Clear Sky Science · en
FGF1 orchestrates circadian hepatic triglyceride secretion
Why the liver’s daily fat rhythm matters
Fatty liver disease is becoming alarmingly common, yet the liver is not just a passive fat sponge. It constantly ships fat out into the bloodstream so other tissues can burn or store it. This study reveals that a natural liver-made protein called FGF1 acts like a timing signal, helping the liver decide when to send fat into circulation across the day and night. Understanding this built-in schedule could open new ways to ease fat buildup in the liver without shutting down other vital metabolic processes.
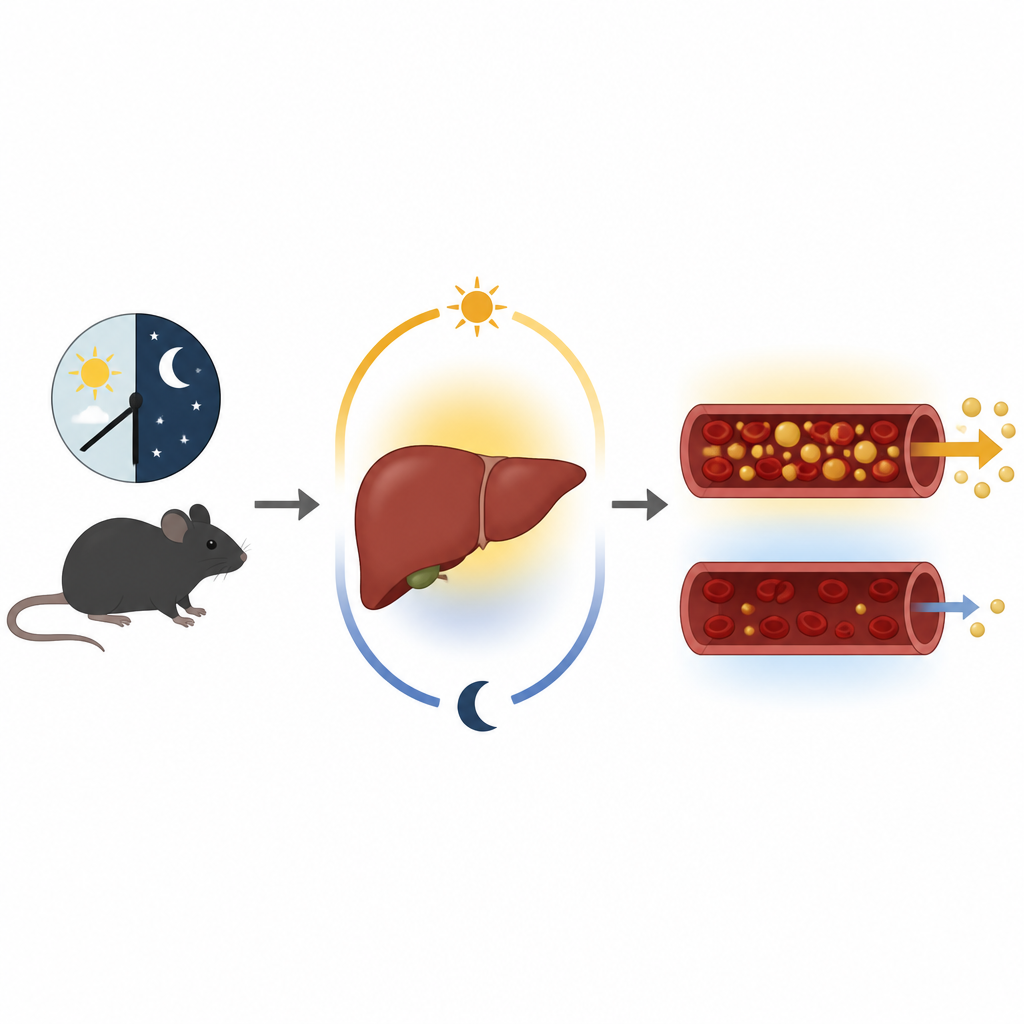
A growing problem in modern livers
Metabolic dysfunction-associated steatotic liver disease, or MASLD, covers a spectrum from simple fat buildup to inflammation, scarring, and even liver cancer. It is now one of the leading reasons people develop end-stage liver disease. In MASLD, the balance between making fat, burning it, and exporting it as tiny fat-rich particles becomes distorted. While drugs that help the liver burn more fat are emerging, many patients still do not respond well. That gap has pushed scientists to ask whether errors in the timing of fat export, not just the amount of fat made, might help drive disease.
The body clock and liver fat traffic
Like sleep and hunger, liver metabolism follows a daily rhythm. In healthy animals, liver fat content rises during the resting phase, then falls when the animal is active and needs energy, as fat is packaged into particles released into the blood. The authors focused on FGF1, a protein known to affect blood sugar and fat storage in other tissues. They discovered that one major form of FGF1 in the liver is turned on and off in a circadian pattern, peaking during the active phase. This makes FGF1 an “output” of the liver’s internal clock, positioned to translate time of day into a pattern of fat release.
What happens when the FGF1 signal is lost
To test how important this timing signal is, the team created mice that lacked FGF1 only in liver cells. On a normal diet, these animals looked outwardly healthy, but closer inspection at different times of day told a different story. Around the time when FGF1 normally peaks, their livers held more triglycerides while blood levels of triglycerides were lower, showing that export was impaired just when it should be strongest. When challenged with a rich, high-fat, high-cholesterol diet, these mice developed worse fatty liver, more liver inflammation, and more scarring than normal animals, even though their body weight and overall metabolism were similar. This pointed to a specific role for liver FGF1 in preventing fat from clogging the liver over the long term.
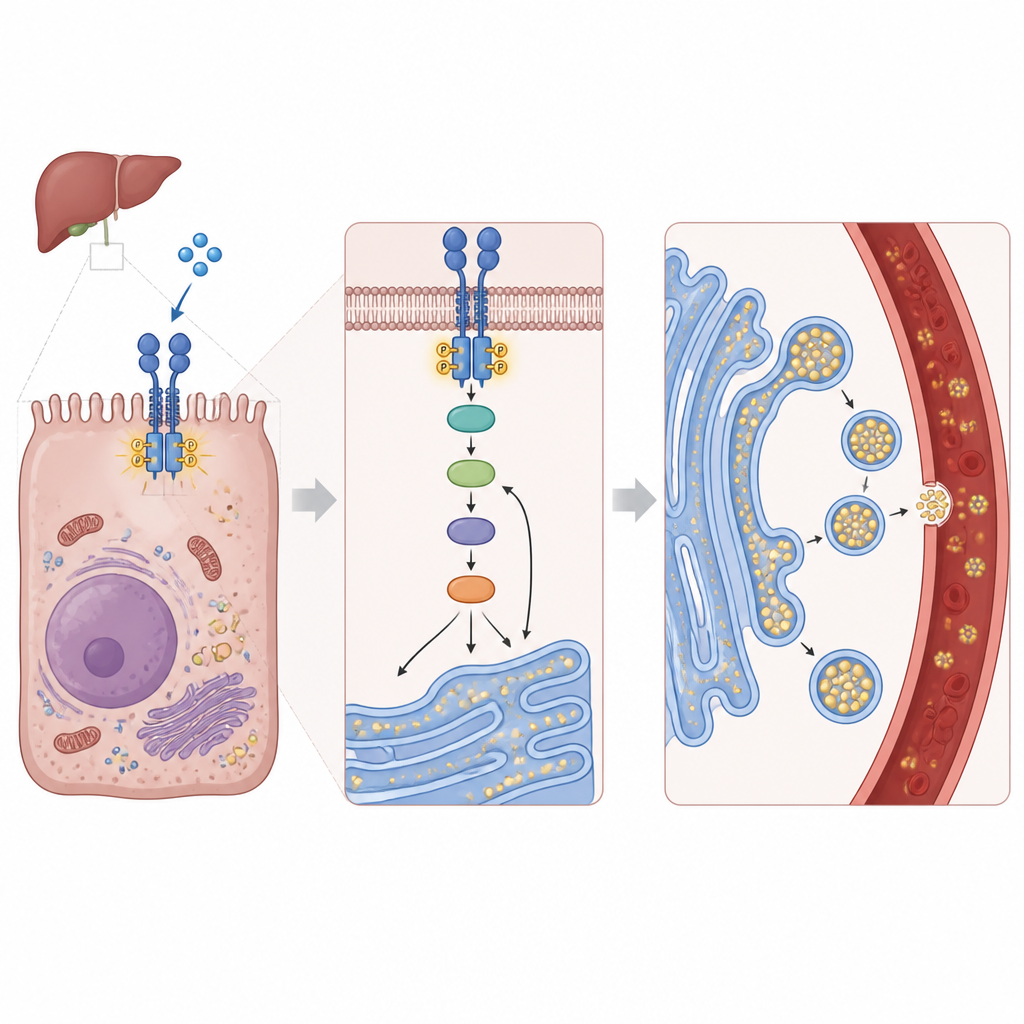
How liver cells turn FGF1 into action
Digging deeper, the researchers traced how FGF1’s signal travels inside liver cells. FGF1 binds to a surface receptor called FGFR4, which kicks off a chain reaction through well-known signaling proteins, including PI3K, AKT, and a growth-control hub called mTORC1. This, in turn, activates a quality-control system in a cell compartment called the endoplasmic reticulum, where fat particles are built. A key player there is IRE1, which, when switched on, processes another protein called XBP1. The activated XBP1 form then boosts the machinery that helps assemble and secrete fat-loaded particles. Remarkably, the team found that FGF1 triggers IRE1 in an unusual way that does not rely on its classic stress-sensing domain, allowing liver cells to ramp up fat export without broadly turning on a full-blown stress response.
Restoring the rhythm to protect the liver
The study also explored whether extra FGF1 could help in disease-like states. In mouse models that mimic human fatty liver and early steatohepatitis, treatment with FGF1 increased triglyceride release from the liver, reduced liver fat content, and eased signs of inflammation and scarring. Blocking FGFR4, IRE1, XBP1, or mTORC1 erased these benefits, underscoring that this signaling route is essential for FGF1’s protective effects. Together, the findings show that FGF1 serves as a timekeeping messenger that tells the liver when to ship out fat, helping match internal fat traffic with daily activity.
What this means for future liver care
For non-specialists, the takeaway is that fatty liver disease is not only about how much fat we eat or make, but also about when the liver moves that fat out. FGF1 appears to be a key part of the liver’s own clock-controlled system that keeps fat flowing at the right time of day. If this timing breaks down, fat lingers in the liver and damage accelerates. By learning how to support or gently tune this FGF1-driven rhythm, future therapies may help clear liver fat and slow disease progression while respecting the body’s natural daily cycles.
Citation: Sermikli, B.P., Liu, S., Kim, K. et al. FGF1 orchestrates circadian hepatic triglyceride secretion. Nat Commun 17, 4241 (2026). https://doi.org/10.1038/s41467-026-70849-7
Keywords: circadian rhythm, fatty liver, triglycerides, FGF1, liver metabolism