Clear Sky Science · en
Experimental Sodalis infection eliminates ancient insect symbiont
Hidden partnerships inside tiny grain pests
Grain stored in silos or pantries is often under siege by beetles too small to notice. Inside these insects live even tinier partners—bacteria that quietly supply missing nutrients and help the beetles survive dry, harsh conditions. This study peeks into that hidden world and shows what happens when scientists deliberately introduce a new bacterium into a beetle that already relies on an ancient helpful microbe. The result is a rapid microbial takeover that highlights how fragile long‑standing alliances between animals and bacteria can be.
A long marriage between beetles and helpful microbes
Many insects depend on bacteria living inside their cells to make vitamins and amino acids they cannot get from their limited diets. In the sawtoothed grain beetle, a common pest of stored cereals, a long‑term bacterial partner called Shikimatogenerans resides in special organs known as bacteriomes. This symbiont produces a chemical precursor that the beetle converts into tyrosine, an amino acid crucial for hardening and darkening its protective outer shell. Over millions of years of close cooperation, such ancient symbionts typically shrink their genomes and lose flexibility, becoming tightly tied to their hosts—and potentially vulnerable to disruption.
Introducing a new bacterial player
To explore how a new microbe might displace an old one, the researchers injected female beetles with a lab‑grown bacterium, Sodalis praecaptivus, which is closely related to symbionts found in other insects. They tracked the bacteria using a fluorescent marker and DNA measurements. Within a week, Sodalis had spread throughout the beetle’s body, including blood‑like fluid, fatty tissues, the nervous system, and the reproductive organs. Crucially, it entered developing eggs and was passed efficiently from mothers to their offspring, generation after generation, without any help from the fathers. This rapid, reliable vertical transmission mimics a key step in the natural evolution of beneficial symbioses. 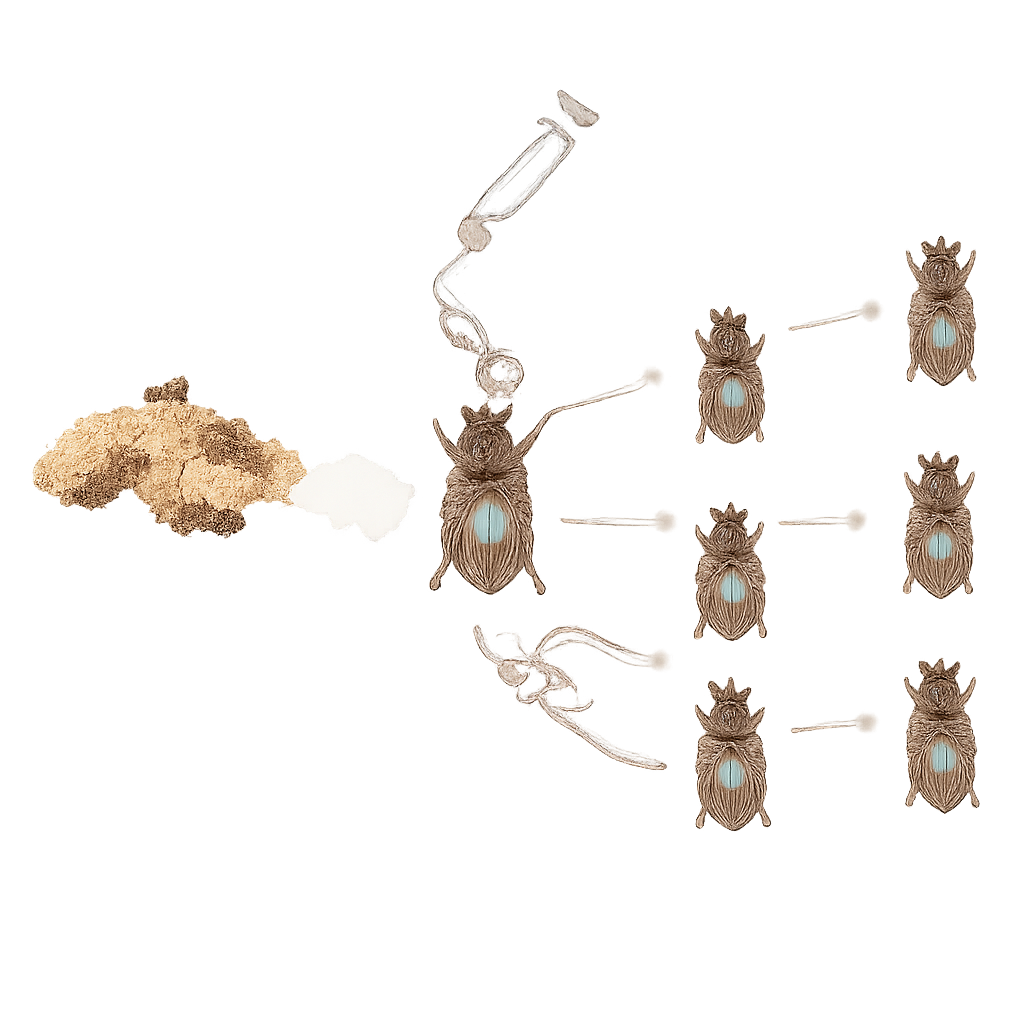
Costs for the beetle and a silent struggle inside
Despite this successful colonization, the new partnership came at a steep price for the beetles. Infected adults died much sooner and produced far fewer larvae than uninfected controls. Young adults carrying Sodalis had paler, less darkened shells, a sign that tyrosine supplies were strained, although shell thickness itself did not change. Inside, imaging and three‑dimensional scans revealed that Sodalis invaded the bacteriomes, sharing space with Shikimatogenerans. Where both bacteria co‑occurred, the native symbiont cells became enlarged and oddly shaped, suggesting stress or damage, even though their overall numbers initially stayed similar.
When the ancient partner disappears
Over just three beetle generations, this uneasy coexistence tipped into outright displacement. By the third generation, every surviving beetle that carried Sodalis had completely lost its original symbiont and often lacked visible bacteriomes altogether, while control beetles retained normal symbiont organs. Gene‑expression analyses of bacteriome tissue showed that the beetle’s immune system strongly reacted to Sodalis, turning on antimicrobial defenses and regulatory proteins. In contrast, Shikimatogenerans barely changed its activity, consistent with its streamlined, inflexible genome. Subtle shifts in both beetle and symbiont metabolism hinted that the invader was competing for key nutrients, including those needed to make tyrosine, further undermining the native partner. 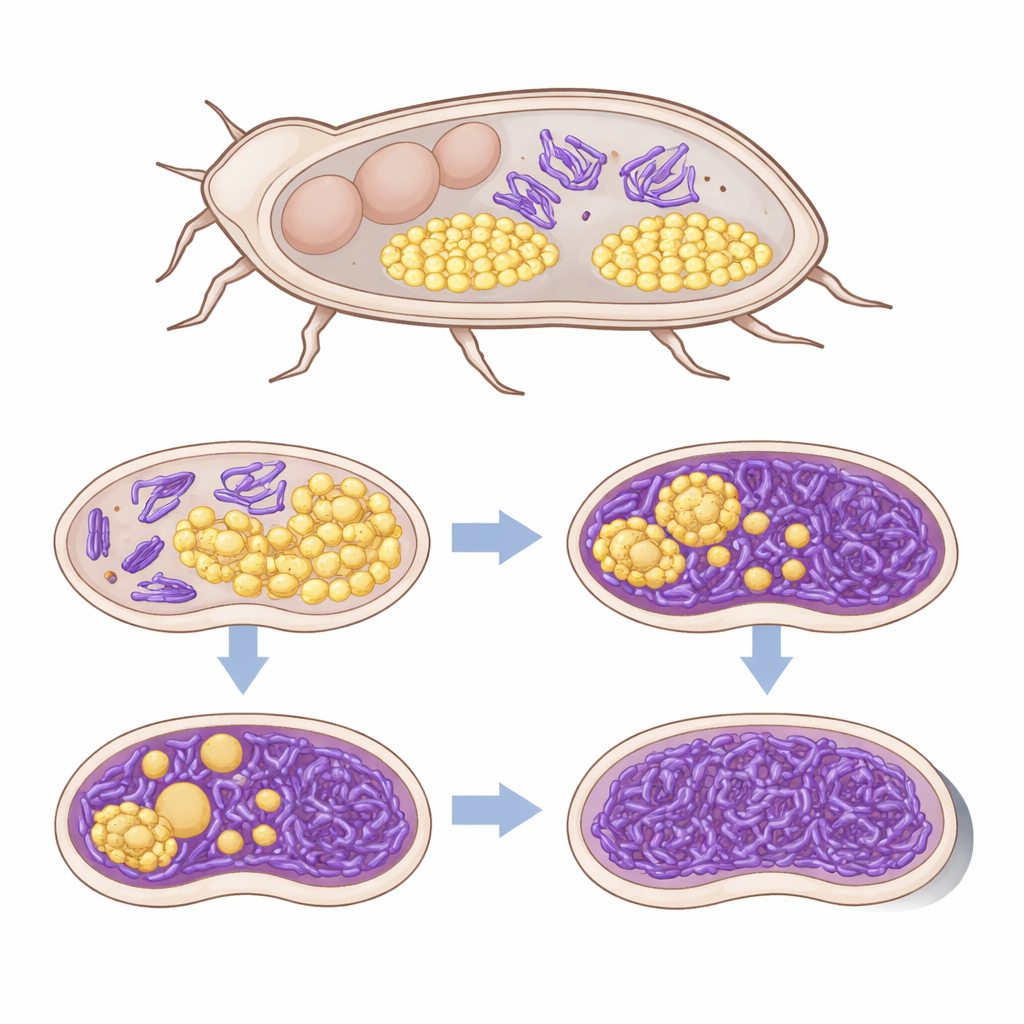
What this tells us about changing microbial alliances
By experimentally recreating an early stage of symbiont replacement, this work shows that a newly arrived bacterium can rapidly eliminate a long‑standing, beneficial partner, even in just a few generations. In this case, Sodalis already satisfies three of four conditions thought necessary for a full replacement: it establishes inside the host, is passed reliably from mother to offspring, and drives the loss of the ancestral symbiont. What it does not yet do is help the beetle—indeed, it harms it. However, because Sodalis is geneticly malleable and tightly linked to host reproduction, evolutionary pressure may eventually favor versions that leak useful nutrients or soften their harmful effects, turning a destructive infection into a new mutualism. The study thus offers a powerful, controllable model for watching, in real time, how animal–microbe partnerships can collapse and potentially be rebuilt with new partners.
Citation: Krüsemer, R., Carvalho, A.S.P., Keller, J. et al. Experimental Sodalis infection eliminates ancient insect symbiont. Nat Commun 17, 3153 (2026). https://doi.org/10.1038/s41467-026-71143-2
Keywords: insect symbiosis, endosymbiont replacement, grain beetle, host microbe interactions, Sodalis bacteria