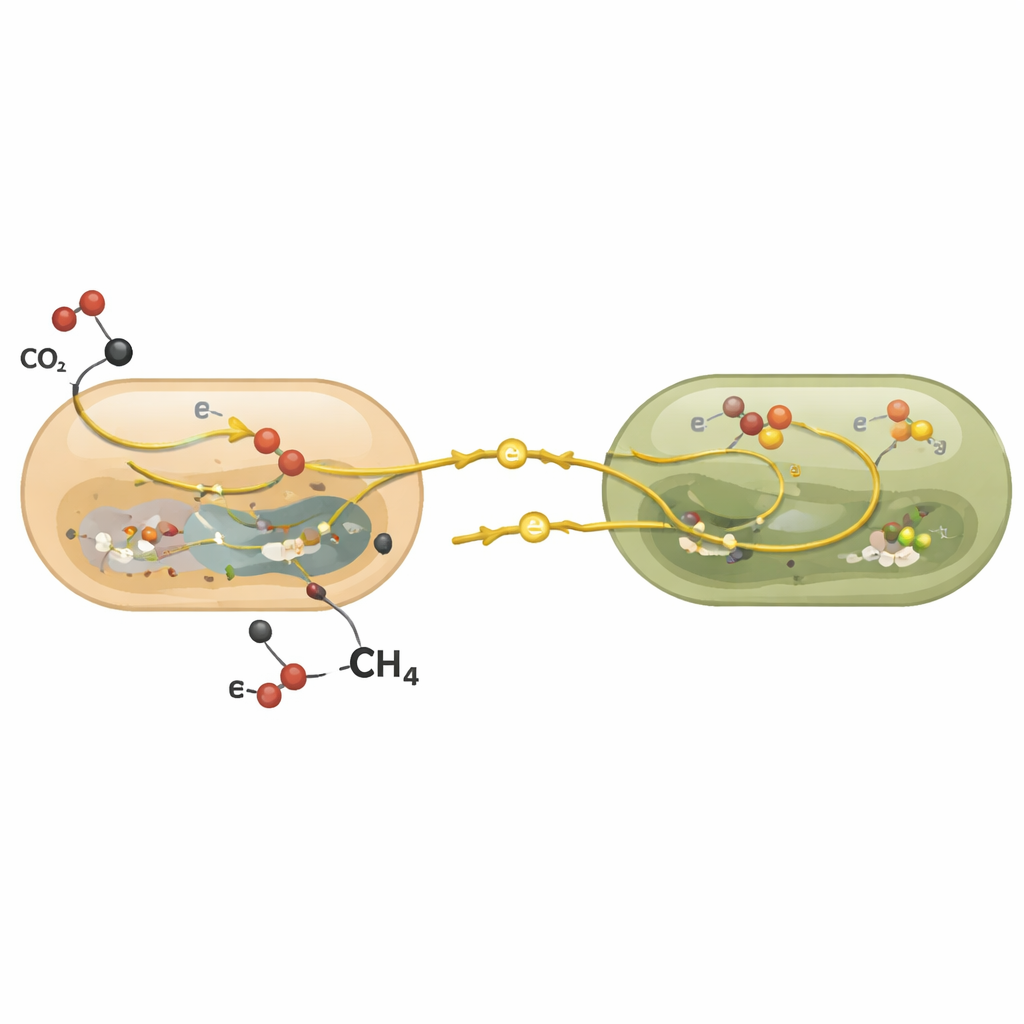
Why This Matters for Earth’s Carbon Balance
To a non-specialist, the key message is that deep-sea methane-eating communities are not one-trick specialists. They can switch to carbon monoxide as an alternate fuel, using it mainly to survive lean times and maintain their metabolism rather than to multiply rapidly. This flexibility helps these consortia persist when methane supplies fluctuate, allowing them to quickly resume their main job of destroying methane when it becomes available again. By uncovering this hidden survival strategy, the study refines our understanding of how carbon and sulfur move through the seafloor and how resilient Earth’s natural methane filter may be under changing environmental conditions.
Citation: Guo, Y., Utter, D.R., Murali, R. et al. Carbon monoxide oxidation expands the known metabolic capacity in anaerobic methanotrophic consortia. Nat Commun 17, 3461 (2026). https://doi.org/10.1038/s41467-026-71433-9
Keywords: methane seeps, carbon monoxide, anaerobic archaea, sulfate-reducing bacteria, deep-sea sediments