Clear Sky Science · en
Comparative genome analysis provides a foundation for defining salvinorin A biosynthesis in Salvia divinorum
A hallucinogenic plant with medical promise
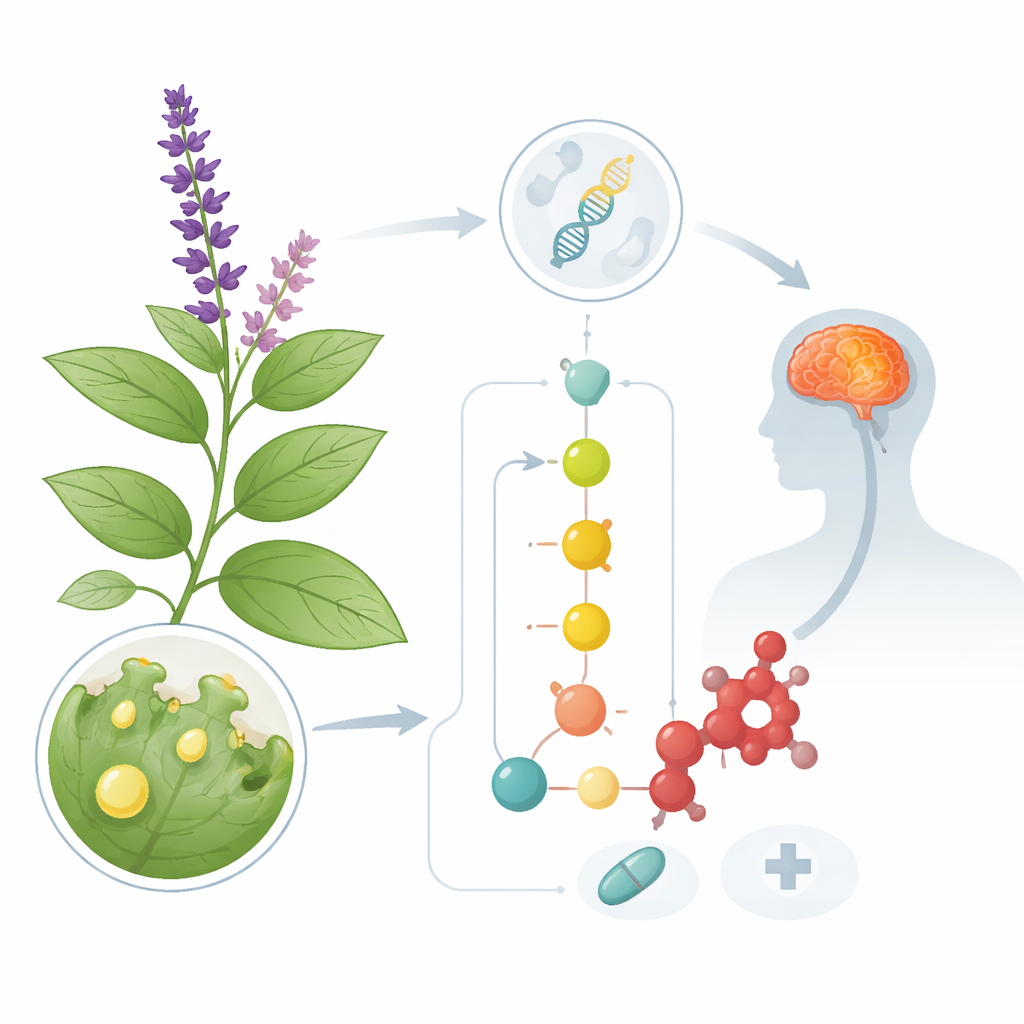
Salvia divinorum, sometimes called “diviner’s sage,” is famous for inducing brief but intense hallucinations when its leaves are consumed. Yet the same compound responsible for these experiences—salvinorin A—is now being explored as a blueprint for new medicines to treat pain, depression, and drug addiction. This study asks a deceptively simple question with big implications: how does the plant make salvinorin A, and how did that chemical pathway evolve? By answering it, scientists hope to recreate the compound, and related molecules, in a controlled way without relying on a difficult-to-grow plant.
From sacred herb to lab subject
Salvia divinorum grows naturally in the cloud forests of Oaxaca, Mexico, where it has long been used by Mazatec healers in ceremonial settings. Modern pharmacology has shown that salvinorin A targets a specific protein in the brain—the kappa opioid receptor—where it can dampen certain forms of pain and reduce the rewarding impact of addictive drugs. Unfortunately, the plant is rare, hard to cultivate, and yields only small amounts of the compound. Total chemical synthesis is possible, but too complicated and costly for large-scale use. To unlock salvinorin A as a practical medicine, researchers need a detailed map of the plant’s internal assembly line so that the pathway can be rebuilt in microbes or other crops.
Reading the plant’s instruction manual
The team generated a high-quality, chromosome-level genome for Salvia divinorum—a complete inventory of its DNA instructions. They then compared this genome to those of several relatives in the sage family, including culinary rosemary and chia, as well as closely allied ornamental and medicinal species. These comparisons revealed when different sage lineages split from each other over tens of millions of years and highlighted bursts of genome duplication, in which the entire set of genes was copied. Such duplication events are a common engine of innovation in plants, because extra gene copies are free to evolve new functions without disrupting old ones.
Building the chemical pathway step by step
Salvinorin A belongs to a family of molecules called furanoclerodane diterpenoids, which the plant produces in tiny factories on the leaf surface known as glandular trichomes. Earlier work had already identified the first stages of the pathway, which build a basic carbon framework from simple starting units. By overlaying gene activity data from trichomes onto the new genome, the authors could pinpoint additional genes that are switched on where salvinorin A is made. They focused on two major groups of enzymes: cytochrome P450 proteins, which decorate the carbon skeleton with oxygen atoms at precise positions, and methyltransferases, which attach small methyl groups.
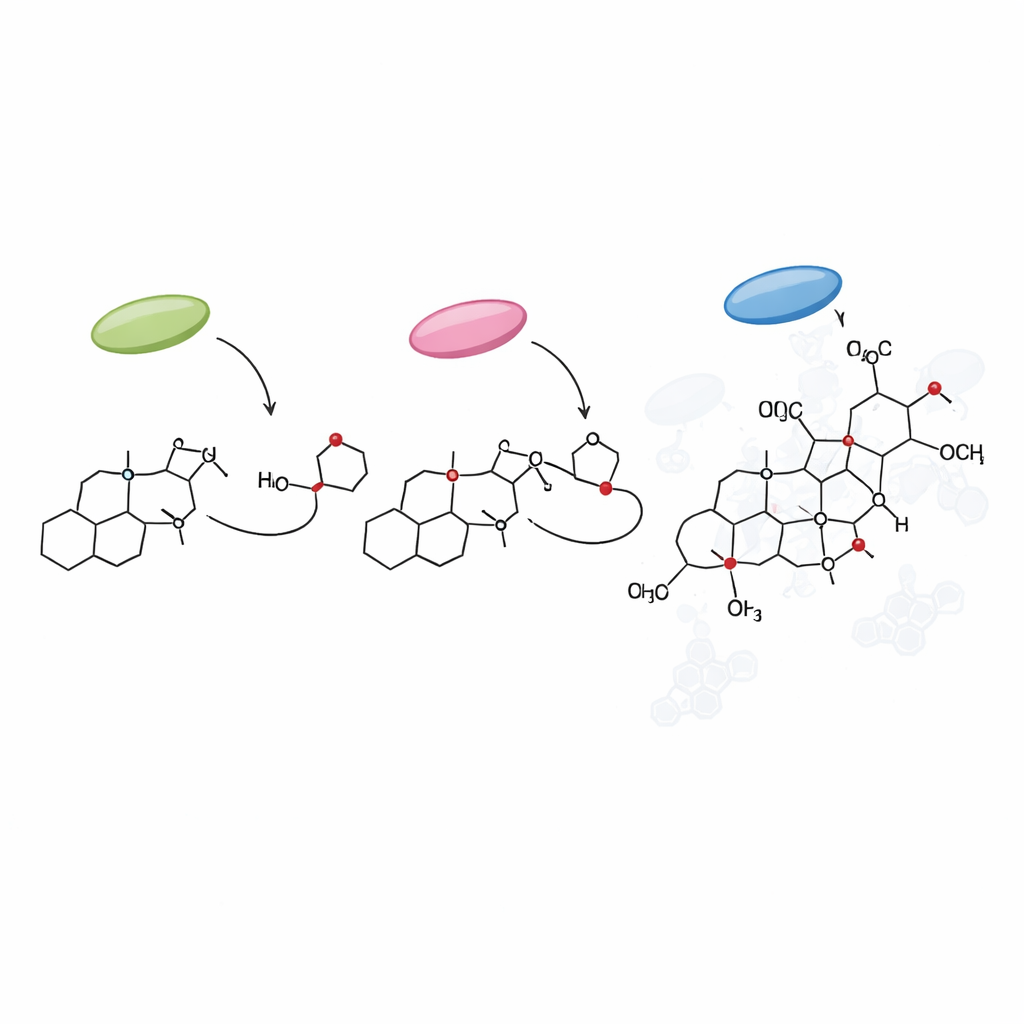
New enzymes that sculpt salvinorin’s core
Through a mixture of evolutionary detective work and practical experiments in tobacco leaves, the researchers uncovered several missing steps in the pathway. They showed that one P450 enzyme, dubbed annonene synthase, likely arose after a whole-genome duplication specific to New World sages and was repurposed from an older enzyme that worked on a different class of plant chemicals. A second cluster of P450 enzymes in the CYP728D family was found to carry out a series of fine-tuned oxidations, transforming an intermediate called hardwickiic acid into “divinatorins,” key stepping stones on the road to salvinorin A. Another enzyme, a SABATH-family methyltransferase, was confirmed to methylate a particular carboxyl group—a subtle chemical tweak that appears unique to Salvia divinorum and crucial for finishing one of the principal intermediates.
How evolution shaped a potent natural product
By tracing where these genes sit in the genome and how they relate to counterparts in other sage species, the study shows that salvinorin A biosynthesis did not appear all at once. Instead, it emerged gradually as duplicated genes were shuffled, locally copied again, and nudged into new roles, especially in leaf trichomes. Some relatives make related clerodane compounds but lack the full complement of specialized enzymes found in Salvia divinorum, underscoring how small, lineage-specific changes can yield strikingly different chemistries. Understanding this evolutionary tinkering provides a roadmap for discovering the final unknown steps and for rationally altering the pathway to create new, potentially therapeutic molecules.
From plant evolution to future medicines
The authors conclude that having a complete genome and a nearly finished pathway for salvinorin A lays the groundwork for producing this compound—and improved variants—without depending on wild or greenhouse-grown plants. In practical terms, it moves researchers closer to engineering yeast, bacteria, or other crops to make furanoclerodanes at scale. For non-specialists, the key message is that by reading and comparing plant genomes, scientists can both reconstruct how powerful natural products evolved and repurpose those same genetic instructions to develop next-generation treatments for pain, addiction, and mental illness.
Citation: Li, H., Sun, Y., Xu, W. et al. Comparative genome analysis provides a foundation for defining salvinorin A biosynthesis in Salvia divinorum. Nat Commun 17, 3414 (2026). https://doi.org/10.1038/s41467-026-70885-3
Keywords: Salvia divinorum, salvinorin A, plant specialized metabolism, comparative genomics, natural product drug discovery