Clear Sky Science · en
Mechanofusion-derived cathode composite microstructures with scalable mixed conducting matrix coatings for solid state batteries
Why Better Batteries Need Better Cathodes
From electric cars to grid-scale storage, next-generation solid-state batteries promise more energy and improved safety compared with today’s lithium-ion cells. But to unlock that promise, engineers must not only invent new materials, they must also figure out how to arrange tiny particles inside the battery so that ions and electrons can move easily and the structure survives thousands of charge–discharge cycles. This study shows a practical way to "pre-build" those tiny structures in a scalable dry process, potentially smoothing the path from lab prototypes to industrial solid-state batteries.
Building a New Kind of Cathode Grain
In a solid-state battery, the cathode is a dense mix of three ingredients: an active material that stores energy, a solid electrolyte that carries lithium ions, and a conductive additive that carries electrons. Traditionally, these powders are simply mixed, leading to a somewhat random arrangement. The authors instead treat each cathode grain as a deliberately engineered "building block." They use single-crystal particles of a nickel-rich oxide as the energy-storing core. Around each core, they wrap a shell made from a soft halide solid electrolyte (Li3InCl6) and, in many cases, a fine carbon powder for conductivity. The result is a core–shell particle designed so that ions and electrons can reach almost every part of the active material.
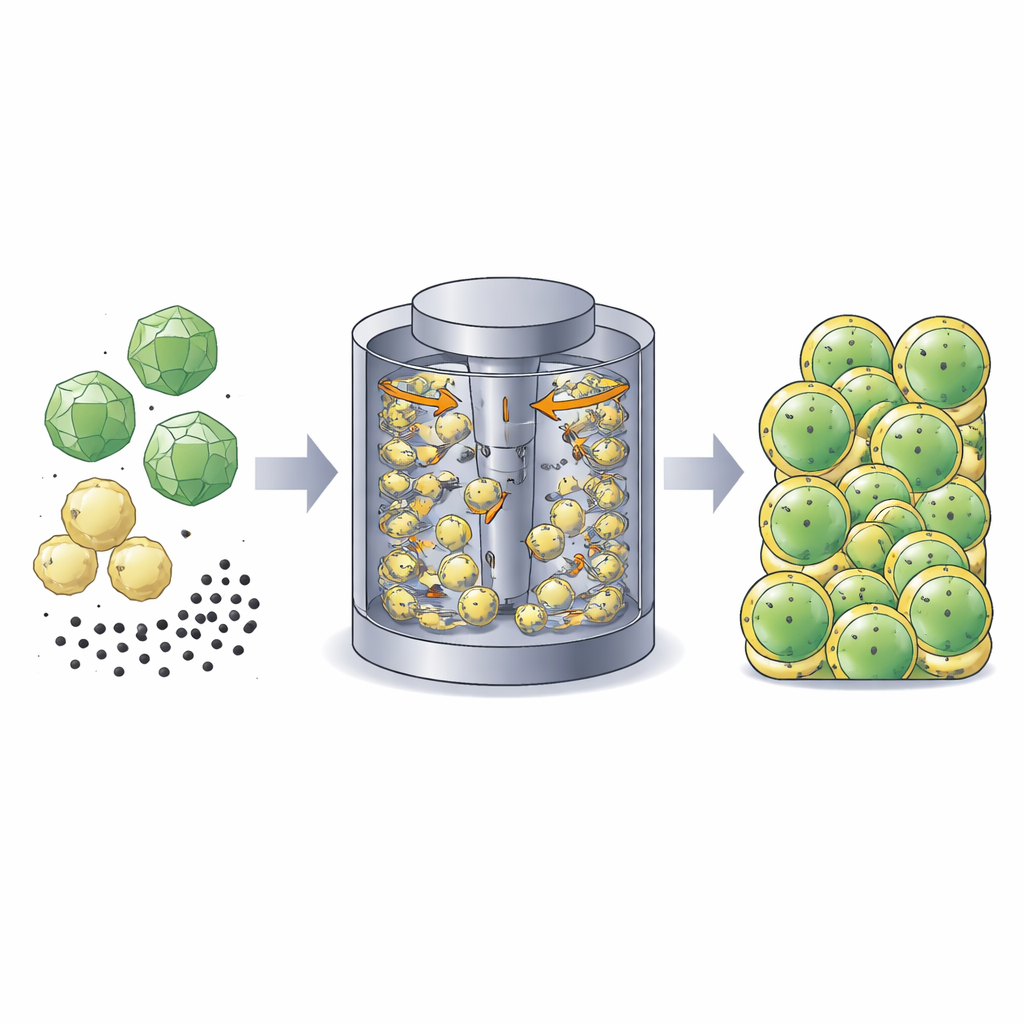
Spinning Powders into Coated Particles
To make these building blocks at scale, the team uses a high-intensity dry mixing technique called mechanofusion. Powders are fed into a compact mixer where a fast-spinning rotor drives them through a narrow gap, subjecting them to intense shear and collisions. Under these conditions, the soft electrolyte particles smear and deform onto the harder active-material crystals, while carbon particles become embedded in this outer layer. By tuning the mixer speed, processing time, and the relative amounts of each component, the researchers can create either ultra-thin, nanometer-scale coatings or thicker shells that form a continuous matrix around several particles. Advanced electron microscopy and surface-sensitive spectroscopy confirm that the coatings can fully cover the active particles without damaging their crystal structure.
Linking Mixer Settings to Microstructure
Because industrial mixers hold vast numbers of particles, the authors combine experiments with computer simulations that track how often and how strongly particles collide in the mixer. These simulations provide two key quantities: the intensity of each collision and how many collisions each particle experiences. They show that high collision intensity is especially important for forming smooth, continuous shells quickly. Lower intensities can eventually reach similar coverage, but only with much longer mixing times and less favorable coating shapes. Crucially, even at the highest tested intensities, the single-crystal cathode particles remain structurally intact when coated with the soft halide layer, suggesting that the process can be both energetic and gentle if the materials are chosen carefully.
Balancing Fast Transport with Mechanical Stability
The carbon-rich shell is meant to create a mixed pathway for both electrons and ions, but there is a trade-off. Adding more carbon improves the chances that every active particle is electronically connected, raising the share of the cathode that actually participates in storing energy. However, carbon dilutes the ion-conducting electrolyte and makes the shell more porous and prone to permanent deformation. Mechanical tests show that carbon-heavy shells behave more like soft, plastic foams that deform and do not fully recover, whereas carbon-lean shells act more elastically. In fast-charge tests, cathodes with too much carbon deliver high capacity only at low rates and rapidly lose performance at higher rates, likely because ion pathways become blocked and contacts are lost during cycling. An intermediate carbon content strikes the best compromise, giving both good utilization of the active material and robust performance at practical charge and discharge speeds.

What This Means for Future Solid-State Batteries
Overall, the study demonstrates that dry, high-intensity mixing can serve as a scalable route to carefully architected cathode particles for solid-state batteries. By treating each grain as a designed object—with a tough, energy-storing core and a soft, ion- and electron-conducting shell—the authors achieve stable cycling at realistic rates while using only industrially relevant processes and batch sizes. Their results highlight that battery performance depends not just on what materials are used, but on how they are mechanically processed into microstructures that balance conductivity and mechanical resilience. This bottom-up approach to cathode design could help bridge the gap between promising solid-state chemistries and practical, manufacturable devices.
Citation: Kissel, M., Frankenberg, F., Demuth, T. et al. Mechanofusion-derived cathode composite microstructures with scalable mixed conducting matrix coatings for solid state batteries. Nat Commun 17, 3215 (2026). https://doi.org/10.1038/s41467-026-71305-2
Keywords: solid-state batteries, cathode coatings, mechanofusion, particle microstructure, energy storage